
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 297
Title
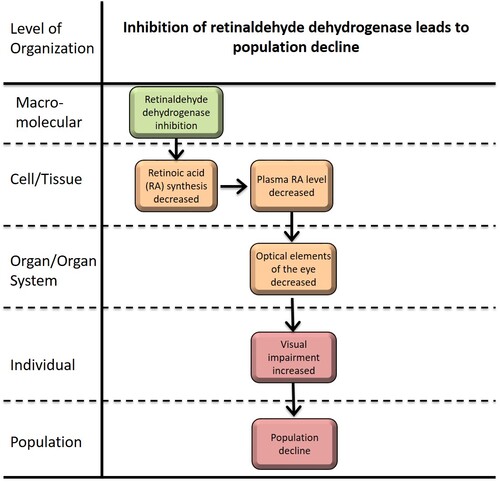
Inhibition of retinaldehyde dehydrogenase leads to population decline
Short name
Graphical Representation
Point of Contact
Contributors
- Young Jun Kim
Coaches
- Dan Villeneuve
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.77 | Under Development |
This AOP was last modified on April 29, 2023 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Retinaldehyde dehydrogenase inhibition | May 22, 2019 05:03 |
| Altered, Photoreceptor patterning | June 16, 2021 06:56 |
| Decreased retinoic acid (RA) synthesis | May 22, 2019 05:10 |
| Decreased plasma RA level | November 03, 2025 03:52 |
| Altered, Visual function | July 08, 2022 07:30 |
| Decrease, Population growth rate | January 03, 2023 09:09 |
| Retinaldehyde dehydrogenase leads to retinoic acid | May 22, 2019 05:13 |
| retinoic acid leads to plasma retinoic acid | May 22, 2019 05:14 |
| plasma retinoic acid leads to Altered, Photoreceptor patterning | May 22, 2019 05:14 |
| Altered, Photoreceptor patterning leads to Altered, Visual function | June 16, 2021 07:24 |
| Altered, Visual function leads to Decrease, Population growth rate | May 25, 2020 10:50 |
| Disulphiram | May 22, 2019 05:17 |
| Diethylaminobenzaldehyde | May 22, 2019 05:17 |
| Citral | May 22, 2019 05:18 |
| Paclobutrazol | May 22, 2019 05:18 |
| nitrofen | May 22, 2019 05:18 |
| 4-biphenyl carboxylic acid | May 22, 2019 05:18 |
| Bisdiamine | May 22, 2019 05:19 |
| SB-210661 | May 22, 2019 05:19 |
Abstract
The present AOP is designed to estimate potential AO of fishes results from the retinaldehyde dehydrogenase (RALDH) inhibition. Visual impairment results from eye development of an embryonic cell might lead to population decline which is the potential endpoint. This AOP will provide a useful risk assessment tool for the toxic assessment of chemicals. Furthermore, this AOP can be applied to the prediction of eco-toxicity caused by the inhibition of RALDH.
AOP Development Strategy
Context
|
This adverse outcome pathway (AOP) represents the potential causative adverse outcomes (AOs) by inhibition of retinaldehyde dehydrogenase (RALDH), which is one of the crucial enzymes participating in retinol metabolism. The role of RALDH in retinol metabolism is to catalyze the chemical reaction converting retinal to retinoic acid (RA). The synthesized RA is associated with the cellular RA-binding protein (CRABP) and enters into the nucleus, and then bind to retinoic acid receptors (RARs) along with retinoid X receptors (RXRs) (Vilhais-Neto and Pourquié, 2008). The activated RARs and RXRs can act as target gene transcription factors regulating embryonic development in fishes (Perz-Edwards et al., 2011). Inhibition of RALDH can be caused by chemical inhibitors such as Disulphiram, Citral, Paclobutrazol, Diethylaminobenzaldehyde, Nitrofen, 4-biphenyl carboxylic acid, Bisdiamine, SB-210661 and etc. (Marsh-Armstrong et al., 1994; Chawla et al., 2018; Wang et al., 2017; Le et al., 2012; Mey et al., 2003). RALDH inhibition, the molecular initiating event (MIE) for this AOP, leads to decreased RA synthesis blocking the reaction converting retinal to RA in embryonic cells (Hyatt and Dowling, 1997; Molotkov et al., 2002; Le et al., 2012; Duester, 2009). Since RA is an essential activator for the RARs and RXRs-mediated gene transcription, low level of plasma RA leads to abnormal development in embryonic cells. A number of previous studies well-elucidated the abnormal developments by RA inhibition including visual function and eye development (Duester et al., 2009; Hyatt and Dowling, 1997; Hyatt et al., 1996; Kam et al., 2012; Le et al., 2012; Luo et al., 2006; Marsh-Armstrong et al., 1994; Matt et al., 2005; Wang et al., 2017), intestinal development (Nadauld et al., 2005), brain patterning and neurogenesis (Begemann et al., 2004; Niederreither and Dollé, 2008; Samarut et al., 2015), and heart development (Niederreither and Dollé, 2008; Samarut et al., 2015). The development of early embryonic cells of fishes plays an essential role in the organism’s young of year survival and adaptation in fluctuated environmental condition. The impact of the development of the optical elements of the eye by RALDH inhibition in fish population trajectory has not been clarified yet, although the importance of the visual function of fishes previously mentioned by previous studies (Fernald, 1984; Sandström, 1999). Acknowledgements: This research was supported by the National Research Council of Science & Technology(NST) grant by the Korea government (MSIP) (No. CAP-17-01-KIST Europe) |
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1637 | Retinaldehyde dehydrogenase inhibition | Retinaldehyde dehydrogenase |
| KE | 1640 | Altered, Photoreceptor patterning | Altered, Photoreceptor patterning |
| KE | 1641 | Decreased retinoic acid (RA) synthesis | retinoic acid |
| KE | 1642 | Decreased plasma RA level | plasma retinoic acid |
| KE | 1643 | Altered, Visual function | Altered, Visual function |
| AO | 360 | Decrease, Population growth rate | Decrease, Population growth rate |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Retinaldehyde dehydrogenase leads to retinoic acid | adjacent | High | |
| retinoic acid leads to plasma retinoic acid | adjacent | High | |
| plasma retinoic acid leads to Altered, Photoreceptor patterning | adjacent | High | |
| Altered, Photoreceptor patterning leads to Altered, Visual function | adjacent | Moderate | Moderate |
| Altered, Visual function leads to Decrease, Population growth rate | adjacent | Low | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Birth to < 1 month | Moderate |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| fish | fish | Moderate | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Mixed | Moderate |
Overall Assessment of the AOP
This AOP is under development supported by the National Research Council of Science & Technology (NST) grant by the Korea government (MSIP) (No. CAP-17-01-KIST Europe) and P11911.
|
To do |
Expected duration |
|
|
Building the AOP frame |
Development of KEs |
3 month |
|
Production of experimental data |
18 month |
|
|
Overall assessment of the AOP |
Biological domain of applicability |
3 month |
|
Essentiality of all KEs |
3 month |
|
|
Evidence supporting all KERs |
5 month |
|
|
Quantitative WoE considerations |
5 month |
|
|
Quantitative understanding for each KER |
6 month |
|
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
The roles of RALDH play in sensory perception of fish mating opportunities via their presence in the visual functions. Significantly, the AO (population decline) can apply to IATA by dysfunction of the retinoid in reproduction in the aquatic environment. It can be applied to retinoid effects on a central nervous system within the OECD EDTA
References
- Chawla, B., Swain, W., Williams, A. L., & Bohnsack, B. L. (2018). Retinoic Acid Maintains Function of Neural Crest–Derived Ocular and Craniofacial Structures in Adult Zebrafish. Investigative ophthalmology & visual science, 59(5), 1924-1935.
- Duester, G. (2009). Keeping an eye on retinoic acid signaling during eye development. Chemico-biological interactions, 178(1-3), 178-181.
- Fernald, R. D. (1984). Vision and Behavior in an African Cichlid fish: Combining behavioral and physiological analyses reveals how good vision is maintained during rapid growth of the eyes. American Scientist, 72(1), 58-65.
- Hyatt, G. A., Schmitt, E. A., Marsh-Armstrong, N., McCaffery, P., Drager, U. C., & Dowling, J. E. (1996). Retinoic acid establishes ventral retinal characteristics. Development, 122(1), 195-204.
- Hyatt, G. A., & Dowling, J. E. (1997). Retinoic acid. A key molecule for eye and photoreceptor development. Investigative ophthalmology & visual science, 38(8), 1471-1475.
- Kam, R. K. T., Deng, Y., Chen, Y., & Zhao, H. (2012). Retinoic acid synthesis and functions in early embryonic development. Cell & bioscience, 2(1), 11.
- Le, H. G. T., Dowling, J. E., & Cameron, D. J. (2012). Early retinoic acid deprivation in developing zebrafish results in microphthalmia. Visual neuroscience, 29(4-5), 219-228.
- Luo, T., Sakai, Y., Wagner, E., & Dräger, U. C. (2006). Retinoids, eye development, and maturation of visual function. Journal of neurobiology, 66(7), 677-686.
- Marsh-Armstrong, N., McCaffery, P., Gilbert, W., Dowling, J. E., & Dräger, U. C. (1994). Retinoic acid is necessary for development of the ventral retina in zebrafish. Proceedings of the National Academy of Sciences, 91(15), 7286-7290.
- Matt, N., Dupé, V., Garnier, J. M., Dennefeld, C., Chambon, P., Mark, M., & Ghyselinck, N. B. (2005). Retinoic acid-dependent eye morphogenesis is orchestrated by neural crest cells. Development, 132(21), 4789-4800.
- Mey, J., Babiuk, R. P., Clugston, R., Zhang, W., & Greer, J. J. (2003). Retinal dehydrogenase-2 is inhibited by compounds that induce congenital diaphragmatic hernias in rodents. The American journal of pathology, 162(2), 673-679.
- Molotkov, A., et al. (2002). Stimulation of retinoic acid production and growth by ubiquitously expressed alcohol dehydrogenase Adh3. Proceedings of the National Academy of Sciences, 99(8), 5337-5342.
- Perz-Edwards, A., Hardison, N. L., & Linney, E. (2001). Retinoic acid-mediated gene expression in transgenic reporter zebrafish. Developmental biology, 229(1), 89-101.
- Sandström, A. (1999). Visual ecology of fish–a review with special reference to percids. Fiskeriverket rapport, 2, 45-80.
- Vilhais-Neto, G. C., & Pourquié, O. (2008). Retinoic acid. Current Biology, 18(5), R191-R192.
- Wang, W. D., Hsu, H. J., Li, Y. F., & Wu, C. Y. (2017). Retinoic acid protects and rescues the development of zebrafish embryonic retinal photoreceptor cells from exposure to paclobutrazol. International journal of molecular sciences, 18(1), 130.