
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 398
Title
Decreased ALDH1A (RALDH) activity leading to decreased fertility via disrupted meiotic initiation of fetal oogonia
Short name
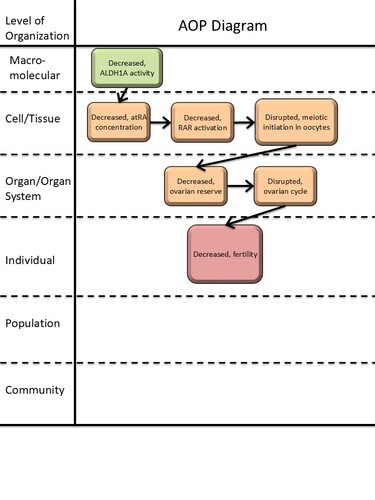
Graphical Representation
Point of Contact
Contributors
- Terje Svingen
- Tommaso Giorgi
Coaches
- You Song
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.97 | Under Development |
This AOP was last modified on May 01, 2026 07:45
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Decreased, ALDH1A (RALDH) enzyme activity | December 17, 2024 12:04 |
| Decreased, all-trans retinoic acid (atRA) concentration | February 13, 2023 08:04 |
| Disrupted, initiation of meiosis of oogonia in the ovary | July 04, 2025 04:15 |
| Decreased, size of the ovarian reserve | July 27, 2025 01:11 |
| decreased, Fertility | December 17, 2024 15:46 |
| disrupted, ovarian cycle | December 17, 2024 15:41 |
| Decreased, Retinoic Acid Receptor Activation | April 23, 2026 05:18 |
| Decreased, ALDH1A activity leads to Decreased, atRA concentration | December 18, 2024 03:16 |
| Decreased, atRA concentration leads to Disrupted, meiotic initiation in oocyte | July 04, 2025 04:35 |
| Decreased, atRA concentration leads to Decreased, RAR activation | April 24, 2026 03:39 |
| Decreased, RAR activation leads to Disrupted, meiotic initiation in oocyte | April 24, 2026 03:40 |
| Disrupted, meiotic initiation in oocyte leads to Decreased, ovarian reserve | December 18, 2024 06:36 |
| Decreased, ovarian reserve leads to disrupted, ovarian cycle | December 18, 2024 06:53 |
| disrupted, ovarian cycle leads to decreased, Fertility | March 18, 2025 12:17 |
Abstract
This AOP links inhibition of ALDH1A during fetal life with female infertility in adulthood. A key step in this AOP is a reduction in all-trans retinoic acid (atRA) locally in the fetal ovary, which prevents resident germ cells (oocytes) from entering meiosis. Evidence for this AOP, especially upstream events, draws heavily from mouse studies, both genetic models and from exposure studies (including explanted ovaries). Human evidence is also available, especially for downstream events where the oocyte pool/ovarian reserve is known to directly impact on fertility. In reproductive toxicity (animal studies and human epidemiology) fertility is an apical endpoint of high importance and has strong utility for chemical safety assessments. Infertility can be caused by many, and varied, factors, but this AOP focusses on linking perturbed meiosis through disrupted atRA signaling during development, thus supporting the use of data from in silico and in vitro measurements for interference with nuclear receptor activity (RAR/RXR) and atRA synthesis/expression to infer potential to cause in vivo effects.
AOP Development Strategy
Context
In mammals, the primordial germ cells are initially ‘bipotential’. They will develop into either oocytes or gonocytes in ovaries or testis, respectively, depending on cues from the somatic environment. Germ cells in the developing testis will enter a quiescent state and reactivate at the onset of puberty. In contrast, germ cells in the developing ovary will enter meiosis (prophase I) during fetal life. A key signaling event for this sexual dimorphic germ cell programming is retinoid signaling, with all-trans retinoic acid (atRA) acting as a meiosis-inducing factor (Spiller & Bowles, 2019).
The source of atRA during ovary development differs to some degree between species. In mice, the adjacent mesonephros, which expresses two enzymes necessary for the final step in atRA production, ALDH1A2 and ALDH1A3, is likely the main source of atRA at early developmental stages (Bowles et al, 2018; Bowles et al, 2006; Koubova et al, 2006; Niederreither et al, 1999). There is also the capacity for atRA to be produced within the ovary itself, due to local expression of the atRA-synthesizing enzyme ALDH1A1 (Bowles et al, 2016; Mu et al, 2013).
In humans, ALDH1A enzymes (ALDH1A, -1B and -1C) are expressed in both testes and ovaries of the developing fetus, which suggest a capacity for de novo synthesis of atRA (Childs et al, 2011; Jørgensen & Rajpert-De Meyts, 2014; le Bouffant et al, 2010), as is also the case in rabbits (Díaz-Hernández et al, 2019). One team studying human fetal ovaries reported a peak of ALDH1A1 expression at the onset of meiosis (le Bouffant et al, 2010), suggesting that meiotic onset in the human ovary depends on provision of atRA at the correct time. There seems to be conservation from rodent to human in terms of the requirement for atRA to induce the pre-meiotic factor STRA8. However, in mice atRA is produced by adjacent tissue and is present at high concentrations in the ovaries, whereas in human ovaries RA is present at only low levels and is then actively produced to induce meiosis in the ovary (Spiller & Bowles, 2019).
Strategy
An initial scoping search of open literature was performed using pre-set search terms (appendix A, 3aztqge6hj_App_A_search_terms.pdf), serving to collate an unbiased catalogue of literature beyond that of the AOP developer’s expertise, as well as content of a Draft Research Report on retinoid signaling and potential links to reproductive toxicity (Nilsson 2020). Based on this literature search (which yielded 97 publications) and expert knowledge, a putative AOP relevant for ovary development and female infertility was identified. A draft AOP including interim KEs and KERs were constructed, followed by preliminary essentiality assessments. Having established which modules to include in the AOP string, all KEs and KERs were developed as described individually.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1880 | Decreased, ALDH1A (RALDH) enzyme activity | Decreased, ALDH1A activity |
| KE | 1881 | Decreased, all-trans retinoic acid (atRA) concentration | Decreased, atRA concentration |
| KE | 2419 | Decreased, Retinoic Acid Receptor Activation | Decreased, RAR activation |
| KE | 1882 | Disrupted, initiation of meiosis of oogonia in the ovary | Disrupted, meiotic initiation in oocyte |
| KE | 1883 | Decreased, size of the ovarian reserve | Decreased, ovarian reserve |
| KE | 405 | disrupted, ovarian cycle | disrupted, ovarian cycle |
| AO | 406 | decreased, Fertility | decreased, Fertility |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Decreased, ALDH1A activity leads to Decreased, atRA concentration | adjacent | High | Moderate |
| Decreased, atRA concentration leads to Decreased, RAR activation | adjacent | ||
| Decreased, RAR activation leads to Disrupted, meiotic initiation in oocyte | adjacent | ||
| Disrupted, meiotic initiation in oocyte leads to Decreased, ovarian reserve | adjacent | High | Moderate |
| Decreased, ovarian reserve leads to disrupted, ovarian cycle | adjacent | Moderate | Low |
| disrupted, ovarian cycle leads to decreased, Fertility | adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Development | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Female | High |
Overall Assessment of the AOP
The majority of evidence supporting this AOP is derived from mouse studies, both in vitro (fetal ovary cultures) and in vivo (incl. genetic mouse models). There is also evidence from humans (in vitro ovary cultures), yet it is also recognized that there are some differences between mice and humans with regard to atRA synthesis, expression and potential role in meiotic initiation. Notably, an important link, yet not described as a separate key event, is the role for Stra8 in meiotic initiation alongside the established role for atRA to control Stra8 expression via RAR/RXR.
The evidence linking MIE with KE1 is considered as strong and regarded as canonical knowledge. Likewise, evidence for the downstream key events linking reduced oocyte pool/ovarian reserve with reduced fertility is very strong and regarded as canonical knowledge. The weak link in the overall AOP is the connection between reduced atRA levels and fertility via loss of oocytes during development. To strengthen this link, more evidence must be obtained; nevertheless, the remaining links are very strong and can be used to assess the impact of chemical stressors on female fertility. Yet, caution should be exercised with directly linking inhibition of ALDH1A2 with reduced fertility.
Domain of Applicability
-
Sex: This AOP applies to females. atRA is also involved in meiosis of testicular gonocytes, but this occurs postnatally. In the female ovaries, atRA induces meiosis of oocytes during gestation, thus the spatiotemporal expression of atRA in the ovaries are tightly controlled. Finally, as this AOP is concerned with establishing the ovarian reserve/follicle pool through mechanisms that are unique to ovaries, restricting the AOP to female only is appropriate.
-
Life stages: This AOP spans the period from mid- to late-gestation in mammals, all the way to adulthood where fertility is manifested. The upstream event pertains to fetal/neonatal life stages, whereas the downstream events pertain to adult reproductive life stages.
-
Taxonomy: Strongest evidence for the role of atRA in regulating oocyte entry into meiosis stems from mouse studies, so the taxonomic applicability is strongest for this animal model. Studies have also been done in rats. Evidence for the same mechanisms in humans is less substantiated (Li & Clagett-Dame, 2009; Griswold et al, 2012; Spiller & Bowles, 2022; Jørgensen & Rajpert-De Meyts, 2014).
Essentiality of the Key Events
The essentiality of each key event (KE) was evaluated, meaning that if an upstream KE is blocked or does not occur, subsequent downstream KEs or the adverse outcome (AO) are prevented or altered. Both direct and indirect evidence of essentiality were assessed according to the OECD developer’s handbook (see Appendix B, 1t3n0mxigk_App_B_Essentiality_2025.pdf) with a summary provided in Table 1.
Table 1: Essentiality assessment of KEs for AOP 398
Retrieved literature and overall assessments are detailed in Appendix B. ****substantial evidence, ***good evidence, **moderate evidence, *some evidence.
|
Event |
Direct evidence |
Indirect evidence |
Contradictory evidence |
Overall essentiality assessment |
|
KE-1880 |
**** |
|
*** |
Low |
|
KE-1881 |
**** |
|
**** |
Low |
|
KE-1882 |
**** |
|
|
High |
|
KE-1883 |
|
*** |
* |
Moderate |
|
KE-405 |
|
** |
|
Moderate |
Uncertainties, inconsistencies and data gaps
In mice, there is strong evidence to support the view that atRA is important for initiating meiosis in germ cells (Bowles et al, 2016; Spiller et al, 2017; Teletin et al, 2017). Some studies suggest that atAR is not critical but important for meiotic entry under normal physiological conditions by evidencing meiosis in Aldh1a1, Aldh1a2 and Aldh1a3 ablated mice, individually and in tandem (Bellutti et al, 2019; Chassot et al, 2020; Kumar et al, 2011); however, additional studies have shown redundant roles between all three Aldha isoforms which can compensate for deletion of one or two (Bowles et al, 2016). More specifically, both double (Aldh1a2/3) and triple (Aldh1a1/2/3) knockout mouse models display reduced Stra8 expression in oocytes, yet oocytes eventually go through meiosis, which could suggest a redundant role for atRA for meiosis in the ovaries (Chassot et al, 2020; Kumar et al, 2011). A similar phenotype with reduced Stra8 expression but eventual meiotic initiation is seen for deletion of atRA receptors RAR-α, -β, -γ) in mice (Vernet et al, 2020). But, although RAR knockouts were also capable of producing offspring, it remains unclear if any of the above-mentioned mouse models display impaired fertility or whether the size of their oocyte pools are affected.
Evidence Assessment
Biological Plausibility, coherence, and consistency of the experimental evidence
The role for ALDH1A2 in the synthesis of atRA is well established as an essential component of regulating regional expression of retinoid species during development. It is also well established that atRA is an inducer of meiosis in germ cells in mice; however, there is some debate about the essentiality of atRA in this process in human fetal ovaries. The requirement for oocytes to enter the first phase of meiosis during fetal development is also well established, hence the biological plausibility linking meiotic failure with loss of oocytes at later developmental stages is strong.
Although non-meiotic oocytes can survive in germ cell nests and during nest breakdown, they will ultimately be eliminated from the oocyte pool of competent follicles. There is therefore a direct link between meiotic entry and fertility during adulthood. Thus, this AOP provides a plausible chain of events linking reduced atRA during fetal life with reduced ovarian reserve and fertility during reproductive age. The strength of the downstream KEs and KER – reduced ovarian reserve and reduced fertility – is very well documented and thus the biological plausibility is very strong. Evidence for a direct link between the AO and perturbed atRA synthesis, or reduced atRA levels, during early development comes mainly from mouse studies; yet the relationship is regarded biologically plausible also in humans, but with weight of evidence not being as strong.
The overall evidence assessment scores for each KER is summarized in the below Table:
Table 2: Biological plausibility of key event relationships of AOP-398.
|
Assessment score |
Rationale |
|
|
KER-2401 |
High |
It is well established that atRA is synthesized from retinaldehyde by the enzyme ALDH1A. |
|
KER-2477 |
Moderate |
A large body of evidence supports that atRA is involved in initiation of meiosis in ovarian germ cells but there also is conflicting data indicating that atRA is not the only determining factor. |
|
KER-2481 |
High |
It is well established that germ cells must enter meiosis prophase I during fetal development to establish the primordial follicles that ultimately make up the ovarian follicle pool. |
|
KER-2525 |
Moderate |
It is highly plausible that a reduced number of primordial follicles leads to irregularities in the ovarian cycle. The main challenge lies with quantification of effect and hence uncertainties regarding when the effect manifests relative to reduction in primordial follicles. |
|
KER-394 |
High |
It is well established that ovarian cyclicity is related to fertility. |
Empirical support
Concordance of dose-response relationships
The quantitative understanding of dose-response relationships in this AOP is limited. Whilst the relative levels of endogenous atRA produced by the ovary (for any species) remains unknown, similarly, the quantitative relationship between atRA levels and induction of meiosis also remains unclear. Nevertheless, it is has been conclusively shown that low levels of exogenous atRA can induce mouse and rat germ cells to enter meiosis both in vitro and ex vivo (Bowles et al, 2006; Livera et al, 2000). Likewise, atRA is necessary to achieve meiosis in in vitro-derived oocytes via PGCLCs (Miyauchi et al, 2017).
Temporal concordance among the key events and the adverse outcome
This AOP bridges two different life stages: fetal/perinatal and adult/reproductive age. The adverse outcome is the result of perturbation taking place during early stages of ovary development. In mice, rats and humans, the oocytes must enter meiosis prophase in order to establish the follicle pool/ovarian reserve postnatally. Thus, the AOP focusses on chemical perturbations during fetal life, which occurs around E13-E16 in mice and E15-E18 is rats, or first trimester in humans (Peters, 1970), but the adverse outcome does not manifest until adulthood.
There is strong temporal concordance between the various key events, from inhibition of ALDH1A2 (RALDH2) that leads to reduced atRA synthesis. In turn, atRA must be present in the fetal ovaries at the time when oocytes are supposed to enter meiosis mid-gestation in mice (or first trimester in human). With a significant reduction in available atRA the oocytes will not enter meiosis, ultimately leading to the downstream key event of loss of oocytes beyond what is normal. The number of oocytes, or the oocyte pool/ovarian reserve, in turn will affect ovary function and fertility at reproductive stages, hence the temporal sequence of events is rational based on the biological process.
Strength, consistency, and specificity of association of adverse effect and initiating event
In mice, there is strong evidence to support the view that atRA is an inducer of meiosis in germ cells, with consistent results from in vitro (PGCLCs), ex vivo (ovary cultures) and in vivo studies as listed under KE 2477. There is strong evidence showing the importance of RA for female fertility, but this relates to many aspects of reproductive development and function from fetal life to adulthood, including maintaining pregnancy (Clagett-Dame & Knutson, 2011). Thus, it can be difficult to distill exactly how atRA-controlled meiotic entry of oocytes directly link to reduced fertility. Nevertheless, a direct relationship is strongly supported by the fact that Stra8-depleted mice are infertile with small ovaries lacking oocytes (Baltus et al, 2006) and that Stra8 induction in germ cells is controlled by atRA in mice, rats and humans (Bowles et al, 2006; Childs et al, 2011; Koubova et al, 2006; Livera et al, 2000). Furthermore, vitamin A-deficient (VAD) mice display delayed or failed meiotic entry of fetal oocytes depending on level of Vitamin A deficiency (Li & Clagett-Dame, 2009).
More detailed descriptions and supporting references are found at respective KER pages on AOPwiki. Overall assessments are summarized in Table 3.
Table 3: Empirical evidence for key event relationships of AOP-398.
|
ID |
Assessment score |
Rationale |
|
KER-2401 |
Moderate |
Evidence exists but only for one stressor, WIN18,466. |
|
KER-2477 |
Low |
There are several studies with several different stressors that affect RA levels or RAR activity also affecting meiosis, but there also is conflicting data indicating that atRA is not the sole determining factor governing this process. |
|
KER-2481 |
High |
There are studies with several stressors showing same effect on meiosis and fertility. |
|
KER-2525 |
High |
There are studies with several stressors showing reduced number of follicles and affected ovarian cycling. |
|
KER-394 |
Moderate |
Although strong empirical evidence exists from numerous studies, they are chiefly based on the assessment of a single substance, DEHP. |
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
This AOP is still largely qualitative, as the quantitative understanding between chemical potency and perturbation of KEs are insufficient. This relates to the dose-response relationship between concentrations of atRA in the ovary relative to meiotic initiation of oocytes. It also relates to the relationship between number of lost oocytes during development relative to the oocyte pool/ovarian reserve, as there naturally is a large loss of oocytes during development.
Considerations for Potential Applications of the AOP (optional)
Currently disrupted retinoid signaling is not directly tested for in OECD TG studies; however, in, for example, the identification of endocrine disruptors, the R-modality is highlighted as a pathway that should be included. Hence, this AOP provides added support for inclusion of retinoid signaling-relevant assays to be included in testing or screening strategies.
This AOP can be used to identify chemicals that inhibit ALDH1A activity (e.g., through in vitro assays for retinoic acid biosynthesis) as potential reproductive toxicants, facilitating prioritization for further testing. It also provides a mechanistic basis for linking molecular-level perturbations to reproductive outcomes, supporting weight-of-evidence approaches in regulatory risk assessment and justifying restrictions on chemicals identified as disrupting this pathway.
References
Clagett-Dame M, Knutson D (2011) Vitamin A in Reproduction and Development. Nutrients 3: 385-428
Griswold MD, Hogarth CA, Bowles J, Koopman P (2012) Initiating meiosis: the case for retinoic acid. Biol Reprod 86: 35
Nilsson, C and Working Group (2020) Retinoids in Mammalian Reproduction, with an Initial Scoping Effort to Identify Regulatory Methods. Available at https://norden.diva-portal.org/smash/record.jsf?pid=diva2%3A1424722&dswid=-4522