
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 4
Title
Ecdysone receptor agonism leading to mortality via suppression of Ftz-f1
Short name
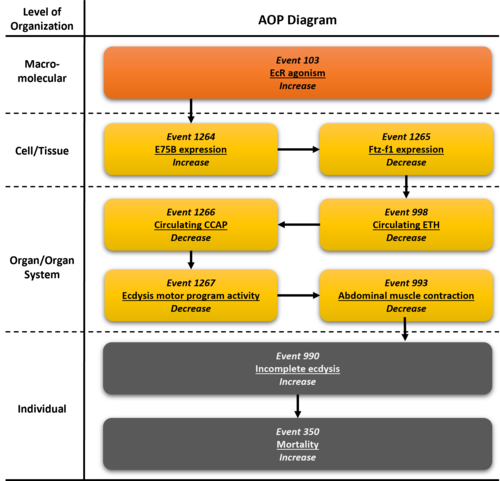
Graphical Representation
Point of Contact
Contributors
- Knut Erik Tollefsen
- You Song
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on October 02, 2025 04:18
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increase, Ecdysone receptor hyperactivation | September 29, 2025 04:47 |
| Decrease, Circulating ecdysis triggering hormone | May 24, 2018 16:34 |
| Increase, Nuclear receptor E75b gene expression | May 24, 2018 16:32 |
| Decrease, Fushi tarazu factor-1 gene expression | September 26, 2025 04:58 |
| Decrease, Circulating crustacean cardioactive peptide | May 24, 2018 16:37 |
| Decrease, Ecdysis motor program activity | September 29, 2025 04:18 |
| Decrease, Abdominal muscle contraction | May 24, 2018 16:41 |
| Increase, Incomplete ecdysis | May 24, 2018 16:41 |
| Increase, Mortality | October 26, 2020 05:18 |
| Increase, EcR hyperactivation leads to Increase, E75b expression | February 09, 2017 03:33 |
| Increase, E75b expression leads to Decrease, Ftz-f1 expression | February 09, 2017 03:33 |
| Decrease, Ftz-f1 expression leads to Decrease, Circulating ETH | February 09, 2017 03:34 |
| Decrease, Circulating ETH leads to Decrease, Circulating CCAP | February 09, 2017 03:34 |
| Decrease, Circulating CCAP leads to Decrease, Ecdysis motor program activity | February 09, 2017 03:35 |
| Decrease, Ecdysis motor program activity leads to Decrease, Abdominal muscle contraction | September 29, 2025 04:17 |
| Decrease, Abdominal muscle contraction leads to Increase, Incomplete ecdysis | December 03, 2016 16:38 |
| Increase, Incomplete ecdysis leads to Increase, Mortality | December 03, 2016 16:38 |
| Tebufenozide | February 09, 2017 03:06 |
| 20-hydroxyecdysone | February 09, 2017 03:06 |
| Ponasterone A | February 09, 2017 03:06 |
| Methoxyfenozide | February 09, 2017 03:42 |
| Halofenozide | February 06, 2017 12:28 |
| Chromafenozide | February 09, 2017 03:41 |
| Cyasterone | February 09, 2017 03:42 |
| Makisterone A | February 09, 2017 03:43 |
| Inokosterone | February 09, 2017 03:43 |
| Ecdysone | February 09, 2017 03:43 |
| RH-5849 | February 09, 2017 03:43 |
Abstract
This Adverse Outcome Pathway (AOP) describes how hyperactivation of the ecdysone receptor (EcR) in arthropods can lead to lethal molting disruption and mortality. Binding of natural ligands (ecdysteroids) to EcR is critical for regulating molting and metamorphosis in insects and crustaceans. However, inappropriate or prolonged activation of EcR by exogenous chemicals disrupts the tightly regulated temporal gene expression cascade required for successful ecdysis. Key events (KEs) include altered expression of early transcription factors (E75B, Ftz-f1), reduction of circulating neuropeptides (CCAP, ETH), impaired motor program activity, and suppression of abdominal muscle contraction, ultimately resulting in incomplete ecdysis and death. This AOP provides mechanistic understanding relevant for environmental chemical safety assessment, particularly regarding pesticides and other compounds targeting insect endocrine systems.
AOP Development Strategy
Context
The development of this AOP is motivated by regulatory needs to understand and predict the impacts of insect growth regulators and other endocrine-active chemicals that target EcR. Such compounds are widely used in pest control but pose risks to non-target arthropods, including pollinators and aquatic invertebrates. The AOP formalizes knowledge of conserved molting endocrine pathways to support hazard identification and potential regulatory screening frameworks.
Strategy
The AOP was developed based on structured literature reviews and expert knowledge. Key sources included primary research on EcR signaling, molting neuropeptides (ETH, CCAP), transcriptional cascades in Drosophila melanogaster and other model insects, as well as crustacean endocrinology. Literature searches were conducted in PubMed, Web of Science, and Scopus using terms such as ecdysone receptor agonists, ecdysis motor program, insect molting disruption, 20-hydroxyecdysone, and ecdysteroid signaling. Priority was given to studies demonstrating experimental perturbation of EcR or downstream KEs and their effects on molting success and survival. Reviews and AOP frameworks (e.g., OECD guidance) were used to ensure structured evaluation and alignment with regulatory relevance.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 103 | Increase, Ecdysone receptor hyperactivation | Increase, EcR hyperactivation |
| KE | 1264 | Increase, Nuclear receptor E75b gene expression | Increase, E75b expression |
| KE | 1265 | Decrease, Fushi tarazu factor-1 gene expression | Decrease, Ftz-f1 expression |
| KE | 988 | Decrease, Circulating ecdysis triggering hormone | Decrease, Circulating ETH |
| KE | 1266 | Decrease, Circulating crustacean cardioactive peptide | Decrease, Circulating CCAP |
| KE | 1267 | Decrease, Ecdysis motor program activity | Decrease, Ecdysis motor program activity |
| KE | 993 | Decrease, Abdominal muscle contraction | Decrease, Abdominal muscle contraction |
| AO | 990 | Increase, Incomplete ecdysis | Increase, Incomplete ecdysis |
| AO | 350 | Increase, Mortality | Increase, Mortality |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Increase, EcR hyperactivation leads to Increase, E75b expression | adjacent | High | |
| Increase, E75b expression leads to Decrease, Ftz-f1 expression | adjacent | High | |
| Decrease, Ftz-f1 expression leads to Decrease, Circulating ETH | adjacent | Moderate | |
| Decrease, Circulating ETH leads to Decrease, Circulating CCAP | adjacent | Moderate | |
| Decrease, Circulating CCAP leads to Decrease, Ecdysis motor program activity | adjacent | Moderate | |
| Decrease, Ecdysis motor program activity leads to Decrease, Abdominal muscle contraction | adjacent | Moderate | |
| Decrease, Abdominal muscle contraction leads to Increase, Incomplete ecdysis | adjacent | Moderate | |
| Increase, Incomplete ecdysis leads to Increase, Mortality | adjacent | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Juvenile | High |
| Adult | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | Moderate |
Overall Assessment of the AOP
The overall weight of evidence supporting this AOP is strong, with high biological plausibility and multiple lines of empirical support linking EcR hyperactivation to lethal molting disruption. The key endocrine and neuropeptide signaling pathways involved are highly conserved across arthropods, increasing confidence in broad taxonomic applicability, particularly to insects and crustaceans undergoing ecdysis. Essentiality of key events is well demonstrated through genetic and pharmacological manipulations, and direct causal linkages between upstream molecular initiating events and downstream organism-level outcomes are supported by both in vitro and in vivo studies. While quantitative understanding remains incomplete, especially regarding cross-species dose-response relationships, the evidence base is sufficient to support application in chemical screening, prioritization, and risk assessment. The AOP is considered reliable for use in evaluating the hazards of EcR agonists and related endocrine-active chemicals, with clear regulatory relevance to the assessment of insect growth regulators and protection of non-target arthropods.
Domain of Applicability
-
Taxa: Arthropods, primarily insects (Diptera, Lepidoptera, Coleoptera) and crustaceans.
-
Life stage: Juvenile and larval stages undergoing molting.
-
Sex: Both sexes are equally affected, as molting regulation is not sex-specific.
-
Other considerations: The pathway is most relevant in holometabolous insects but is applicable across arthropods where molting is controlled by EcR signaling.
Essentiality of the Key Events
-
EcR hyperactivation (MIE): Genetic or pharmacological overactivation prevents correct timing of molting cascades, leading to lethality.
-
E75B and Ftz-f1 expression changes: Knockout or overexpression experiments demonstrate disruption of subsequent endocrine signals and molting success.
-
Circulating ETH/CCAP: Blocking or reducing peptide release suppresses ecdysis behavior. Rescue experiments with exogenous peptides restore motor program activity.
-
Motor program activity and abdominal contractions: Neurophysiological studies show that impaired muscle activity directly prevents exuviation.
-
Incomplete ecdysis: Universally essential for linking endocrine dysfunction to mortality.
Overall, essentiality of KEs is supported by direct experimental evidence in multiple model arthropods.
Evidence Assessment
KER 103 → 1264 (EcR hyperactivation → Increased E75B expression)
-
Biological plausibility: Strong. E75B is a primary-response gene in the ecdysone signaling cascade. Overactivation of EcR drives prolonged or elevated expression of E75B.
-
Empirical support: Multiple in vitro and in vivo studies (e.g., Drosophila, lepidopterans) show dose-dependent induction of E75B following EcR agonist exposure.
-
Uncertainties: Quantitative thresholds vary across taxa.
KER 103 → 1265 (EcR hyperactivation → Decreased Ftz-f1 expression)
-
Biological plausibility: Strong. Ftz-f1 is normally induced after a decline in ecdysone signaling, serving as a competence factor for subsequent developmental transitions. Sustained EcR activity suppresses Ftz-f1 expression.
-
Empirical support: Genetic experiments in Drosophila demonstrate that EcR hyperactivation prevents Ftz-f1 induction, leading to molting defects.
-
Uncertainties: The precise timing of downregulation differs between insect species.
KER 1265 → 998 (Decreased Ftz-f1 expression → Decreased circulating ETH)
-
Biological plausibility: Moderate to strong. ETH release from Inka cells requires proper transcriptional programming, in which Ftz-f1 plays a permissive role.
-
Empirical support: ETH levels are reduced in Ftz-f1 mutant or RNAi knockdown insects, with corresponding ecdysis failure.
-
Uncertainties: Direct mechanistic links in crustaceans are less studied.
KER 1264 → 1266 (Increased E75B expression → Decreased circulating CCAP)
-
Biological plausibility: Moderate. E75B dysregulation affects downstream neural peptide release patterns, including crustacean cardioactive peptide (CCAP).
-
Empirical support: Studies in Manduca sexta and Drosophila show disrupted CCAP neuron activation in response to EcR agonists.
-
Uncertainties: Empirical dose-response data linking E75B overexpression directly to CCAP suppression are limited.
KER 1266 → 1267 (Decreased circulating CCAP → Decreased ecdysis motor program activity)
-
Biological plausibility: Strong. CCAP is required to initiate and maintain the motor patterns driving ecdysis behavior.
-
Empirical support: Ablation or silencing of CCAP neurons abolishes normal ecdysis behavior in insects. Exogenous CCAP restores motor program activity in some experimental systems.
-
Uncertainties: Quantitative thresholds for peptide levels triggering full motor program are not well established.
KER 998 → 993 (Decreased circulating ETH → Decreased abdominal muscle contraction)
-
Biological plausibility: Strong. ETH directly triggers ecdysis motor output by activating central nervous system circuits.
-
Empirical support: ETH knockout or peptide inhibition prevents abdominal contractions in Drosophila larvae. ETH injection can rescue the phenotype.
-
Uncertainties: Effects may be stage-dependent.
KER 1267 → 990 (Decreased ecdysis motor program activity → Incomplete ecdysis)
-
Biological plausibility: Strong. Motor program activity is essential for successful shedding of the old cuticle.
-
Empirical support: Neurophysiological and behavioral studies show that impaired motor activity directly correlates with failed or incomplete ecdysis.
-
Uncertainties: Variation in motor outputs among species may influence severity.
KER 993 → 990 (Decreased abdominal muscle contraction → Incomplete ecdysis)
-
Biological plausibility: Strong. Abdominal contractions generate the mechanical force required for cuticle shedding.
-
Empirical support: Pharmacological or genetic inhibition of abdominal muscle contraction prevents complete ecdysis in Drosophila and other insects.
-
Uncertainties: Contribution of other body muscles (e.g., thoracic) not fully quantified.
KER 990 → 350 (Incomplete ecdysis → Increased mortality)
-
Biological plausibility: Strong. Inability to shed the old cuticle is incompatible with survival.
-
Empirical support: High mortality rates are consistently observed in laboratory and field studies where molting is disrupted by EcR agonists or neuropeptide blockers.
-
Uncertainties: Mortality timing (immediate vs delayed) may vary with species and stage.
Known Modulating Factors
-
Temperature: Affects hormone turnover and molting periodicity.
-
Nutritional status: Influences steroid hormone synthesis and peptide release.
-
Species-specific sensitivity: Different insects and crustaceans vary in susceptibility to EcR agonists.
-
Developmental stage: Early versus late larval stages may have differing vulnerability.
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Quantitative relationships between EcR activation and downstream transcriptional responses are partially characterized, especially in Drosophila. Dose-response data for diacylhydrazine insecticides provide empirical linkage between exposure and incomplete ecdysis. However, quantitative models are still limited and largely taxon-specific.
Considerations for Potential Applications of the AOP (optional)
This AOP has clear utility for both scientific and regulatory applications related to the environmental assessment of endocrine-active substances targeting arthropod molting. Because EcR is the primary molecular target of many insect growth regulators (IGRs), this pathway provides a mechanistic framework for interpreting how chemical binding at the receptor level translates to population-level adverse outcomes such as mortality.
From a regulatory perspective, the AOP can inform the development and refinement of OECD test guidelines addressing arthropod development and molting. It can also support the design of integrated approaches to testing and assessment (IATA), in which data from in vitro receptor-binding assays, transcriptomic biomarkers (e.g., E75B, Ftz-f1), and neuropeptide measurements can be combined with higher-tier organismal studies to streamline hazard characterization. Furthermore, the pathway may facilitate the identification of molecular biomarkers that can be incorporated into early screening assays, reducing reliance on animal-intensive in vivo tests.
The AOP is also relevant for chemical grouping and read-across approaches, particularly for compounds within the diacylhydrazine class and other EcR agonists. Structure–activity relationship ((Q)SAR) models or chemical profilers trained on these endpoints could help predict EcR activity and prioritize substances for further testing.
For ecological risk assessment, this AOP highlights the potential for population-level impacts on non-target arthropods, including beneficial insects (e.g., pollinators) and aquatic crustaceans. Given the essential role of molting for growth and reproduction, disruptions captured in this pathway provide a mechanistic basis to link molecular initiating events to ecologically relevant endpoints.
Overall, this AOP offers opportunities to improve chemical safety decision-making by providing a structured framework to integrate mechanistic data into regulatory contexts, enabling screening, prioritization, and risk assessment of chemicals that act through EcR hyperactivation.
References
Song, Y.; Villeneuve, D. L.; Toyota, K.; Iguchi, T.; Tollefsen, K. E., 2017. Ecdysone receptor agonism leading to lethal molting disruption in arthropods: review and adverse outcome pathway development. Environ Sci Technol, 51, (8), 4142-4157.
Song, Y., Evenseth, L.M., Iguchi, T., Tollefsen, K.E., 2017. Release of chitobiase as an indicator of potential molting disruption in juvenile Daphnia magna exposed to the ecdysone receptor agonist 20-hydroxyecdysone. J Toxicol Environ Health A, 1-9
Fay, K. A., Villeneuve, D. L., LaLone, C. A., Song, Y., Tollefsen, K. E. and Ankley, G. T., 2017. Practical approaches to adverse outcome pathway (AOP) development and weight of evidence evaluation as illustrated by ecotoxicological case studies. Environ. Toxicol. Chem. 36(6):1429-1449.
Miyakawa, H., Sato, T., Song, Y., Tollefsen, K.E., Iguchi, T., 2017. Ecdysteroid and juvenile hormone biosynthesis, receptors and their signaling in the freshwater microcrustacean Daphnia. J Steroid Biochem Mol Biol. pii: S0960-0760(17), 30370-30379.