
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 567
Title
Binding to plastoquinone B site leading to decreased population growth rate via photosystem II inhibition
Short name
Graphical Representation
Point of Contact
Contributors
- Li Xie
- Knut Erik Tollefsen
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 10, 2026 14:47
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Binding of plastoquinone B (QB) within D1 protein of Photosystem II | February 04, 2026 06:58 |
| Decrease, Photosystem II efficiency | January 16, 2026 04:26 |
| Decrease, Photosynthesis | January 20, 2026 03:44 |
| Decreased, mitochondrial oxidative phosphorylation | February 04, 2026 03:52 |
| Decrease, ATP production | February 04, 2026 05:16 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| Decrease, Growth | July 06, 2022 07:36 |
| Binding to the QB site D1 protein leads to Decrease, Photosystem II efficiency | February 23, 2026 08:10 |
| Decrease, Photosystem II efficiency leads to Decrease, Photosynthesis | February 23, 2026 07:43 |
| Decrease, Photosynthesis leads to Decrease in mitochondrial oxidative phosphorylation | February 23, 2026 08:46 |
| Decrease in mitochondrial oxidative phosphorylation leads to Decrease, ATP production | February 26, 2026 07:50 |
| Decrease, ATP production leads to Decrease, Growth | April 10, 2026 08:16 |
| Decrease, Growth leads to Decrease, Population growth rate | October 08, 2020 08:17 |
| Diuron | May 24, 2018 15:29 |
| atrazine | November 29, 2016 18:42 |
| Metribuzin | February 23, 2026 08:51 |
| Chlorotoluron | February 23, 2026 08:52 |
Abstract
Photosystem II (PSII) is essential for photosynthesis in primary producers, facilitating the primary photochemical reaction to oxidize water and drive the production of ATP and NADPH. A critical interaction within PSII occurs at the plastoquinone B (QB) site on the D1 protein, where electron transfer from QA to QB ensures the continuity of photosynthetic electron flow and energy transduction. PSII-inhibitors can competitively bind to this QB site, blocking electron flow and suppressing the production of ATP and NADPH in chloroplasts. This disruption constrains downstream processes at different biological levels and consequently result in growth inhibition. PSII inhibitors are frequently detected in surface and ground waters, especially in agricultural regions. Their prolonged mobility results in chronic exposure of non-target organisms, particularly primary producers such as algae, aquatic macrophytes, and terrestrial plants that share conserved QB binding sites with target weeds. To improve mechanistic understanding and strengthen ecological risk assessment of PSII-inhibitor, such as herbicides, AOP #567 was developed. This AOP delineates a linear cascade of key events, beginning with the binding of PSII inhibitors to the QB site on the D1 protein, leading through successive reductions in PSII efficiency, photosynthesis, mitochondrial OXPHOS, and ATP production, and culminating in decreased growth rates of primary producer populations. By systematically linking molecular interactions to population-level outcomes, AOP #567 provides a transparent and biologically plausible framework to assess the environmental hazards posed by PSII inhibitors.
AOP Development Strategy
Context
Photosynthesis is the fundamental biological process to convert solar energy into chemical energy and stored as carbohydrates in plants, algae, and cyanobacteria. It aids energy needs of both terrestrial and aquatic ecosystems. Being at the centre of the photosynthesis, photosystem II (PSII) performs the most important photochemical reaction through light capture resulting in oxidation of the water molecules releasing oxygen, protons and electrons (Chen et al., 2007). Such electrons are then passed on through the photosynthetic electron transport pathway, and eventually to the generation of ATP and NADPH. In a situation within the PSII complex, a momentous interaction that is in place is the plastoquinone B (QB) interaction capsule with the D1 protein, a pivotal location which receives electrons at the plastoquinone A (QA) (Ohad and Hirschberg, 1992). This is an essential electron transfer between QA and QB which is essential to the constancy of electron flow and energy transduction in photosynthesis (Vermaas, Renger and Arntzen, 1984). The binding site of the QB is common among herbicidal molecular targets because it is the key protein to electron transport (Fuerst and Michael, 1991).
PSII-inhibiting herbicides exert their phytotoxic effects by competitively binding to the QB site on the D1 protein. This binding disrupts the natural electron flow between QA and QB that stops electron flow in photosynthesis apparatus. This blockade direct suppressed the production of ATP and NADPH in chloroplast, which has serious impact on the followed metabolic process, such as such as carbon fixation and carbohydrate synthesis (Wilkinson et al., 2015). Reduced amount of generated carbohydrates output results in shorter amount of photosynthate to serve as mitochondrial oxidative phosphorylation (OXPHOS) substrate and consequently less ATP formation via the respiratory pathway (Hanson et al., 2023). At the whole-plant scale, about 50 percent of carbon dioxide fixed through photosynthesis is usually respired (Amthor and Baldocchi, 2001). Thus, constriction of carbon assimilation achieved by PSII inhibitors magnifies the cellular energy deficiency by restraining not only photosynthetic but as well as respiratory ATP synthesis. This deconvoluted energy deficiency is the root cause of metabolism malfunction and major reduction of growth in PSII-stressed plants.
Although useful as agricultural compounds, PSII inhibitors have elicited extreme environmental concerns considering how chemically stable they are, moderately water soluble, and resistance to breakdown and movement throughout the ecosystem (Brock, Lahr and Van den Brink, 2000). These chemicals are often found in surface and ground water, especially in agriculture areas as it is introduced through run off and leaching. Their prolonged environmental persistence and mobility mean that they can affect non-target organisms in aquatic and terrestrial habitats long after application. Primary producers, such as algae, aquatic macrophytes and terrestrial plants, are particularly susceptible to PSII herbicides, since they contain highly conserved QB binding sites the same as the targeted weed species. Even low environmentally relevant concentrations can negatively affect photosynthetic efficiency, growth rates and normal primary productivity (Oettmeier, 1999). These effects may cascade through ecosystems and change species composition, community structures, and stability and functioning of whole ecosystems. To improve mechanistic insight and risk assessment of herbicides that inhibit photosystem II, AOP#567 (Binding of plastoquinone B causing a lowered population growth rate through a reduced population photosystem II efficiency) was developed to illustrate how the binding of PSII inhibitor to the QB site of the D1 protein interferes with photosynthetic electron transport and leads to a decreased growth rate of populations on primary producers.
Strategy
The development of AOP 567 followed the guidance outlined in the OECD Users’ Handbook Supplement to the Guidance Document for Developing and Assessing AOPs (OECD, 2018). The pathway was constructed using a structured, evidence-based, and transparent process integrating systematic literature review, expert consultation, and formal weight-of-evidence (WoE) evaluation consistent with modified Bradford Hill considerations.
3.1 Problem Formulation and AOP Scoping
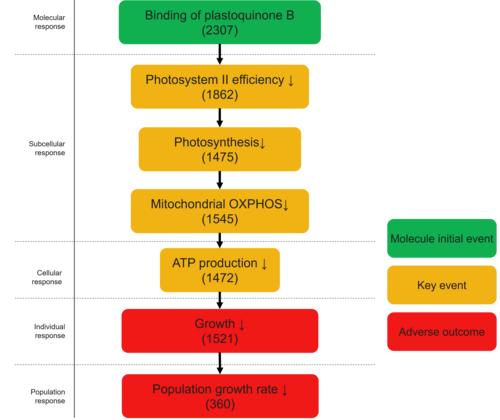
The AOP was initiated based on the well-characterised mode of action of PSII-inhibiting chemicals that interact with the Q_B binding niche of the D1 protein. The molecular initiating event (MIE) was defined as interference at the Q_B site within PSII. Downstream key events (KEs) were sequentially identified based on established biological continuity: decreased PSII efficiency, reduced photosynthesis, impaired mitochondrial oxidative phosphorylation (OXPHOS), decreased ATP production, and reduced population growth rate. These relationships correspond to KER#3556, KER#2333, KER#3557, KER#3558, and KER#3559.
3.2 Systematic Literature Identification and Screening
A tiered and reproducible literature screening pipeline was applied. The AOP-helpFinder (v3.0) was used to screen bibliographic datasets for stressor–event and event–event relationships. Swift-Reviewer (Version 1.43.1063) was subsequently used to refine the evidence corpus through structured tagging, relevance scoring, and confidence ranking. This combined approach ensured traceability and reproducibility in evidence selection while reducing manual bias.
3.3 Weight-of-Evidence Evaluation
The data extraction and assessment were done according to a weight-of-evidence (WOE) methodology in resonance to the modified Bradford Hill requirements (Becker et al., 2015; Collier et al., 2016). In particular, each key event relationship (KER) was evaluated by: (3) Essentiality of individual key events (KEs); (2) biological plausibility in that it is based on known photosynthetic and bioenergetic principles; (3) empirical evidence such as the dose response, temporality, and occurrence of effects.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2307 | Binding of plastoquinone B (QB) within D1 protein of Photosystem II | Binding to the QB site D1 protein |
| KE | 1862 | Decrease, Photosystem II efficiency | Decrease, Photosystem II efficiency |
| KE | 1475 | Decrease, Photosynthesis | Decrease, Photosynthesis |
| KE | 1545 | Decreased, mitochondrial oxidative phosphorylation | Decrease in mitochondrial oxidative phosphorylation |
| KE | 1472 | Decrease, ATP production | Decrease, ATP production |
| AO | 1521 | Decrease, Growth | Decrease, Growth |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Binding to the QB site D1 protein leads to Decrease, Photosystem II efficiency | adjacent | High | Low |
| Decrease, Photosystem II efficiency leads to Decrease, Photosynthesis | adjacent | High | High |
| Decrease, Photosynthesis leads to Decrease in mitochondrial oxidative phosphorylation | adjacent | Moderate | Moderate |
| Decrease in mitochondrial oxidative phosphorylation leads to Decrease, ATP production | adjacent | High | High |
| Decrease, ATP production leads to Decrease, Growth | adjacent | High | Moderate |
| Decrease, Growth leads to Decrease, Population growth rate | adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
Overall Assessment of the AOP
The present AOPdescribes the potential causal events initiated by the binding of photosystem II (PSII) inhibitors to the plastoquinone B (QB) site of the D1 protein within the PSII complex (MIE, event 1975), leading to a decrease in population growth rate in primary producers (adverse outcome, event 2181) via a cascade of intermediate key events (KEs), including inhibition of PSII electron transport (event 1976), decrease in PSII efficiency (event 1977), and ultimately, decrease in growth rate (event 2031). The Table 1 provides a summary of the KEs that constitute the AOP and the representative methods for measurement of each KE.
The MIE constitutes the competitive binding of PSII inhibitors to the QB site, which disrupts the transfer of electrons between QA and QB. This blockage has a direct inhibitory impact on the functioning of the PSII complex and causes the first downstream KE: the inhibition of PSII efficiency, which is typically reported as Fv/Fm ratio or ΦPSII. The lower PSII efficiency is an indication of reduced energy conversion in the light-dependent components of photosynthesis. As a logical physiological consequence, this leads to a decrease in carbon fixation and the availability of ATP and NADPH, both of which are essential for cellular growth. here is a consequent decrease in the rate of organismal growth, particularly on the taxa that grows fast like phytoplankton and aquatic plants.
This series of KEs represents the minimal essential path of causally linked events required to describe how a molecular-level interaction with a chemical stressor produces an ecologically relevant adverse outcome. Each KE and its connection to the adjacent events are evidenced by the mechanistic and empirical evidence, dose-response relationships, as well as taxonomic concordance, as it is elaborated in the section “Summary of scientific evidence assessment” below. Although the subsequent secondary effects, such as oxidative stress and thylakoid membrane damage, also can happen after the PSII disruption, they do not constitute a necessary part of this AOP but rather play a modifying role. The mechanisms of the reactions discussed are very well conserved and the events are consequently found in very wide range of photosynthetic organisms i.e. algae, cyanobacteria, and aquatic vascular plants. This linear AOP can be included in a network of herbicide-related inhibition of photosynthesis and downstream effects at the population level that can occur across primary producers.
|
Table 1. Summary of key events in AOP #567 and related measurement methods
|
Domain of Applicability
Taxonomic Applicability
Since the MIE (binding of an inhibitor to the QB site of the D1 protein) targets one of the most highly conserved components of the photosynthetic apparatus across taxa, AOP 567 is broadly applicable to all oxygenic photosynthetic organisms that contain Photosystem II (PSII), which included photosynthetic primary producers of many different taxonomic groups. The underlying mechanism, PSII inhibitor binding to D1 QB site, is conserved across these groups. Sensitivity differences reflect physiological traits (e.g., pigment composition, repair rates, metabolic flexibility) rather than differences in molecular target presence. Heterotrophic organisms lacking PSII are not directly affected, though they may experience indirect impacts through reduced primary productivity. For regulatory purposes, a representative set of test species (e.g., green alga, cyanobacterium, diatom, aquatic plant) can adequately capture the taxonomic domain of applicability for this AOP.
Higher plants (aquatic angiosperms). Aquatic vascular plants such as seagrasses (Halophila ovalis, Zostera muelleri) and freshwater macrophytes (e.g., Lemna gibba, Myriophyllum spicatum) rely on PSII for photosynthesis and are highly sensitive to PSII herbicides. In seagrasses, diuron or atrazine at 1–10 µg/L can cause ~50% inhibition of PSII yield within 72 h, with chronic exposures leading to growth and survival impacts. Laboratory tests with Lemna show frond multiplication inhibited at low tens of µg/L. The D1 protein structure in higher plants is essentially identical to that in algae, and resistance mutations (e.g., psbA Ser264→Gly in triazine-resistant weeds) confirm cross-taxa conservation of the binding site. While terrestrial plants are the intended targets of these herbicides, aquatic plants are affected via environmental exposure.
Diatoms and algae groups. Diatoms can be amongst the most sensitive taxa; Chaetoceros muelleri has a 72h reduced growth under 1.5 ug/l diuron exposure. There is clear PSII inhibition in cryptophytes( e.g., Rhodomonas salina; 72-h growth EC50 with 6 µg/L diuron). The structure of PSII is the same across rhodophytes and other macroalgae, except that the composition of other pigments can affect the scaling of repair and the sensitivity of repair.
Cyanobacteria. These prokaryotic phototrophs have PSII functionally equivalent to that in eukaryotes and are susceptible to PSII herbicides. Laboratory studies show inhibition of PSII activity and growth in species such as Nostoc, Aphanocapsa, and Aulosira (96-h atrazine EC₅₀ ~147 µg/L). Some cyanobacteria exhibit greater tolerance or can exploit ecological shifts (e.g., nitrogen fixation) to gain competitive advantage when co-occurring algae are suppressed, but direct effects occur at sufficiently high or prolonged exposures.
Other photosynthetic protists that contains PSII, Euglenophytes, dinoflagellates, and coral symbionts (Symbiodiniaceae) are also affected. For example, Symbiodinium spp. show significant declines in Fv/Fm at 1–3 µg/L diuron, contributing to coral bleaching events.
Sex and life stage
Sex is not a relevant domain of applicability for this AOP, as the key events are rooted in the universally conserved physiology of PSII and cellular energy metabolism.
All life stages that rely on functional photosynthesis are within the domain of applicability. Early developmental stages, such as germinating seeds, algal spores, or propagules of aquatic plants, may be particularly vulnerable due to their high energy demands and limited capacity for metabolic compensation (Amthor & Baldocchi, 2001).
Biological context:
The AOP is applicable under both laboratory and environmentally realistic exposure conditions. While higher doses are often used in controlled studies to establish mechanistic linkages, environmentally relevant, sublethal concentrations have been shown to impair PSII efficiency, photosynthesis, and growth in aquatic systems (Wilkinson et al., 2015; Hanson et al., 2023). Given the persistence and mobility of PSII inhibitors in surface and ground waters, chronic exposures in aquatic environments are particularly relevant.
Essentiality of the Key Events
Evidence for essentiality in an AOP refers to demonstrating that if an upstream key event is prevented or reversed, the subsequent downstream events do not occur (or normal recovery). In AOP 567, the essentiality of events were determined based on exposure-recovery experiments, the inhibition-rescue experiments as well as the mechanistic studies of aquatic plants and algae. The essentiality of the MIE (Event 2307: Binding of plastoquinone B to the D1 protein) is considered high, since this is the well characterised target site of most PSII-inhibitor herbicides. A number of reports show that the herbicide can be artificially removed or that replacement of the PSII acceptors by oxidants with alternative electron acceptors (e.g., decyl-plastoquinone) will allow restoration of the activity of PSII and avoid downstream consequences on photosynthesis and subsequent growth. Similarly, KE1 (Event 1862: Decrease in PSII efficiency) is also considered high in essentiality. Inhibition of PSII activity directly results in reduced quantum yield and electron transport, and restoration of PSII activity has been consistently associated with recovery of photosynthetic performance. There is strong empirical evidence showing that PSII efficiency (e.g., Fv/Fm, ΦPSII) strongly correlates with primary productivity in photosynthetic organisms. And recovery study on seagrass after exoposure to diuron indicated the ΔF/Fm′ and growth rate returned over few weeks after removal to clean water. The KE2 (Event 1472: Decrease in photosynthesis) is assigned to high essentiality as well. Studies in both algae and aquatic plants have shown that when photosynthesis is suppressed, growth rapidly declines, and normal function resumes when photosynthetic activity is restored. In contrast, KE3 (Event 1545: Decrease in mitochondrial oxidative phosphorylation) is considered to have moderate essentiality. While evidence indicates that mitochondrial ATP production is reduced following photosynthetic inhibition due to limited carbohydrate substrate availability, the capacity for partial compensation through glycolysis or alternative pathways reduces the strength of causal inference. Finally, KE4 (Event 1472: Decrease in ATP production) also holds moderate essentiality. Growth inhibition is mechanistically connected to a lack of ATP. However, few direct rescue studies, using ATP precursors or supply of energy, provide uncertainty.
Evidence Assessment
1. Biological Plausibility
Biological plausibility means the known structural, or functional correlations between upstream key event and downstream key event in unperturbed biology or normal conditions (OECD, 2018). Such basic knowledge forms what can be used to extrapolate or make a hypothesis of the probable effects of a biological perturbation induced by a stressor.
The biological plausibility of KER3556 (Binding of plastoquinone B leads to Decrease in Photosystem II efficiency) is considered high. Under normal photosynthetic conditions, plastoquinone at the QB site of Photosystem II (PSII) accepts electrons from QA and enables the continuation of electron flow through the thylakoid membrane. Binding PSII-inhibitor to the QB site blocks this electron transfer, directly impairing photochemical efficiency (Battaglino, Grinzato and Pagliano, 2021). Such a structure-function relationship is known well within plant and algal systems and forms the basis MoA of many commercial herbicides.
The biological plausibility of KER2333 (Decrease in PSII efficiency leads to Decrease in Photosynthesis) has been rated high as well. The effectiveness of PSII determines the ability of the plant to utilize the light available to generate biochemical energy by the process of ATP and NADPH synthesis and subsequently carbon fixation. Decreased energy transmission causes a downstream decrease in CO2 assimilation, and hence this relationship is well supported by both mechanistic knowledge and experimentation.
The biological plausibility of KER3557 (Decrease in Photosynthesis leads to Decrease in Mitochondrial OXPHOS) is considered high. Mitochondrial respiration in photosynthetic organisms relies on photosynthetically produced oxygen-dependent oxidative phosphorylation with carbohydrates. In normal physiological states, the functions of chloroplast and mitochondria are in highly coordinated patterns. A drop in photosynthetic carbon fixation diminishes the availability of substrate to the mitochondrial respiration, therefore restricting the OXPHOS capability. The interdependence in this functional nature is clearly proven in plots and algae systems and this offers a good mechanism to this relationship.
The biological plausibility is also high in the case of KER3558 (Decrease in Mitochondrial OXPHOS leads to Decrease in ATP production). In eukaryotes mitochondrial oxidative phosphorylation is the primary route of cellular ATP production and any impairment of the electron transport chain or proton gradient will directly reduce the efficiency of ATP synthesis. The structural and functional reliance of generation of ATP on OXPHOS is evolutionarily preserved, and as a result this is an extremely probable causative relationship.
The biological plausibility of KER3559 (Decrease in ATP production leads to Decrease in Population growth rate) is similarly high. ATP at the cellular level is important in facilitating energy-demanding functions of DNA replication, protein synthesis and cell division all of which are preparatory to the expansion of the population. Photoautotrophic organisms suffer with ATP deficits maintained, which suppresses the cell proliferation and lowers biomass gain. Mathematical prediction of ATP limitation of cellular and population level growth has been borne out by numerous experiments making this KE relationship quite convincing.
2. Empirical Support
Empirical support is moderate to high.
According to OECD AOP guidance, the empirical support of a key event relationship (KER) is determined based on evidence to illustrate dose-response consistency (the upstream KE is impacted at a concentration or dose equal to or lower than that which impacts the downstream KE), temporal concordance (the upstream KE occurs before the downstream KE is observed) and incidence concordance (the upstream KE is observed in an equal or greater proportion of the test population compared to the downstream KE). For AOP 567, empirical support for the KERs ranges from moderate to high, with particularly strong evidence for the early, photochemical stages.
The empirical evidence in support of KER3556 (Binding of plastoquinone B results in Decrease in photosystem II efficiency) is high, as many experiments on photosystem II (PSII) inhibitors (including the herbicides diuron, atrazine, and etc.) show instantaneous, dose-dependent reductions in PSII efficiency when the QB-binding site is occupied by an inhibitor. For instance, in isolated pea thylakoid membranes, QB site herbicides with higher inferred QB affinity (diuron, terbuthylazine, metribuzin) inhibited PSII electron transfer and fluorescence-based PSII performance at substantially lower concentrations than lower-affinity inhibitors (e.g., bentazon, metobromuron). Reported I50 values were around 7–8 × 10−8M for diuron and 1–2 × 10−7 M for terbuthylazine/metribuzin, derived from both DPIP photoreduction (PSII activity) and OJIP fluorescence (1–Vj, reflecting QA reduction and QB site interference). (Battaglino, Grinzato and Pagliano, 2021).
The empirical evidence in support of KER2333 (Decrease in PSII efficiency leads to Decrease in Photosynthesis) is high, as multiple studies demonstrate linear or proportional correlations between PSII inhibition (e.g., reduced Fv/Fm or ΔF/Fm′) and declines in net photosynthetic rate, CO2 fixation, or overall photosynthetic efficiency. Dose-dependent reductions in photosynthesis follow PSII blockage, with EC50 values often aligning closely with those for PSII efficiency (e.g., diuron causing ~50% Pn reduction at ~1 µM in algae; terbuthylazine EC50 ≈0.3 µM in sugar beet). Temporal evidence shows photosynthetic decline occurring shortly after PSII inhibition, typically within 2–4 hours, with chronic exposure amplifying effects through photodamage and ROS accumulation (Morin et al., 2018). In freshwater biofilms, diuron induced rapid photosynthesis inhibition (t1/2 for 50% inhibition ranging from <30 s at high doses to ~7 min at lower doses), directly linking PSII blockage to downstream photosynthetic output (Morin et al., 2018). This linkage holds robustly in microalgae (e.g., Chlorella, Scenedesmus), seagrasses, and higher plants via OJIP kinetics and gas exchange data.
The empirical evidence in support of KER3557 (Decrease in Photosynthesis leads to Decrease in Mitochondrial OXPHOS) is moderate, primarily inferred from aquatic models with limited direct data in terrestrial plants. Photosynthetic inhibition restricts carbohydrate substrate supply, reducing NADH/FADH2 availability for mitochondrial respiration and leading to decreased oxygen consumption rates (e.g., 30–50% reduction at 1 µM diuron in Chlamydomonas reinhardtii). Temporal patterns indicate mitochondrial effects follow photosynthetic decline, measurable within 6–12 hours and intensifying under chronic exposure (days–weeks) due to persistent energy deficits (Seloto et al., 2024). Bioenergetic principles support this cascade in algae, with analogous inferences for terrestrial species (e.g., Arabidopsis thaliana), though direct terrestrial measurements remain sparse.
The empirical evidence in support of KER3558 (Decrease in Mitochondrial OXPHOS leads to Decrease in ATP production) is moderate, based on studies showing direct proportionality between reduced OXPHOS and ATP depletion. Dose-response data indicate ATP levels decline with OXPHOS impairment (e.g., ~40% reduction at 1 µM diuron in Chlorella; EC50 ≈0.5 µM for diuron in algae), as diuron inhibits photochemical (but not oxidative) ATP synthesis, while related compounds affect both pathways (John, 1971). Temporal dynamics reveal ATP depletion following OXPHOS reduction within 12–24 hours in aquatic models, with sustained exposure causing severe cellular energy shortages. This linkage is conserved across mitochondrial bioenergetics in algae and inferred for terrestrial plants (e.g., Pisum sativum, Arabidopsis thaliana), though direct evidence in plants is limited (Seloto et al., 2024).
The empirical evidence in support of KER3559 (Decrease in ATP production leads to Decrease in Population growth rate) is high, as ATP depletion limits energy for cell division, growth, and reproduction, resulting in clear dose-dependent reductions in population growth rates across primary producers. Dose-response curves show consistent EC50 values for growth inhibition (e.g., diuron 50% inhibition in Chlorella vulgaris at ≈0.5 µM; terbuthylazine ≈0.4 µM in diatoms; diuron EC50 3.1–206 µg/L in marine microalgae like Tisochrysis lutea and Tetraselmis sp.) (Flores et al., 2024). Temporal evidence indicates growth declines lag ATP reduction, becoming significant after 24–48 hours in microalgae and weeks in terrestrial/macrophyte systems (e.g., reduced biomass, seed germination). Chronic exposures (weeks) severely restrict population expansion in aquatic primary producers (e.g., seagrasses, microalgae), with field-relevant diuron levels (e.g., 0.3 µg/L) causing sublethal PSII inhibition and growth suppression (Magnusson et al., 2008; Thomas et al., 2020).
Overall WoE Considerations
The AOP is biologically plausible and mechanistically conserved across primary producers. Dose-response and temporal concordance are strong for early KEs (MIE to photosynthesis) and moderate-to-strong for downstream linkages, supported by consistent patterns in dose- and time-resolved experiments (Morin et al., 2018; Battaglino et al., 2021). Evidence gaps remain for direct terrestrial plant data on mitochondrial/ATP KEs, warranting further targeted studies. This AOP supports predictive risk assessment for PSII herbicides in aquatic and terrestrial ecosystems.
Uncertainties, inconsistencies, and critical gaps
Overall, AOP 567 still constrains several uncertainties and knowledge gaps that need to be addressed to strengthen the reliability. First, for KER3557, the direct causal relationship between decreased photosynthesis (KE1472) and subsequent reduction in mitochondrial oxidative phosphorylation (OXPHOS; KE1545) remains not well-established. While it is logical that loss of chloroplast-derived ATP/NADPH due to PSII inhibition will lead to cellular energy deficits, empirical evidence for a direct impairment of mitochondrial function is limited. Studies on PSII-inhibiting herbicides (e.g., atrazine) have primarily documented chloroplast effects (reduced CO₂ fixation and chloroplast ATP synthesis) and oxidative stress in the chloroplast, with much less focus on mitochondria. Any mitochondrial impacts may occur only under prolonged or high exposures, if at all, introducing uncertainty in temporal and dose concordance for KER3557. In other words, mitochondrial dysfunction might lag behind the initial photosynthesis decline or require higher doses, rather than occurring in parallel at lower exposure levels. This gap in evidence leaves the Photosynthesis→OXPHOS linkage supported more by biological rationale than by consistent experimental data.. Additionally, Some studies on PSII inhibitors show no significant mitochondrial disruption, attributing growth declines solely to photosynthetic energy loss. For example, in certain algal species, ATP deficits are predominantly linked to chloroplastic ATP synthase inhibition rather than mitochondrial OXPHOS, creating inconsistencies with the proposed pathway. This may reflect species-specific differences or assay sensitivities, but it challenges the universality of the KER between photosynthesis decrease and mitochondrial OXPHOS impairment. Empirical data from population growth assays (e.g., in Lemna or Chlamydomonas) occasionally show non-monotonic responses, where low doses stimulate growth (hormesis) despite PSII efficiency drops, inconsistent with linear KER predictions. Such inconsistencies could arise from compensatory mechanisms like enhanced respiration, which are not fully accounted for in the AOP. The AOP is primarily applicable to photosynthetic eukaryotes (e.g., algae, aquatic plants), but gaps exist for non-photosynthetic taxa or prokaryotes, where plastoquinone analogs (e.g., ubiquinone in bacteria) might alter the MIE. Life stage-specific data are limited; juvenile stages may be more sensitive due to higher metabolic demands, but few studies address this. Sex applicability is generally not relevant for hermaphroditic or asexual species but requires exploration in dioecious plants. Important data gaps include the paucity of long-term studies linking the early key events (e.g., decreases in cellular ATP production) to the final adverse outcome (population growth decline) under realistic environmental conditions. Most evidence supporting this AOP comes from short-term laboratory exposures with constant conditions, whereas in nature, organisms experience fluctuating light, nutrient levels, and intermittent exposures. We lack chronic or multi-generation experiments that demonstrate a direct, quantitative progression from sustained ATP/OXPHOS deficits to measurable population-level effects (such as reduced reproduction or population growth rates) in photosynthetic organisms. Additionally, the AOP in its current form does not incorporate alternative or parallel pathways that could also lead to growth impairment. For instance, severe energy stress from PSII inhibition can trigger oxidative damage leading to cell death (e.g., via programmed cell death/apoptotic-like pathways in plants) or could impair other physiological functions like nutrient uptake and assimilation. These outcomes could contribute to growth and survival impacts independently of—or alongside—ATP shortage, but they are not accounted for in the linear sequence of key events. This represents a gap in the network completeness of the AOP. Addressing these uncertainties will require targeted research. Future studies should focus on experiments that probe the interaction between chloroplast and mitochondrial function under stress (to clarify the causality and timing in KER3557) and include a broader array of species (including different algal/plant taxa and life stages) to test the AOP’s generality. Conducting longer-duration exposures that monitor recovery or compounding effects over time would help establish whether short-term energy deficits indeed translate to long-term population declines. Moreover, incorporating emerging omics data is a promising avenue: transcriptomic and proteomic analyses under PSII-inhibitor exposure can reveal early biomarkers and compensatory changes (e.g., upregulation of respiratory genes or stress-response pathways), thereby refining our understanding of each key event and its quantitative relationship. Integrating such omics insights in future AOP updates could improve the weight of evidence for the KERs and support the development of predictive models for regulatory use.
Overall, these uncertainties, inconsistencies, and gaps highlight the need for caution when applying AOP 567 and point to specific areas (temporal concordance, dose-response shape, taxonomic scope, and additional pathways) where further research and data generation would greatly enhance the AOP’s robustness and utility in risk assessment.
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
| Light intensity | High irradiance can amplify the impact of PSII inhibition by increasing excitation pressure and ROS formation; low light may partially buffer translation from PSII efficiency decline to measurable reductions in photosynthesis. | KER#3556; KER#2333 |
| Temperature | Elevated temperatures can exacerbate PSII instability and D1 turnover, increasing sensitivity to Q_B inhibitors; low temperatures may slow metabolic propagation to mitochondrial processes. | KER#3556; KER#2333; KER#3557 |
| Nutrient availability (N, Fe) | Nutrient limitation reduces baseline photosynthetic capacity and mitochondrial activity, potentially increasing susceptibility to energy limitation or masking proportional declines. | KER#2333; KER#3557; KER#3558 |
| Species-specific photoprotective capacity (NPQ, cyclic electron flow) | Enhanced non-photochemical quenching or alternative electron sinks can mitigate the functional impact of PSII inhibition on net photosynthesis. | KER#3556; KER#2333 |
| Carbohydrate reserves | High intracellular carbohydrate storage may buffer short-term reductions in photosynthesis, delaying downstream OXPHOS and ATP decline. | KER#3557; KER#3558 |
| Mitochondrial respiratory flexibility | Alternative oxidase pathways or metabolic reprogramming may partially compensate for reduced substrate supply, moderating ATP decline. | KER#3557; KER#3558 |
| Exposure duration | Short exposures may produce transient PSII inhibition without measurable population-level consequences; chronic exposure increases likelihood of ATP-mediated growth reduction. | KER#3556; KER#2333; KER#3557; KER#3558; KER#3559 |
| Chemical potency and binding affinity at Q_B site | Higher affinity inhibitors produce PSII inhibition at lower concentrations, shifting the quantitative threshold for downstream effects. | KER#3556 |
| Life stage metabolic demand | Rapidly growing or reproducing stages have higher ATP demand and may exhibit earlier population-level consequences following ATP depletion. | KER#3558; KER#3559 |
| Environmental stress co-exposure (e.g., oxidative stressors) | Additional stressors that impair mitochondrial function can synergistically enhance ATP depletion and growth suppression. | KER#3557; KER#3558; KER#3559 |
Quantitative Understanding
The quantitative understanding of KER#3556 (Q_B binding → PSII efficiency) is considered low, because although target-site inhibition occurs at nanomolar concentrations (I₅₀ diuron ≈ 7–8 × 10⁻⁸ M in PSII assays; Battaglino et al., 2021), the quantitative relationship between fractional Q_B occupancy and functional decline in Fv/Fm has not been parameterised. The available data demonstrate high potency at the binding site, but there is no established response–response model linking molecular occupancy to photochemical efficiency reduction.
The quantitative understanding of KER#2333 (PSII efficiency → Photosynthesis) is considered high, because concordant EC₅₀ values have been reported between fluorescence-derived PSII endpoints and functional photosynthetic measures. In Rhodomonas salina, diuron produced an EC₅₀ of 1.71 µg/L for ΔF/Fm′ compared to 6.27 µg/L for growth (Thomas et al., 2020). Similarly, atrazine EC₅₀ values for fluorescence (232 µg/L) closely matched those for oxygen evolution (222 µg/L) (USEPA, n.d.). These datasets demonstrate strong quantitative alignment between reduced PSII efficiency and decreased photosynthetic performance, although compensatory non-photochemical quenching introduces variability across taxa.
The quantitative understanding of KER#3557 (Photosynthesis → Mitochondrial OXPHOS) is considered moderate, because limited simultaneous multi-endpoint studies are available. In Lemna minor, the effect dose rate for a 10% reduction in CO₂ uptake (EDR₁₀ = 2.8 mGy/h) was substantially lower than that for mitochondrial membrane potential (EDR₁₀ = 21.8 mGy/h) (Xie et al., 2019), indicating an approximate eightfold separation between upstream functional decline and downstream mitochondrial impairment. While this supports directional linkage, a fully parameterised quantitative response–response model has not yet been established.
The quantitative understanding of KER#3558 (Mitochondrial OXPHOS → ATP production) is considered moderate, because direct respiratory inhibition studies demonstrate clear concentration–response relationships. Rotenone inhibits Complex I with IC₅₀ values of approximately 5–20 nM and produces 40–80% ATP reduction, with nonlinear behaviour observed when OXPHOS inhibition exceeds 70–80% (Hatefi, 1985; Nicholls, 2004). However, within the context of AOP 567, the relevant mechanism is substrate-limited OXPHOS following reduced photosynthesis rather than direct electron transport inhibition, limiting direct quantitative extrapolation.
The quantitative understanding of KER#3559 (ATP production → Population growth rate) is considered moderate, because quantitative concordance between ATP depletion and growth inhibition has been demonstrated but not yet fully parameterised across the entire concentration range. In Chlamydomonas reinhardtii, the EC₅₀ for ATP reduction (0.16 µM) was lower than that for growth inhibition (0.41 µM), representing an approximately 2.6-fold separation (Nestler et al., 2012). Additionally, Bayesian modelling in Lemna minor provides a quantitative framework linking energetic impairment to demographic outcomes (Moe et al., 2021). Nevertheless, a fully integrated quantitative AOP model spanning all key events has not yet been developed.
Table 1. Summary of quantitative understanding for each KER in AOP 567
| KER | KE Upstream → KE Downstream | Rating | Key Quantitative Data | Limiting Factor |
| 3556 | QB binding → PSII efficiency | Low | I₅₀ diuron ≈ 7–8 × 10⁻⁸ M (PSII assays; Battaglino et al., 2021) | Fractional QB occupancy not coupled to Fv/Fm decline |
| 2333 | PSII efficiency → Photosynthesis | High | EC₅₀ diuron: 1.71 µg/L (ΔF/Fm′) vs. 6.27 µg/L (growth) in Rhodomonas salina (Thomas et al., 2020); atrazine EC₅₀ fluorescence 232 µg/L ≈ O₂ evolution 222 µg/L (USEPA, n.d.) | Compensatory NPQ varies across taxa |
| 3557 | Photosynthesis → Mitochondrial OXPHOS | Moderate | Lemna minor: EDR₁₀ CO₂ uptake = 2.8 mGy/h vs. EDR₁₀ MMP = 21.8 mGy/h (Xie et al., 2019) | Limited simultaneous multi-endpoint studies under PSII herbicide exposure |
| 3558 | Mitochondrial OXPHOS → ATP production | Moderate | Rotenone IC₅₀ Complex I ≈ 5–20 nM → 40–80% ATP reduction; nonlinear at >70–80% OXPHOS inhibition (Hatefi, 1985; Nicholls, 2004) | AOP-relevant mechanism is substrate-limited OXPHOS, not direct ETC inhibition |
| 3559 | ATP production → Population growth rate | Moderate | EC₅₀ ATP = 0.16 µM vs. EC₅₀ growth = 0.41 µM (~2.6-fold ratio) in C. reinhardtii (Nestler et al., 2012); Bayesian model for Lemna minor (Moe et al., 2021) | No fully parameterised qAOP model across the full concentration range |
Considerations for Potential Applications of the AOP (optional)
This AOP provides a mechanistically anchored framework linking PSII-targeting stressors to ecologically relevant growth outcomes and is particularly suitable for application in environmental hazard assessment of PSII-inhibiting herbicides and related compounds. Structural and functional evidence demonstrating Q_B site inhibition and suppression of PSII electron transport (Broser et al., 2011; Battaglino et al., 2021) supports its relevance for identifying chemicals acting through this conserved molecular target.
For regulatory applications, AOP 567 can inform mode-of-action classification, weight-of-evidence evaluations, and ecological risk assessment of plant protection products and other PSII-active chemicals. The mechanistic continuity from PSII inhibition to growth impairment aligns with population-level risk frameworks linking individual performance to population growth rate (Forbes and Calow, 2002; Kramer et al., 2011). The pathway may also support mixture risk assessment where multiple stressors converge on photosynthetic or energetic processes.
Limitations include environmental modulating factors such as light intensity and species-specific photoprotective mechanisms (e.g., NPQ), which may influence translation from PSII impairment to functional photosynthetic decline (Maxwell and Johnson, 2000). Additionally, while quantitative anchors exist for individual KERs, a fully parameterised qAOP model spanning the entire cascade has not yet been developed.
References
Alfonso, M., Pueyo, J.J., Gaddour, K., Etienne, A.-L., Kirilovsky, D., & Picorel, R. (1996). Induced new mutation of D1 serine-268 in soybean photosynthetic cell cultures produced atrazine resistance, increased stability of S₂QB⁻ and S₃QB⁻ states, and increased sensitivity to light stress. Plant Physiology, 112(4), 1499–1508.
Battaglino, B., Grinzato, A., & Pagliano, C. (2021). Binding properties of photosynthetic herbicides with the QB site of the D1 protein in plant Photosystem II: A combined functional and molecular docking study. Plants, 10(8), 1501.
Broser, M., Glöckner, C., Gabdulkhakov, A., Guskov, A., Buchta, J., Kern, J., Müh, F., Dau, H., Saenger, W., & Zouni, A. (2011). Structural basis of cyanobacterial photosystem II inhibition by the herbicide terbutryn. Journal of Biological Chemistry, 286(18), 15964–15972.
Chance, B., & Williams, G.R. (1955). Respiratory enzymes in oxidative phosphorylation. I. Kinetics of oxygen utilization. Journal of Biological Chemistry, 217, 383–393.
Chaube, R., et al. (2012). AMP-activated protein kinase and energy balance in fish. General and Comparative Endocrinology, 176, 366–374.
Delieu, T., & Walker, D.A. (1981). Polarographic measurement of photosynthetic oxygen evolution by leaf discs. New Phytologist, 89(2), 165–178.
Ermakova-Gerdes, S., & Vermaas, W. (1998). Mobility of the primary electron-accepting plastoquinone QA of Photosystem II in a Synechocystis sp. PCC 6803 strain carrying mutations in the D2 protein. Biochemistry, 37(17), 5918–5924.
Forbes, V.E., & Calow, P. (2002). Population growth rate as a basis for ecological risk assessment of toxic chemicals. Philosophical Transactions of the Royal Society B, 357, 1299–1306.
Gilbin, R., et al. (2008). Effects of chronic gamma irradiation on Daphnia magna. Journal of Environmental Radioactivity, 99, 134–145.
Graymore, M., Stagnitti, F., & Allinson, G. (2001). Impacts of atrazine in aquatic ecosystems. Environment International, 26(7–8), 483–495.
Hardie, D.G. (2011). AMP-activated protein kinase: An energy sensor that regulates all aspects of cell function. Genes & Development, 25(18), 1895–1908.
Hatefi, Y. (1985). The mitochondrial electron transport and oxidative phosphorylation system. Annual Review of Biochemistry, 54, 1015–1069.
Heckmann, L.H., et al. (2008). Reproduction recovery of Daphnia magna after chronic exposure to ibuprofen. Ecotoxicology, 17(3), 175–182.
Igamberdiev, A.U. (2023). Mitochondria in photosynthetic cells: Coordinating redox and energy metabolism. Plant Physiology, 191(4), 2104–2120.
Jones, R.J., Muller, J., Haynes, D., & Schreiber, U. (2003). Effects of herbicides diuron and atrazine on corals of the Great Barrier Reef, Australia. Marine Ecology Progress Series, 251, 153–167.
Jose, C., Bellance, N., & Rossignol, R. (2011). Choosing between glycolysis and oxidative phosphorylation: a tumor's dilemma? Biochimica et Biophysica Acta, 1807(6), 552–561.
Kortenkamp, A., Backhaus, T., & Faust, M. (2009). State of the Art Report on Mixture Toxicity. Report to the European Commission, Directorate General for the Environment.
Kramer, V.J., Etterson, M.A., Hecker, M., Murphy, C.A., Roesijadi, G., Spade, D.J., Spromberg, J.A., Wang, M., & Ankley, G.T. (2011). Adverse outcome pathways and ecological risk assessment: Bridging to population-level effects. Environmental Toxicology and Chemistry, 30(1), 64–76.
Kuruvilla, S., et al. (2003). Mechanistic and toxicokinetic data reduce uncertainty in the extrapolation of in vitro toxicity data. Toxicological Sciences, 76(1), 138–152.
Lambreva, M.D., et al. (2014). Structure/function/dynamics of Photosystem II plastoquinone binding sites. Current Protein & Peptide Science, 15(4), 332–345.
Macinnis-Ng, C.M.O., & Ralph, P.J. (2003). Short-term response and recovery of the seagrass Zostera capricorni to the herbicide diuron. Marine Environmental Research, 55(2), 153–166.
Maxwell, K., & Johnson, G.N. (2000). Chlorophyll fluorescence — a practical guide. Journal of Experimental Botany, 51(345), 659–668.
Milligan, A.J., Halsey, K.H., & Behrenfeld, M.J. (2015). Advancing interpretations of ¹⁴C-uptake measurements in the context of phytoplankton physiology and ecology. Journal of Plankton Research, 37(4), 692–698.
Mitchell, P. (1961). Coupling of phosphorylation to electron and hydrogen transfer by a chemiosmotic type of mechanism. Nature, 191, 144–148.
Moe, S.J., et al. (2021). Quantification of an adverse outcome pathway network by Bayesian regression and Bayesian network modeling. Integrated Environmental Assessment and Management, 17, 147–164.
Nestler, H., Groh, K.J., Schönenberger, R., Behra, R., Schirmer, K., Eggen, R.I.L., & Suter, M.J.F. (2012). Multiple-endpoint assay provides a detailed mechanistic view of responses to herbicide exposure in Chlamydomonas reinhardtii. Aquatic Toxicology, 110–111, 214–224.
Nicholls, D.G. (2004). Mitochondrial membrane potential and aging. Aging Cell, 3(1), 35–40.
Nicholls, D.G., & Ferguson, S.J. (2013). Bioenergetics 4. Academic Press.
Nieminen, A.L., Saylor, A.K., Tesfai, S.A., Herman, B., & Lemasters, J.J. (1994). Contribution of the mitochondrial permeability transition to lethal injury after exposure of hepatocytes to t-butylhydroperoxide. Biochemical Journal, 307, 99–106.
Noctor, G., De Paepe, R., & Foyer, C.H. (2007). Mitochondrial redox biology and homeostasis in plants. Trends in Plant Science, 12(3), 125–134.
Noguchi, K., & Yoshida, K. (2008). Interaction between photosynthesis and respiration in illuminated leaves. Mitochondrion, 8(1), 87–99.
OECD (2013). Guidance Document on Developing and Assessing Adverse Outcome Pathways. OECD Series on Testing and Assessment, No. 184. OECD Publishing, Paris.
OECD (2014). Guidance on Grouping of Chemicals, Second Edition. OECD Series on Testing and Assessment, No. 194. OECD Publishing, Paris.
OECD (2022). Uncoupling of Oxidative Phosphorylation Leading to Growth Inhibition via Decreased Cell Proliferation. OECD Series on Adverse Outcome Pathways. OECD Publishing, Paris.
Oettmeier, W. (1999). Herbicide resistance and supersensitivity in photosystem II. Cellular and Molecular Life Sciences, 55(10), 1255–1277.
Ohad, N., & Hirschberg, J. (1992). Mutations in the D1 subunit of Photosystem II distinguish between quinone and herbicide binding sites. Plant Cell, 4(3), 273–282.
Padmasree, K., Padmavathi, L., & Raghavendra, A.S. (2001). Essentiality of mitochondrial oxidative metabolism for photosynthetic performance in plant cells. Plant Physiology, 125(2), 617–626.
Parisot, F., Bourdineaud, J.P., Plaire, D., Adam-Guillermin, C., & Alonzo, F. (2015). DNA alterations and effects on growth and reproduction in Daphnia magna during chronic exposure to gamma radiation over three successive generations. Aquatic Toxicology, 163, 27–36.
Raghavendra, A.S. (1994). Interdependence of photosynthesis and respiration in plant cells. Photosynthesis Research, 38, 3–14.
Raghavendra, A.S., & Padmasree, K. (2003). Beneficial interactions of mitochondrial metabolism with photosynthetic carbon assimilation. Trends in Plant Science, 8(11), 546–553.
Song, Y., et al. (2020). Integrative assessment of low-dose gamma radiation effects on Daphnia magna reproduction: Toxicity pathway assembly and AOP development. Science of the Total Environment, 705, 135912.
Sundby, C., Chow, W.S., & Anderson, J.M. (1993). Effects on Photosystem II function, photoinhibition, and herbicide binding caused by mutation of the D1 protein. Photosynthesis Research, 36(2), 123–135.
Thomas, M.C., et al. (2020). Toxicity of ten herbicides to the tropical marine microalgae Rhodomonas salina. Scientific Reports, 10, Article 7521.
Tischer, W., & Strotmann, H. (1977). Relationship between inhibitor binding by chloroplasts and inhibition of photosynthetic electron transport. Biochimica et Biophysica Acta (Bioenergetics), 460(1), 113–125.
U.S. Environmental Protection Agency (EPA) (n.d.). Ambient Aquatic Life Water Quality Criteria for Atrazine. US EPA.
Vermaas, W.F., Renger, G., & Arntzen, C.J. (1984). Herbicide/quinone binding interactions in photosystem II. Zeitschrift für Naturforschung C, 39(5), 368–373.
Wallace, D.C. (1999). Mitochondrial diseases in man and mouse. Science, 283(5407), 1482–1488.
Wilkinson, A.D., Collier, C.J., Flores, F., & Ralph, P.J. (2015). Assessing the toxicity of herbicides to tropical seagrasses using chlorophyll fluorescence. Marine Pollution Bulletin, 95(2), 449–455.
Xie, L., Solhaug, K.A., Song, Y., Brede, D.A., Lind, O.C., Salbu, B., & Tollefsen, K.E. (2019). Modes of action and adverse effects of gamma radiation in an aquatic macrophyte Lemna minor. Science of the Total Environment, 680, 23–34.