
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 474
Title
Succinate dehydrogenase inactivation leads to cancer by promoting EMT
Short name
Graphical Representation
Point of Contact
Contributors
- Sylvie Bortoli
- Xavier COUMOUL
- Arnaud TETE
- Karine Audouze
Coaches
- Rex FitzGerald
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.106 | Under Development |
This AOP was last modified on April 08, 2026 12:25
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Succinate dehydrogenase, inhibited | May 04, 2023 11:49 |
| Increase, DNA methyltransferase inhibition | April 22, 2019 05:09 |
| Epithelial Mesenchymal Transition | April 24, 2024 00:44 |
| Succinate Accumulation | July 18, 2024 13:24 |
| Increase, Cancer | August 22, 2023 14:32 |
| SDH, inhibited leads to Succinate Accumulation | July 18, 2024 13:50 |
| Succinate Accumulation leads to Increase, DNMT inhibition | July 18, 2024 13:50 |
| Increase, DNMT inhibition leads to EMT | July 18, 2024 13:51 |
| EMT leads to Increase, Cancer | July 18, 2024 13:52 |
| Boscalid | June 10, 2024 10:27 |
| Bixafen | June 10, 2024 10:27 |
| Sedaxane | June 10, 2024 10:29 |
Abstract
Succinate dehydrogenase (SDH) is a key enzymatic complex involved in two interconnected metabolic processes for energy production: the transfer of electrons in the mitochondrial respiratory chain and the oxidation of succinate to fumarate in the Krebs cycle. In humans, inherited SDH deficiencies may cause major pathologies including cancers. The cellular and molecular mechanisms related to genetic SDH inactivation have been well described in neuroendocrine tumors, in which it induces an oxidative stress, a pseudohypoxic phenotype, a metabolic, epigenetic and transcriptomic remodeling, and alterations in the migration and invasion capacities of cancer cells, in connection with the accumulation of succinate, an oncometabolite, substrate of the SDH. SDH complex is the molecular target of Succinate Dehydrogenase Inhibitors (SDHi), a family of pesticides widely used to limit the proliferation of pathogenic fungi. This AOP aims to describe the relationship between SDH inactivation and cancer development (PMID: 37778286).
AOP Development Strategy
Context
Succinate dehydrogenase (SDH) is a key enzyme of mitochondria, organelles that play a crucial role in the production of energy, the metabolic and calcium homeostasis, the control of apoptosis, and the production of reactive oxygen species. SDH is involved in two interconnected metabolic processes for energy production: 1) cellular respiration, where it allows the transfer of electrons to ubiquinone as complex II of the mitochondrial respiratory chain, and 2) the Krebs cycle, where it catalyzes the oxidation of succinate to fumarate (PMID: 33112834)
Numerous studies show that a complete inactivation of SDH caused by a first constitutional mutation associated with a second somatic mutation, leads to cancerous pathologies in young adults, including particularly aggressive forms of cancer such as paragangliomas (neuroendocrine tumors of the head and neck, thorax, abdomen and pelvis), pheochromocytomas (tumors of the adrenal medulla), renal cancers and gastrointestinal stromal tumors. The cellular and molecular mechanisms related to the genetic inactivation of SDH have been well described in neuroendocrine tumors, where it induces an oxidative stress, a pseudohypoxia phenotype, a metabolic, epigenetic and transcriptional remodeling, and alterations in tumor cell migration and invasion capacities, in connection with the accumulation of succinate, the substrate of SDH (PMID: 40285898).
The succinate dehydrogenase inhibitors (SDHi) are fungicides used to control the proliferation of pathogenic fungi in cereal, fruit and vegetable crops, with a mode of action based on blocking the activity of SDH. The analysis of literature data shows that the impact of SDHi on health remains largely unexplored to date, despite a growing number of studies reporting toxic effects in non-target organisms (PMID: 37778286). This is supported by our recent work highlighting 1) the high degree of conservation of the SDH catalytic site (i.e. the SDHi binding site) during the evolution and 2) the ability of SDHi to inhibit SDH in the mitochondria of non-target species, including humans (PMID: 31697708). These observations show that SDHi are not specific to fungal SDH and that their use may present a risk to human health, particularly in the context of chronic exposure through the diet. Moreover, the analysis of regulatory assessment reports shows that most SDHi induce tumors in animals without evidence of genotoxicity. Thus, for these substances, the mechanisms of carcinogenicity are, to date, not clearly established.
Our hypothesis is that, if SDHi fungicides are able to alter SDH activity in humans, the consequences of SDHi exposure on cellular and mitochondrial functions may resemble those observed in SDH-mutated tumors and SDH-deficient cells. We assume that the development of an AOP deciphering the different steps leading to cancer following a genetically-SDH inactivation could help to propose the exploration of relevant key events and adverse effects upon chronic exposure to SDHi fungicides.
Strategy
The development strategy for this AOP is based on the current knowledge on molecular and cellular events triggered by a genetic inactivation of SDH, and on the hypothesis that a chemical SDH inactivation may lead to similar events.
This AOP will be part of the development of an AON with AOP 534.
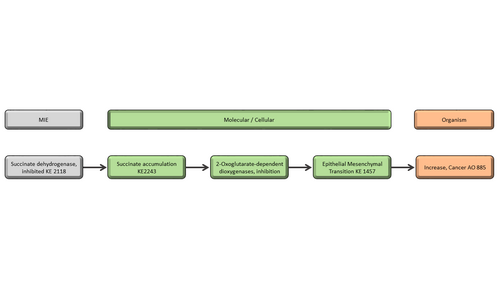
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2118 | Succinate dehydrogenase, inhibited | SDH, inhibited |
| KE | 2243 | Succinate Accumulation | Succinate Accumulation |
| KE | 1619 | Increase, DNA methyltransferase inhibition | Increase, DNMT inhibition |
| KE | 1457 | Epithelial Mesenchymal Transition | EMT |
| AO | 885 | Increase, Cancer | Increase, Cancer |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| SDH, inhibited leads to Succinate Accumulation | adjacent | High | High |
| Succinate Accumulation leads to Increase, DNMT inhibition | adjacent | High | High |
| Increase, DNMT inhibition leads to EMT | adjacent | Moderate | Moderate |
| EMT leads to Increase, Cancer | adjacent | High | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Adult | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| human and other cells in culture | human and other cells in culture | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | Moderate |
Overall Assessment of the AOP
This AOP describes the molecular and cellular consequences of succinate dehydrogenase (SDH) inactivation, leading to adverse biological outcomes specifically cancer. It integrates evidence from genetic and chemical inhibition models, supporting the hypothesis that chemical inactivation of SDH can mimic the effects observed in genetic SDH deficiencies.
This AOP is broadly applicable across multiple taxa, as SDH is a highly conserved enzyme among eukaryotes. The key events have been observed in mammalian models (rodents, humans) as well as in some invertebrate systems, though most empirical evidence comes from vertebrate studies. The AOP is relevant to both sexes, as SDH-related dysfunctions have not shown sex-specific susceptibility.
Regarding life stage, available data suggest that embryonic and juvenile stages may be more vulnerable to SDH inactivation due to their higher metabolic demands and reliance on mitochondrial function. However, further studies are needed to delineate life-stage-dependent sensitivities.
The overall weight of evidence for this AOP is moderate to strong, with well-established mechanistic understanding of early molecular events but some uncertainties in later-stage key event relationships. - Biological plausibility: Strong, as SDH inactivation is well-documented to disrupt mitochondrial metabolism, leading to accumulation of oncometabolites (e.g., succinate) and altered cellular signaling. SDH inactivation is clearly associated with the occurence of multiple cancers. - Empirical support: Moderate to strong, particularly for the initial key events. In vitro and in vivo studies demonstrate consistency in key event progression, but quantitative understanding of dose-response relationships remains limited. - Concordance and consistency: Strong, with observed reproducibility across multiple experimental models, despite moderate species-specific differences.
Regulatory Considerations This AOP contributes to a broader Adverse Outcome Network (AON) alongside AOP 534, providing a mechanistic basis for regulatory applications such as: - Priority setting: identifying chemicals that may interfere with SDH function as potential environmental and occupational hazards. This is the case for SDHi fungicides. - Testing strategies: informing the development of in vitro assays targeting early key events to reduce reliance on animal models (SDH activity) - Risk assessment: Supporting the integration of mitochondrial toxicity (mitotoxicity) into chemical risk assessment frameworks, particularly for compounds suspected of disrupting cellular metabolism.
Domain of Applicability
This AOP is applicable across multiple biological domains, considering its relevance to various taxa, sexes, life stages, and broader biological contexts. The key molecular and cellular events triggered by SDH inactivation are largely conserved across eukaryotes, making this AOP broadly applicable.
Sex Applicability This AOP is considered applicable to both sexes, as no significant sex-specific differences have been reported in SDH inactivation studies. While hormonal and metabolic differences could theoretically modulate susceptibility in certain contexts, available data do not indicate a strong sex-dependent effect. Further research may be needed to assess potential sex-specific variations in sensitivity.
Life Stage Applicability The consequences of SDH inactivation are relevant across multiple life stages, but susceptibility may vary. - Early developmental stages (embryonic, juvenile): more vulnerable due to higher metabolic demands and reliance on mitochondrial function for growth and differentiation. Disruptions in energy metabolism during early development may lead to severe consequences, including impaired organogenesis.
- Adult stage: the impact of SDH inhibition is likely tissue-specific, with high-energy-demand organs (e.g., brain, heart, muscle) being particularly affected. While metabolic plasticity in adults may provide some resilience, chronic SDH inhibition can still lead to progressive dysfunction, particularly in tissues with limited regenerative capacity.
- Vulnerable stage: furthermore, the invalidation or inhibition of SDH in already vulnerable populations is likely to aggravate their pathophysiological condition. Furthermore, the invalidation or inhibition of SDH in already vulnerable populations is likely to aggravate their pathophysiological condition.
Taxonomic Applicability SDH is an essential enzyme in the mitochondrial electron transport chain, highly conserved across eukaryotic species. Strong empirical evidence supporting this AOP comes from mammalian models (rodents, humans), where SDH inactivation has been linked to metabolic dysregulation and disease. Additionally, SDH impairment has been studied in invertebrate models (Drosophila melanogaster, Caenorhabditis elegans), demonstrating similar disruptions in mitochondrial function and cellular signaling (https://doi.org/10.1016/j.jmb.2009.02.028 ; PMID: 24954416). While fundamental mechanisms are preserved across species, variations in metabolic compensation and stress response pathways may influence the severity of downstream effects in different taxa.
Other Biological Contexts - Cell and tissue specificity: the effects of SDH inactivation are particularly relevant in metabolically active tissues such as the nervous system, cardiac and skeletal muscles, and endocrine organs, where mitochondrial function is critical for normal physiological processes.
- Physiological and pathological conditions: this AOP is especially relevant in the context of mitochondrial diseases, neurodegenerative disorders, and cancer, where SDH dysfunction has been implicated. The findings may also have implications for toxicological assessments of environmental and pharmaceutical compounds targeting mitochondrial function.
Essentiality of the Key Events
The essentiality of each Key Event (KE) was assessed based on experimental evidence demonstrating whether its manipulation (e.g., inhibition, knockdown, overexpression, or supplementation) prevents the occurrence of subsequent KEs and/or the final Adverse Outcome (AO).
In this AOP, the Molecular Initiating Event (MIE) is the inhibition of succinate dehydrogenase (SDH, KE 2118), which leads to succinate accumulation (KE2243), inhibition of DNA methyltransferase (DNMT) activity (KE 1619), and activation of epithelial–mesenchymal transition (EMT, KE 1457). Strong evidence supports the essentiality of SDH inhibition and EMT activation, while succinate accumulation and DNMT inhibition are also supported by experimental data, although the direct causal links have varying strength depending on the studies. Overall, the evidence suggests that preventing any of these KEs can significantly alter the progression toward the AO.
| Key Event (KE) | Essentiality Level | Evidence Summary | References |
|---|---|---|---|
| KE 2118 - SDH inhibited | Strong | The SDH complex uses succinate as a substrate. Invalidation, genetic knockdown or inhibition of SDH leads to succinate accumulation. Restoration of SDH activity normalizes mitochondrial function and blocks epigenetic and phenotypic changes, leading to EMT. | PMID: 40359599; PMID: 33112834 |
| KE 2243 - Succinate accumulation | Moderate | Exogenous succinate administration can induce epigenetic alterations and EMT markers in some in vitro models, but effects are partial and context-dependent. Direct suppression of succinate accumulation to block downstream KEs is less frequently reported. | REF |
| KE 1619 - Increase, DNA methyltransferase inhibition | Strong | Elevated succinate inhibits α-KG-dependent dioxygenases (including TET enzymes), resulting in altered DNA methylation patterns. DNMT inhibition or knockdown experimentally reproduces these patterns and related gene expression changes. Conversely, DNMT overexpression or α-KG supplementation can attenuate these epigenetic effects. | REF |
| KE 1457 - Epithelial Mesenchymal Transition (EMT) | Strong | Pharmacological or genetic inhibition of EMT-associated transcription factors (e.g., SNAI1, TWIST) blocks tumor progression and cell migration, even in the presence of upstream perturbations such as SDH inhibition or succinate accumulation. | REF |
Evidence Assessment
KER1: SDH inhibited (KE 2118) → Succinate accumulation (KE 2243) Strong. SDH catalyzes oxidation of succinate to fumarate in the TCA cycle; loss or inhibition of SDH predictably causes intracellular succinate build-up. This is well supported in SDH-deficient tumors and models (REF)
KER2: Succinate accumulation (KE 2243) → Increase, DNA methyltransferase (DNMT) inhibition (KE 1619) Mixed. Robust evidence shows succinate competitively inhibits α-KG–dependent dioxygenases (e.g., TET DNA demethylases, Jumonji histone demethylases), leading to DNA hypermethylation in SDH-deficient tumors. However, this mechanism reflects TET inhibition, not DNMT inhibition. Direct evidence that succinate inhibits DNMT enzymes is limited and, mechanistically, DNMT inhibition would be expected to favor hypomethylation, opposite to what is seen in SDH-deficient contexts. Thus, the biological plausibility is strong for “succinate → inhibition of α-KG–dependent demethylases (e.g., TET) → hypermethylation,” but weak/inconsistent for “succinate → DNMT inhibition.”
KER3: Increase, DNMT inhibition (KE 1619) → Epithelial–Mesenchymal Transition (EMT, KE 1457) Moderate/conditional. EMT programs are influenced by epigenetic remodeling (e.g., methylation at CDH1 and other epithelial genes). There is ample evidence that increased DNA methylation promotes EMT and invasion, and that epigenetic drugs can modulate EMT. But again, most mechanistic support involves hyper-methylation via TET inhibition or DNMT activity supporting EMT—whereas “DNMT inhibition” would usually counter hypermethylation. Therefore, the general epigenetic control → EMT is biologically plausible, but the directionality specified by KE 1619 (“DNMT inhibition”) is poorly aligned with the SDH/succinate epigenetic signature.
(Non-adjacent reinforcement): Independent lines link SDH loss/succinate accumulation to EMT-like phenotypes via pseudohypoxia (PHD/HIF), ROS, and epigenetic routes; exogenous succinate can enhance EMT-related functions in several models.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|