
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 641
Title
Activation, estrogen receptor alpha leads to increased, phenotypic female-biased sex ratio via increased, differentiation to ovaries
Short name
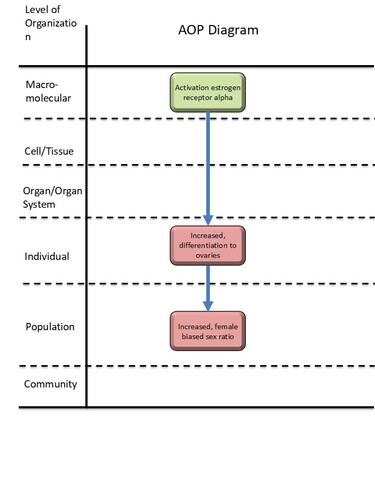
Graphical Representation
Point of Contact
Contributors
- John Frisch
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 22, 2026 09:09
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, estrogen receptor alpha | January 28, 2026 14:32 |
| Increased, differentiation to ovaries | April 17, 2026 13:59 |
| Increased, phenotypic female-biased sex ratio | April 17, 2026 14:57 |
| Activation, ERα leads to Increased, differentiation to ovaries | April 17, 2026 14:47 |
| Increased, differentiation to ovaries leads to Increased, female-biased sex ratio | April 17, 2026 14:52 |
Abstract
Estrogen receptor alpha (ERa) is a nuclear transcription factor involved in regulation of many physiological processes in vertebrates. Binding by estrogen induces the transcription of target genes. Here we focus on the role of ERa in the hypothalamus- pituitary-gonadal (HPG) axis involved in reproductive development. Estrogens have a central role in pathways leading to ovarian differentiation, with androgens causing testicular differentiation (Guiguen et al., 2010).
In species with genetic sex determination (GSD) whether an individual develops into a male or female is based on chromosome composition. In species with environmental sex determination (ESD), environmental factors such as temperature influence whether an individual develops into a male or female (in vertebrates, mainly fish and reptiles per Nagahama et al., 2021). For a review of sex determination systems in invertebrates, see (Picard et al., 2021).
In fish husbandry, long standing practice has demonstrated that adding estrogens during development can increase the number of individuals that develop ovaries. This is of interest when females achieve greater growth than males (in many fish species per review by Piferrer, 2001). Similarly exposure to estrogenic endocrine disrupting compounds during development can result in an increased number of fish that develop ovaries as seen in sex ratios (in many fish species per review by Dang and Kienzler, 2019). An adverse outcome of female-biased sex ratio occurs when a disruptor skews more individuals to developing ovaries and resulting in more females and intersex individuals, which has the potential to reduce effective population size and have negative effects on population dynamics. A 7-year whole lake study showed a near population collapse of fathead minnows (Pimephales promelas) exposed to doses of 5-6 ng/L 17β-ethynylestradiol (EE2) in a treated lake compared to a reference lake due to arrested male testicular development and genotypic males exhibiting ova-testes, followed by loss of young-of-the year age classes (Kidd et al., 2007).
Although the relative importance of genetic and environmental factors for determining the sex of an individual differ among classes of vertebrates, there are some evolutionarily conserved consistencies in development (Ditewig and Yao, 2005; Nichol et al., 2022). The gonadal primordium (genital ridge) develops on the surface of the mesonephros (intermediate mesoderm), which has the capability to develop either into ovaries or testes. Upon receiving cues to undergo female development, cells from the gonadal primordium differentiate into granulosa cells. Subsequent differentiation results in increased specialization of cells in the developing ovary, including germ cells (follicles that develop into oocytes) and somatic cells (granulosa and thecal cells that produce hormones among other duties, stromal cells that provide connective tissue, epithelium surface cells).
This AOP contributes to the scientific understanding of the mechanistic foundations for linking estrogen receptor agonism to the adverse outcome female-biased sex ratio, with female-biased sex ratio representing an apical endpoint in guideline tests associated with the Endocrine Disruptor Screening Program (US EPA, 1998).
AOP Development Strategy
Context
This AOP was part of an Environmental Protection Agency effort to develop AOPs that establish scientifically supported causal linkages between alternative endpoints measured using new approach methodologies (NAMs) and guideline apical endpoints measured in Tier 1 and Tier 2 test guidelines (U.S. EPA, 2024) employed by the Endocrine Disruptor Screening Program (EDSP). A series of key events that represent significant, measurable, milestones connecting molecular initiation to apical endpoints indicative of adversity were identified based on scientific review articles and empirical studies. Additionally, scientific evidence supporting the causal relationships between each pair of key events was assembled and evaluated. The present effort focused primarily on empirical studies with fish.
Strategy
The scope of the aforementioned EPA project was to develop AOP(s) relevant to apical endpoints employed in the test guidelines, based on mechanisms consistent with empirical studies. The literature used to support this AOP and its constituent pages began with the test guidelines and followed to primary, secondary, and/or tertiary works concerning the relevant underlying biology. KE and KER page creation and re-use was determined using Handbook principles where page re-use was preferred.

Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1065 | Activation, estrogen receptor alpha | Activation, ERα |
| KE | 2417 | Increased, differentiation to ovaries | Increased, differentiation to ovaries |
| AO | 2418 | Increased, phenotypic female-biased sex ratio | Increased, female-biased sex ratio |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Activation, ERα leads to Increased, differentiation to ovaries | non-adjacent | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Development | Moderate |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | High |
| Mixed | Moderate |
Overall Assessment of the AOP
|
1. Support for Biological Plausibility of Key Event Relationships: Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
|
|
Key Event Relationship (KER) |
Level of Support Strong = Extensive understanding of the KER based on extensive previous documentation and broad acceptance. |
|
Relationship 3773: ERa activation AVPV leads to increased, differentiation to ovaries |
Strong support. The relationship between activation of estrogen receptor alpha and increased differentiation to ovaries is broadly accepted and supported among animals, particularly fish data. Activation of estrogen receptor alpha is often studied in vitro, with activation of estrogen receptor alpha inferred in organism studies when downstream effects are consistent with in vitro observations. |
|
Relationship 3774: Increased, differentiation to ovaries leads to Increased, female-biased sex ratio |
Strong support. The relationship between increased differentiation to ovaries and increased female biased sex ratio is broadly accepted and supported among animal data. When an increased number of individuals develop ovaries there is an increase in females in a population, directly resulting in increased female biased sex ratio. |
|
Overall |
Strong support. Extensive understanding of the relationships between events from empirical studies from animals, particularly fish, with some inference of estrogen receptor alpha activation from in vitro studies when performing in vivo studies. |
Domain of Applicability
Life Stage: Development.
Sex: Applies to females, with some mixed genders observed.
Taxonomic: Possible in any animal species that has females and males. Largely studied in vertebrates, with some research in sexually reproducing invertebrate. Vertebrates differ in prevalence of environmental sex determination (Nagahama et al., 2021), with amphibians, reptiles and fish most likely to have increased differentiation to ovaries from environmental factors.
Essentiality of the Key Events
|
2. Essentiality of Key Events: Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
|
|
Key Event (KE) |
Level of Support Moderate = Direct evidence from specifically designed experimental studies illustrating essentiality between key events, best characterized as non-adjacent due to known hormone effects. Strong = Direct evidence from specifically designed experimental studies illustrating essentiality and direct relationship between key events. |
|
MIE 1065 Activation estrogen receptor alpha |
Moderate support. Activation of estrogen receptor alpha leads to increased differentiation to ovaries. Evidence is available from in vitro studies to establish estrogenicity by estrogen receptor competitive binding assays and from in vivo exposure to estrogens, endocrine-disrupting compounds, and toxicants. Best evidence for essentiality for activation of estrogen receptor alpha is from fish husbandry studies that have shown that in a wide variety of species adding estrogen compounds during development results in an increase in the number of individuals that develop ovaries, of economic interest when females achieve greater growth than males (Reviewed in Piferrer, 2001). Gene knockout studies suggest that the relationship between activation of estrogen receptor alpha and development of ovaries is best characterized as non-adjacent, with Estrogen receptor alpha having a key signalling role for enzyme production responsible for hormone levels leading to differentiation of ovaries with normal function (e.g. aromatase (cyp19a1a); Lau et al, 2016; Zhang et al., 2017; Chen et al. 2018; Yan et al. 2019). |
|
KE 2417 Increased, differentiation to ovaries |
Strong support. Increased differentiation to ovaries leads to increased female biased sex ratio. Sex ratio is determined by the proportion of females to males in a population. Development of more organisms with ovaries will lead directly to a female-biased sex ratio. |
|
AO 2418 Increased, female-biased sex ratio |
This is the final event of the AOP. |
|
Overall |
Moderate to strong support. Direct evidence from empirical studies from animals, particularly fish, and cell lines for all key events. |
Evidence Assessment
|
3. Empirical Support for Key Event Relationship: Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? |
|
|
Key Event Relationship (KER) |
Level of Support Strong = Experimental evidence from exposure to toxicant shows consistent change in both events across taxa and study conditions. |
|
Relationship 3773: ERa activation AVPV leads to increased, differentiation to ovaries |
Strong support. Activation of estrogen receptor alpha leads to increased differentiation to ovaries. Evidence is available from in vivo estrogen compound studies, endocrine disruptor studies, toxicant studies, temperature alteration studies, and in vitro studies to establish estrogenicity by estrogen receptor competitive binding assays. Activation of estrogen receptor alpha occurred earlier in the time-course of exposure than increased differentiation to ovaries, and the concentrations that activated estrogen receptor alpha were equal to or lower than the concentrations that increased differentiation to ovaries. Therefore, the data support a causal relationship. In some in vivo laboratory studies, activation of estrogen receptor alpha is inferred by response from a stressor known to be an ERa agonist from in vitro studies. |
|
Relationship 3774: Increased, differentiation to ovaries leads to Increased, female-biased sex ratio |
Strong support. Increased differentiation to ovaries leads to increased female biased sex ratio. Evidence is available from vivo estrogen compound studies, endocrine disruptor studies, toxicant studies, and temperature alteration studies. Increased differentiation to ovaries occurred earlier in the time-course of exposure than increased female biased sex ratio, and the concentrations of stressors that increased differentiation to ovaries were equal to or lower than the concentrations that increased female biased sex ratio. When an increased number of individuals develop ovaries there is an increase in females in a population, directly resulting in increased female biased sex ratio. Therefore, the data support a causal relationship. |
|
Overall |
Strong support. Evidence from empirical studies shows consistent relationships in upstream and downstream events, with upstream events occurring earlier in the time-course of exposure and at equal or lower concentrations than downstream events, supporting causal relationships. |
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
References
Chen Y, Tang H, Wang L, He J, Guo Y, Liu Y, Liu X, Lin H. 2018. Fertility Enhancement but Premature Ovarian Failure in esr1-Deficient Female Zebrafish. Frontiers in Endocrinolology 9: 567.
Dang Z, Kienzler A. 2019. Changes in fish sex ratio as a basis for regulating endocrine disruptors. Environment International 130: 104928.
Ditewig AC, Yao HH. 2005. Organogenesis of the ovary: a comparative review on vertebrate ovary formation. Organogenesis 2(2): 36-41.
Guiguen Y, Fostier A, Piferrer F, Chang CF. 2010. Ovarian aromatase and estrogens: a pivotal role for gonadal sex differentiation and sex change in fish. General and Comparative Endocrinology 165(3): 352-366.
Kidd KA, Blanchfield PJ, Mills KH, Palace VP, Evans RE, Lazorchak JM, Flick RW. 2007. Collapse of a fish population after exposure to a synthetic estrogen. Proceedings of the National Academy of Sciences of the United States of America 104(21): 8897-8901.
Lau ES, Zhang Z, Qin M, Ge W. Knockout of Zebrafish Ovarian Aromatase Gene (cyp19a1a) by TALEN and CRISPR/Cas9 Leads to All-male Offspring Due to Failed Ovarian Differentiation. 2016. Scientific Reports 6:37357.
Nagahama Y, Chakraborty T, Paul-Prasanth B, Ohta K, Nakamura M. 2021. Sex determination, gonadal sex differentiation, and plasticity in vertebrate species. Physiological Reviews 101(3): 1237-1308.
Nicol B, Estermann MA, Yao HH, Mellouk N. 2022. Becoming female: Ovarian differentiation from an evolutionary perspective. Frontiers in Cell and Development Biology 10: 944776.
Piferrer F. 2001. Endocrine sex control strategies for the feminization of teleost fish. Aquaculture 197: 229–281.
Picard MAL, Vicoso B, Bertrand S, Escriva H. 2021. Diversity of Modes of Reproduction and Sex Determination Systems in Invertebrates, and the Putative Contribution of Genetic Conflict. Genes 12(8): 1136.
U.S. Environmental Protection Agency. 1998. Health Effects Test Guidelines OPPTS 870.3800 Reproduction and Fertility Effects. https://ntp.niehs.nih.gov/sites/default/files/iccvam/suppdocs/feddocs/epa/epa_870_3800.pdf (retrieved 24 December 2025)
U.S. Environmental Protection Agency. 2004. EDSP Test Guidelines and Guidance Document. https://www.epa.gov/test-guidelines-pesticides-and-toxic-substances/edsp-test-guidelines-and-guidance-document (retrieved 25 July 2025).
Yan L, Feng H, Wang F, Lu B, Liu X, Sun L, Wang D. 2019. Establishment of three estrogen receptors (esr1, esr2a, esr2b) knockout lines for functional study in Nile tilapia. The Journal of Steroid Biochemistry and Molecular Biology 191:105379.
Zhang X, Li M, Ma H, Liu X, Shi H, Li M, Wang D. Mutation of foxl2 or cyp19a1a Results in Female to Male Sex Reversal in XX Nile Tilapia. 2017. Endocrinology. 158(8): 2634-2647.