
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 167
Title
Early-life estrogen receptor agonism leading to endometrial adenosquamous carcinoma via promotion of sine oculis homeobox 1 progenitor cells
Short name
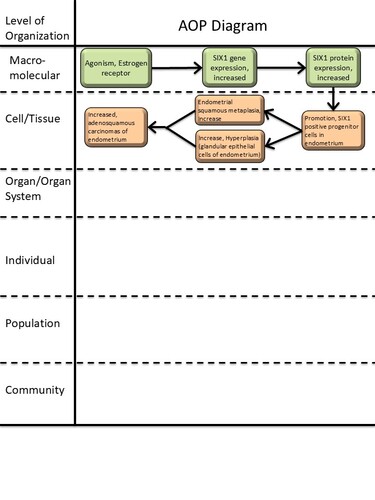
Graphical Representation
Point of Contact
Contributors
- Charles Wood
- Travis Karschnik
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on January 11, 2026 16:56
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Promotion, Sine oculis homeobox 1 positive progenitor cells in endometrium | September 12, 2025 12:32 |
| Endometrial squamous metaplasia, Increase | September 18, 2025 15:31 |
| Increased, adenosquamous carcinomas of endometrium | September 18, 2025 15:32 |
| Sine oculis homeobox 1 gene expression, increased | September 11, 2025 16:35 |
| Sine oculis homeobox 1 protein expression, increased | December 17, 2025 14:59 |
| Agonism, Estrogen receptor | September 11, 2025 15:13 |
| Increase, Hyperplasia (glandular epithelial cells of endometrium) | September 18, 2025 15:31 |
| Agonism, Estrogen receptor leads to SIX1 gene expression, increased | September 12, 2025 17:24 |
| SIX1 gene expression, increased leads to SIX1 protein expression, increased | September 18, 2025 11:04 |
| SIX1 protein expression, increased leads to Promotion, SIX1 positive progenitor cells in endometrium | September 18, 2025 11:05 |
| Promotion, SIX1 positive progenitor cells in endometrium leads to Endometrial squamous metaplasia, Increase | September 18, 2025 11:08 |
| Promotion, SIX1 positive progenitor cells in endometrium leads to Increase, Hyperplasia (glandular epithelial cells of endometrium) | September 18, 2025 11:05 |
| Endometrial squamous metaplasia, Increase leads to Increased, adenosquamous carcinomas of endometrium | September 18, 2025 11:06 |
| Increase, Hyperplasia (glandular epithelial cells of endometrium) leads to Increased, adenosquamous carcinomas of endometrium | September 18, 2025 11:06 |
| Diethylstilbestrol | November 29, 2016 18:42 |
| Genistein | November 29, 2016 18:42 |
Abstract
Developmental estrogen receptor agonism induces localized aberrant sine oculis homeobox 1 (six1) progenitor cell populations in the uterine epithelium. In adulthood, these six1+ progenitor cells drive abnormal epithelial differentiation. This differentiation manifests, in part, as squamous metaplasia and glandular hyperplasia which can both undergo malignant transformation resulting in adenosquamous carcinoma.
This AOP illustrates how early lifestage estrogen receptor agonism, and by extension estrogen receptor agonists, can drive molecular changes resulting in long-term adverse outcomes i.e., uterine cancer. The endometrial histological measurements: metaplasia, hyperplasia, and eventual adenosquamous carcinoma, are important markers/endpoints in the human health field. This is particularly relevant to the US EPA Endocrine Disruptor Screening Program (EDSP), which uses Tier 1 assays to identify potential estrogenic compounds and Tier 2 assays to evaluate adverse outcomes across lifestages and generations.
It also applies to a number of international regulatory frameworks concerned with endocrine disruption including but not limited to:
- OECD for international harmonization.
- The European Union (EU) by way of the European Chemicals Agency (ECHA), and the Registration, Evaluation, Authorization, and Restricting of Chemicals (REACH) regulation.
- Canada, who evaluates endocrine disruption under the Canadian Environmental Protection Act (CEPA).
AOP Development Strategy
Context
This AOP was as part of an Environmental Protection Agency effort to develop AOPs that establish scientifically supported causal linkages between alternative endpoints measured using new approach methodologies (NAMs) and guideline apical endpoints measured in Tier 1 and Tier 2 test guidelines (U.S. EPA, 2024) employed by the Endocrine Disruptor Screening Program (EDSP). A series of key events that represent significant, measurable, milestones connecting molecular initiation to apical endpoints indicative of adversity were identified based on scientific review articles and empirical studies. Additionally, scientific evidence supporting the causal relationships between each pair of key events was assembled and evaluated. The present effort focused primarily on empirical studies with laboratory rodents and other mammals.
Strategy
The scope of the aforementioned EPA project was to develop AOP(s) relevant to apical endpoints observed in the test guidelines, based on mechanisms consistent with empirical studies. The literature used to support this AOP and its constituent pages began with the test guidelines and followed to primary, secondary, and/or tertiary works concerning the relevant underlying biology. KE and KER page creation and re-use was determined using Handbook principles where page re-use was preferred.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 111 | Agonism, Estrogen receptor | Agonism, Estrogen receptor |
| KE | 2329 | Sine oculis homeobox 1 gene expression, increased | SIX1 gene expression, increased |
| KE | 2330 | Sine oculis homeobox 1 protein expression, increased | SIX1 protein expression, increased |
| KE | 1066 | Promotion, Sine oculis homeobox 1 positive progenitor cells in endometrium | Promotion, SIX1 positive progenitor cells in endometrium |
| KE | 1068 | Endometrial squamous metaplasia, Increase | Endometrial squamous metaplasia, Increase |
| KE | 772 | Increase, Hyperplasia (glandular epithelial cells of endometrium) | Increase, Hyperplasia (glandular epithelial cells of endometrium) |
| AO | 1070 | Increased, adenosquamous carcinomas of endometrium | Increased, adenosquamous carcinomas of endometrium |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Agonism, Estrogen receptor leads to SIX1 gene expression, increased | adjacent | High | Not Specified |
| SIX1 gene expression, increased leads to SIX1 protein expression, increased | adjacent | High | Not Specified |
| SIX1 protein expression, increased leads to Promotion, SIX1 positive progenitor cells in endometrium | adjacent | High | Not Specified |
| Promotion, SIX1 positive progenitor cells in endometrium leads to Endometrial squamous metaplasia, Increase | adjacent | High | Moderate |
| Promotion, SIX1 positive progenitor cells in endometrium leads to Increase, Hyperplasia (glandular epithelial cells of endometrium) | adjacent | High | Moderate |
| Endometrial squamous metaplasia, Increase leads to Increased, adenosquamous carcinomas of endometrium | adjacent | Moderate | Not Specified |
| Increase, Hyperplasia (glandular epithelial cells of endometrium) leads to Increased, adenosquamous carcinomas of endometrium | adjacent | Moderate | Not Specified |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Juvenile | Moderate |
| Embryo | High |
| Fetal | High |
| Adult | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | High |
Overall Assessment of the AOP
|
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
Extensive understanding of the KER based on extensive previous documentation and broad acceptance. |
KER is plausible based on analogy to accepted biological relationships, but scientific understanding is incomplete |
Empirical support for association between KEs , but the structural or functional relationship between them is not understood. |
|
Support for Biological Plausibility of Key Event Relationships: Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
|
|
Key Event Relationship (KER) |
Level of Support |
|
Relationship 3561: Agonism, Estrogen receptor leads to SIX1 gene expression, increased |
Strong support. A number of plausible mechanisms have been proposed for this relationship. |
|
Relationship 3562: SIX1 gene expression, increased leads to SIX1 protein expression, increased |
Strong support. This is an example of the broadly accepted and well-known transcription -> translation process. |
|
Relationship 3563: SIX1 protein expression, increased leads to Promotion, SIX1 positive progenitor cells in endometrium |
Strong support. Protein expression levels and their relationship to the cells where that protein is measured are well documented. Six1’s developmental role is also plausibly linked to the increased incidence of progenitor cells specifically. |
|
Relationship 3626: Promotion, SIX1 positive progenitor cells in endometrium leads to Endometrial squamous metaplasia, Increase |
Moderate/Strong support. Six1’s role in tissue developmental and the mechanisms by which that occurs are plausibly linked in subsequent metaplasia. |
|
Relationship 3627: Promotion, SIX1 positive progenitor cells in endometrium leads to Increase, Hyperplasia (glandular epithelial cells of endometrium) |
Moderate/Strong support. Six1’s role in tissue developmental and the mechanisms by which that occurs are plausibly linked in subsequent hyperplasia. |
|
Relationship 3610: Endometrial squamous metaplasia, Increase leads to Increased, adenosquamous carcinomas of endometrium |
Strong support. Well supported from a biological standpoint given the nature cell progression to malignancy. |
|
Relationship 3608: Increase, Hyperplasia (glandular epithelial cells of endometrium) leads to Increased, adenosquamous carcinomas of endometrium |
Strong support. Well supported from a biological standpoint given the nature cell progression to malignancy. |
|
Overall: |
Strong support. Well established plausibility arguments at each step in the pathway. |
Domain of Applicability
Taxonomic Applicability
The empirical evidence presented throughout is derived from mice and human models. The mechanisms by which estrogen receptor agonism interacts with six1 expression leading to altered cell differentiation and eventually adenosquamous carcinoma could plausibly be extended to all uterine bearing mammals.
Lifestage Applicability
The empirical evidence presented throughout extends from developmental periods (embryogenesis and neonatal periods) through adulthood. Estrogen receptor agonism and the subsequent changes to six1 gene, protein, and progenitor populations occur early in development. The conditions that they create are then re-encountered at adulthood to kick-off the changes to cellular differentiation which can result in uterine cancer.
Sex Applicability
Limited to females as the outcome is limited by a sex-specific organ (uterus).
Essentiality of the Key Events
|
Defining question |
High (Strong) |
Moderate |
Low (Weak) |
|
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from specifically designed experimental studies illustrating essentiality for at least one of the important KEs |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE |
No or contradictory experimental evidence of the essentiality of any of the KEs. |
|
Essentiality of Key Events: Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
|
|
Key Event (KE) |
Level of Support |
|
MIE 111: Agonism, Estrogen receptor |
Strong support. KE2329 is not prevented, generally, in the absence of early lifestage ER agonism. However, KE2329 localized to the uterus and specific tissues in the uterus is. AO1070 does not require early lifestage ER agonism as it can be cause through a number of other mechanisms. |
|
KE 2329: SIX1 gene expression, increased |
Strong support. KE2330 is prevented in the absence of six1 gene expression. AO1070 does not require aberrant SIX1 gene expression as it can be caused through a number of other mechanisms. |
|
KE 2330: SIX1 protein expression, increased |
Strong support. KE1066 is prevented in the absence of six1 protein expression. AO1070 does not require aberrant SIX1 protein expression as it can be caused through a number of other mechanisms. |
|
KE 1066: Promotion, SIX1 positive progenitor cells in endometrium |
Moderate support. For both KE1068 and KE772, a promotion of six1 positive progenitor cells is but one mechanism to create the conditions for squamous metaplasia and glandular hyperplasia. Both can occur independently of six1+ progenitor cells. |
|
KE 1068: Endometrial squamous metaplasia, Increase |
Strong support. AO1070 can’t be diagnosed without a malignant squamous metaplasia element. |
|
KE 772: Increase, Hyperplasia (glandular epithelial cells of endometrium) |
Strong support. AO1070 can’t be diagnosed without a malignant glandular hyperplasia element. |
|
AO 1070: Increased, adenosquamous carcinomas of endometrium |
Strong support. This AO isn’t possible in subjects without malignant squamous metaplasia and glandular hyperplasia of the epithelial endometrium. However, the pathway leading from aberrant six1 expression leading to altered cell differentiation is far from the only one documented to achieve the AO. |
|
Overall: |
Moderate support. MIE111 and the downstream KEs 2329, 2330, and 1066 are supported by direct evidence, some including causal elements. AO1070 requires malignant versions of KE1068 and KE772 to be diagnosed so those are inextricably linked. AO1070 can come about by mechanisms outside of six1 expression leading to the required aberrant differentiation. |
Evidence Assessment
|
Defining Questions |
High (Strong) |
Moderate |
Low (Weak) |
|
Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? Does KEup occur at lower doses and earlier time points than KE down and is the incidence of KEup > than that for KEdown? Inconsistencies? |
if there is dependent change in both events following exposure to a wide range of specific stressors (extensive evidence for temporal, dose- response and incidence concordance) and no or few data gaps or conflicting data |
if there is demonstrated dependent change in both events following exposure to a small number of specific stressors and some evidence inconsistent with the expected pattern that can be explained by factors such as experimental design, technical considerations, differences among laboratories, etc. |
if there are limited or no studies reporting dependent change in both events following exposure to a specific stressor (i.e., endpoints never measured in the same study or not at all), and/or lacking evidence of temporal or dose- response concordance, or identification of significant inconsistencies in empirical support across taxa and species that don’t align with the expected pattern for the hypothesized AOP |
|
Empirical Support for Key Event Relationship: Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? |
|
|
Key Event Relationship (KER) |
Level of Support |
|
Relationship 3561: Agonism, Estrogen receptor leads to SIX1 gene expression, increased |
Strong support. Experimentally confirmed in mice including an ER antagonist exposure producing the opposite result. |
|
Relationship 3562: SIX1 gene expression, increased leads to SIX1 protein expression, increased |
Strong support. Experimentally confirmed in human cell lines including knockout experiments producing the opposite result. |
|
Relationship 3563: SIX1 protein expression, increased leads to Promotion, SIX1 positive progenitor cells in endometrium |
Strong support. Mice and human data are presented supporting the plausibility arguments that six1 protein expression encourages proliferation, expands progenitor populations, suppresses or otherwise alters differentiation, and stabilizes a transcriptional feedback loop. |
|
Relationship 3626: Promotion, SIX1 positive progenitor cells in endometrium leads to Endometrial squamous metaplasia, Increase |
Moderate/Strong support. Evidence presented here focus on concurrent six1+ cells and squamous metaplasia. One study is included demonstrates six1 deletion results in less metaplasia implying that DES induced six1 could be partially causal. |
|
Relationship 3627: Promotion, SIX1 positive progenitor cells in endometrium leads to Increase, Hyperplasia (glandular epithelial cells of endometrium) |
Moderate support. Evidence presented here focus on concurrent six1+ cells and glandular hyperplasia. The evidence supports the idea that more six1+ cells are found in areas of more hyperplasia. |
|
Relationship 3610: Endometrial squamous metaplasia, Increase leads to Increased, adenosquamous carcinomas of endometrium |
Moderate support. This is empirically supported in humans. Although, the evidence is presented as co-occuring rather than strictly causal in nature. |
|
Relationship 3608: Increase, Hyperplasia (glandular epithelial cells of endometrium) leads to Increased, adenosquamous carcinomas of endometrium |
Moderate support. This is empirically supported in humans. Although, the evidence is presented as co-occuring rather than strictly causal in nature. |
|
Overall: |
Moderate/Strong support. These KERs are a blend of direct causal evidence and co-occurrence at various stages. Because the AO is a histological diagnosis the two required constituent findings, malignant squamous metaplasia and malignant glandular hyperplasia are directly tied. |
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
As it pertains to OECD TG440 this AOP could inform the refinement of assay endpoints to include transcriptional and/or protein biomarkers. The timescale at which the overall AOP plays out, depending on species, could inform changes to, or the inclusion of, additional medium-term measurements related to those transcriptional and/protein changes.
The information could be used as justification for testing Diethylstilbestrol and Genistein at the Tier 2 level in the EDSP.
The AOP establishing links between short-term, early-life changes resulting in increased long-term cancer risk supports greater focus on early key events to establish safe exposure levels/timing. Six1 and any other associated genetic and protein markers could also serve as mechanistic biomarkers for early-life estrogenic perturbations and as predictive indicators for long-term uterine cancer outcomes.
References
OECD (2007), Test No. 440: Uterotrophic Bioassay in Rodents: A short-term screening test for oestrogenic properties, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris, https://doi.org/10.1787/9789264067417-en