
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 490
Title
Co-activation of IP3R and RyR leads to reduced IQ and increased socio-economic burden through non-cholinergic mechanisms
Short name
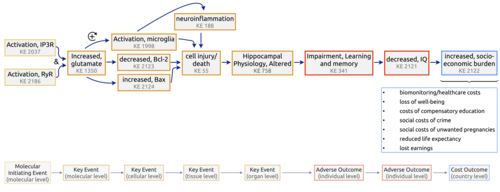
Graphical Representation
Point of Contact
Contributors
- Thibaut Coustillet
- Karine Audouze
- Xavier COUMOUL
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on December 23, 2025 08:41
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increased, glutamate | October 11, 2021 14:58 |
| Activation, Microglia | April 07, 2022 09:25 |
| Neuroinflammation | July 15, 2022 09:54 |
| Hippocampal Physiology, Altered | July 24, 2024 23:15 |
| Impairment, Learning and memory | April 27, 2026 05:48 |
| decreased, Bcl-2 expression | May 29, 2025 08:01 |
| increased, Bax expression | May 20, 2025 11:22 |
| decreased, Intellectual Quotient | May 29, 2025 12:51 |
| Increase, Cell injury/death | May 27, 2024 07:23 |
| Ryanodine receptor activation | May 29, 2025 05:51 |
| Inositol triphosphate receptor activation | May 29, 2025 05:58 |
| increased, socio-economic burden | June 18, 2025 03:37 |
| Activation, IP3R leads to Increased, glutamate | September 08, 2023 09:12 |
| Activation, RyR leads to Increased, glutamate | September 08, 2023 09:12 |
| Increased, glutamate leads to decreased, Bcl-2 | April 04, 2023 10:36 |
| Increased, glutamate leads to increased, Bax | April 04, 2023 10:36 |
| Increased, glutamate leads to Microglia activation | April 03, 2023 06:05 |
| Microglia activation leads to Neuroinflammation | April 03, 2023 06:06 |
| decreased, Bcl-2 leads to Cell injury/death | April 04, 2023 10:37 |
| increased, Bax leads to Cell injury/death | April 04, 2023 10:38 |
| Neuroinflammation leads to Cell injury/death | November 07, 2019 10:27 |
| Cell injury/death leads to Hippocampal Physiology, Altered | April 05, 2023 03:48 |
| Hippocampal Physiology, Altered leads to Impairment, Learning and memory | April 03, 2023 06:07 |
| Impairment, Learning and memory leads to decreased, IQ | April 04, 2023 10:39 |
| decreased, IQ leads to increased, socio-economic burden | April 04, 2023 10:35 |
| Chlorpyrifos | July 27, 2022 04:02 |
| Chlorpyrifos oxon | April 03, 2023 03:45 |
| Organophosphates | November 29, 2016 21:20 |
Abstract
Prenatal and neonatal periods are critical windows of vulnerability to environmental chemical contaminants. A growing number of epidemiological and toxicological studies suggest that exposure to pesticides during these periods may adversely affect children's health at birth as well as their developmental trajectory, with potential delayed long-term consequences. An Adverse Outcome Pathway (AOP) initiated after exposure to organophosphate pesticides, such as chlorpyrifos (CPF), resulting in decreased Intellectual Quotient (IQ) through non-cholinergic mechanisms was constructed using expert knowledges and artificial intelligence tool (such as the third-party tool AOP-helpFinder).
Indeed, despite the well-known mode of action of organophosphates as acetylcholinesterase (AchE) inhibitors, some studies have demonstrated adverse effects on neurodevelopment at doses insufficient to inhibit AchE, thus suggesting the involvement of so-called non-cholinergic mechanisms. Additionally, we have enriched the AOP by connecting the KE ‘decreased, IQ’ to a special KE ‘increased, socio-economic burden’ (see corresponding article for details). This is based on an increasing number of studies and models that highlight the long-term socio-economic impacts of stress exposure. Considering the culminating 'population' level of organization in the AOP framework (i.e., adverse effects occuring in multiple individuals within a community), we propose that this new concept, termed Cost Outcome Pathway (COP), can have significant implications at a national scale (country level).
AOP Development Strategy
Context
The exposome concept, proposed by C.P. Wild in 2005, gathers all exposure sources (environmental chemicals, biological agents, physical & socio-economic factors...) to which an individual is subjected, from conception to death (Wild, C. P., 2005). Complementary to the genome approach, the exposome is an important notion for the understanding of how gene-environment interactions trigger diseases (Vineis, P., et al., 2020). The environmental contribution to the development of chronic diseases could be in the range of 70% to 90% (Rappaport, S. M., & Smith, M. T., 2010).
Agrochemicals that include biocides, herbicides, fungicides, and insecticides contribute chronically and significantly to the chemical exposome worldwide due to their extensive use in agriculture and domestic environments. In Europe, different families of man-made pesticides have been used in waves since the 1950s, notably in agriculture. For example, the organochlorines (e.g., DDT) were replaced by organophosphates (OPs), which are now progressively replaced by pyrethroids. Despite their gradual substitutions by pyrethroids in Europe, some OPs including chlorpyrifos (CPF), are still widely used and sometimes benefit from exemptions for their use (e.g., France, spinach cultivation). Recently, the National Institute of Health and Medical Research in France (INSERM) published a collective expert report identifying strong presumed risks associated with exposure to OPs (INSERM, 2022) (non-Hodgkin's lymphomas or cognitive disorders in adults and altered motor, cognitive and sensory capacities in children due to exposure during pregnancy).
Although exposure to risk factors increases the probability of acquiring an adverse outcome, the window, timespan, and dose of exposure are elements to be considered when assessing the hazardousness of a chemical. In line with the Developmental Origins of Health and Disease (DOHaD) theory, which emphasizes linkage between prenatal and postnatal exposure to environmental factors and the risk of developing several diseases later in life (Gluckman, P. D., et al., 2008), the perinatal period is a critical window of high vulnerability for neurodevelopment (Silbereis, J. C., et al., 2016). Therefore, children’s exposure to environmental pollutants (through contaminated food/air/water, direct contact through crop spraying, domestic use, etc.), may result in behavioral and/or cognitive disorders, potentially persistent through adult life (Arora, M., et al., 2017).
To have a better understanding of the putative involvement from the chemical exposome to health effects, new and innovative methods are needed. New Approaches Methodologies (NAMs) or next generation risk assessment (NGRA), referring to non-animal- based approaches, can provide information on chemical hazards and inform risk assessment (van der Zalm, A. J., et al., 2022). In 2010, Ankley et al. formalized the Adverse Outcome Pathway (AOP) framework which compile existing biological knowledge in a structured linear representation at various levels of the biological organization (molecular, cellular, individual, etc.) (Ankley, G. T., et al., 2010). Advancements in technologies, coupled with an uptick in data volume and the availability of diverse toxicological data from various sources (omics, high throughput assays, literature), facilitate the development of complex, but realistic toxicological models including AOPs (Kleinstreuer, N., & Hartung, T., 2024). As the AOP construction could be very time-intensive to compile existing heterogeneous knowledge from structured- and non-structured data, innovative computational methods based on artificial intelligence (AI), and data mining technologies are well suited. AI allows to identify, extract, and compile relevant sparse information from the wealth of available open-source data, and can be used for predictive toxicology (e.g., Abstract Sifter (Baker, N., et al., 2017), the ComptoxAI tools (Hartung T., 2023; Lin, Z., & Chou, W. C. 2022)) or others computational approaches (Baudiffier, D., et al., 2024).
Recently, AI and text-mining were used to develop AOP-helpFinder (Carvaillo, J. C., et al., 2019; Jaylet, T., et al., 2023), a tool to automatically identify, extract, and prioritize knowledge from the literature (PubMed). AOP-helpFinder has already been successfully used in several studies, as well as in the development of novel AOPs that were submitted to the AOP-Wiki database (e.g., AOP IDs 439, 441, 490, 493, 494 and 497).
The AOP concept can be extended to the socio-economic consequences of AOs (or diseases) beyond its biological information (MIE and KE). Diseases may cause suffering, moral distress, loss of earnings, and set limits in the present and future life of affected individuals in addition to decrease their quality of life, as many dimensions that are not captured by the depiction of the biological KEs in AOP. Due to these consequences, and the need for healthcare and other social cares (psychological support, economic support, ...), AOs may imply costs to the society that need to be reflected in AOPs as relevant consequences from chemical exposure. Notwithstanding the fact that there are studies that have already outlined, e.g., disability-adjusted life years due to diseases (Menzies, N. A., et al., 2023), the socio-economic costs of disease need to be more often considered and formalized, particularly in the case of exposome-induced diseases.
Here we proposed a new concept, the Cost Outcome Pathway (COP) that integrates the socio-economical cost (Cost Outcome, CO) into the AOP framework. As a case study, one AOP related to agrochemicals exposure (OPs including CPF) during neurodevelopment was designed using the AOP-helpFinder tool and scientific expertise’s. The proposed AOP was extended to a COP, by adding a ‘socio-economic KER’ describing socio-economic consequence.
Strategy
- Text-mining tool
- Scientific expertise & manual curation
- Refinement
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 2186 | Ryanodine receptor activation | Activation, RyR |
| MIE | 2037 | Inositol triphosphate receptor activation | Activation, IP3R |
| KE | 1350 | Increased, glutamate | Increased, glutamate |
| KE | 2123 | decreased, Bcl-2 expression | decreased, Bcl-2 |
| KE | 2124 | increased, Bax expression | increased, Bax |
| KE | 1998 | Activation, Microglia | Microglia activation |
| KE | 188 | Neuroinflammation | Neuroinflammation |
| KE | 55 | Increase, Cell injury/death | Cell injury/death |
| KE | 758 | Hippocampal Physiology, Altered | Hippocampal Physiology, Altered |
| AO | 341 | Impairment, Learning and memory | Impairment, Learning and memory |
| AO | 2121 | decreased, Intellectual Quotient | decreased, IQ |
| AO | 2122 | increased, socio-economic burden | increased, socio-economic burden |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Activation, IP3R leads to Increased, glutamate | adjacent | Moderate | |
| Activation, RyR leads to Increased, glutamate | adjacent | Moderate | |
| Increased, glutamate leads to decreased, Bcl-2 | adjacent | High | |
| Increased, glutamate leads to increased, Bax | adjacent | High | |
| Increased, glutamate leads to Microglia activation | adjacent | High | |
| Microglia activation leads to Neuroinflammation | adjacent | High | |
| decreased, Bcl-2 leads to Cell injury/death | adjacent | High | |
| increased, Bax leads to Cell injury/death | adjacent | High | |
| Neuroinflammation leads to Cell injury/death | adjacent | High | |
| Cell injury/death leads to Hippocampal Physiology, Altered | adjacent | High | |
| Hippocampal Physiology, Altered leads to Impairment, Learning and memory | adjacent | High | |
| Impairment, Learning and memory leads to decreased, IQ | adjacent | Moderate | |
| decreased, IQ leads to increased, socio-economic burden | adjacent | High |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Perinatal |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Mixed |
Overall Assessment of the AOP
The evidence supporting all the KERs and the ensuing COP was reviewed according to 7 features. These criteria are a set of requirements for providing adequate evidence of a causal relationship between two KEs.
| Upstream event | Downstream event | KER ID | Biological applicability domain | Biological plausibility | Essentiality | Empirical evidence | Uncertainties & Inconsistencies | Modulation factors | Measurement method (downstream event) | References |
|---|---|---|---|---|---|---|---|---|---|---|
|
Activation, RyR |
Increased, glutamate |
3036 |
Mammals |
Moderate |
Moderate |
Moderate |
Low |
Co-activation with IP3R |
Microdialysis, high-performance liquid chromatography & o-phthalalde hyde-derived fluorescence detection |
Mori et al., 2005 |
|
Activation, IP3R |
Increased, glutamate | 3037 | Mammals | Moderate | Moderate | Moderate | Low |
Co-activation with RyR |
Extreme liquid chromatograph, o-phthalalde hyde derivatized fluorescence detection |
Yamamura et al., 2009 |
|
Increased, glutamate |
decreased, Bcl-2 expression |
2868 |
Mammals |
High |
Moderate |
High |
Low |
- |
Western Blot |
Nishi et al., 1999 Phoraksa et al., 2023 |
|
Increased, glutamate |
increased, Bax expression |
2869 |
Mammals |
High |
Moderate |
High |
Low |
- |
Western Blot |
Nishi et al., 1999 Phoraksa et al., 2023 |
|
Increased, glutamate |
Activation, Microglia |
2861 |
Mammals |
Moderate |
High |
High |
Low |
- |
Light microscopic examination Flowcytometry (CD11b) |
Domercq et al., 2013 Sunkaria et al., 2014 |
|
Activation, Microglia |
Neuroinflammation |
2862 |
Mammals |
High |
High |
High |
Low |
- |
Sandwich ELISA |
Sunkaria et al., 2014 |
|
Neuroinflammation |
Cell injury/death |
1687 |
Mammals |
High |
High |
High |
Low |
- |
Trypan blue staining |
Boje & Arora, 1992 |
|
decreased, Bcl-2 expression |
Cell injury/death |
2870 |
Mammals |
High |
Moderate |
High |
Low |
- |
Western Blot of cleaved caspase 3, Hoechst 33258 staining |
Lee et al., 2014 |
|
increased, Bax expression |
Cell injury/death |
2871 |
Mammals | High | Moderate | High | Low | - |
Western Blot of cleaved caspase 3, Hoechst 33258 staining |
Lee et al., 2014 |
| Cell injury/death |
Hippocampal Physiology, Altered |
2875 |
rodents |
High |
High |
High |
Low | - |
Hoechst staining & GFAP staining TUNEL |
Wu et al., 2021 Huang et al., 2012 |
|
Hippocampal Physiology, Altered |
Impairment, Learning and memory |
2864 |
rodents |
High | High | High | Low | - |
Morris water maze test |
Huang et al., 2012 Su et al., 2014 |
|
Impairment, Learning and memory |
decreased, Intellectual Quotient |
2872 |
7-year-old children |
Moderate |
High |
Moderate |
Moderate |
lifestyle habits |
WISC-IV |
Rauh et al., 2011 Bouchard et al., 2011 |
Regarding the uncertainties and inconsistencies, most KERs were labeled as ‘low’ as the scientific support was considered strong. However, the KER ID 2872 ‘Impairment, Learning and memory leads to decreased, Intellectual Quotient’ was considered with moderate uncertainty. In children aged 8 to 9, 3rd and 4th grade academic performances were both significantly influenced by IQ and study habits but not by short memory (Quilez-Robres, A., et al., 2021).
Study habits are non-cognitive variables considered to be the learning patterns that pupils set outside school, i.e. the systematic or disorganized, efficient, or non-productive way in which each student studies. As these so-called study habits can partly shape school results in the same way as IQ, we believe we can treat them as KER Known Modulating Factors, i.e. ‘modulating factors/variables known to alter quantitative aspects of the response-response function that describes the relationship between the two KEs’. In a wider context, we considered the personal lifestyle habits to be the KER Known Modulating Factors, i.e. habits not necessarily school dependent. By way of example, regular breakfast consumption has been correlated with a higher IQ in children (Liu, J., et al., 2013) while consuming screens such as television while eating has been linked to delayed language development in children (Yang, S., et al., 2024).
Domain of Applicability
- The biological applicability domain of the COP focuses on human beings. Although IQ can decline at any age, we aimed to characterize the reduction during neurodevelopment. However, the consequences of a drop in IQ can be felt over the long term in adults since several socio-economic impacts of IQ loss in early life (compensatory education, reduction in earnings, ...) occurs throughout life.
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
| lifestyle habits | modulates Intellectual Quotient |
|
- See section Overall Assessment of the AOP for details.
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
This COP developed at an individual level, could be used at society level to assess chemicals risk reduction policies, and maybe linked to kinetics and toxicodynamic models such as quantitative AOP. At the EU level, consideration, and evaluation of IQ points loss due to exposure to chemicals has been used in policymaking in the framework of the EU REACH regulation, to help decide on market restrictions for lead in ammunition (ECHA, 2022) and lead in PVC (ECHA, 2017). Another example of use in policymaking is in France to justify new monitoring and intervention thresholds for lead blood levels (HCSP, 2014). This use in policy making relies on the addition of individual effects over the population affected by that policy. It is of note that there could be, in the case of IQ, economic systemic effects since a collective decrease in cognitive capacities could have long-term and far-reaching implications in terms of functioning of the society in many areas (education, innovation and research, ...).
The ongoing interest for AOPs is still growing in recent years. The scientific community is making a collaborative endeavor to enhance existing AOPs and propose new ones. The AOP framework is constantly evolving, and new proposals are put forward thanks to the crowdsourced collaboration. Six years after the original concept was proposed, J. Teeguarden et al., proposed the notion of Aggregate Exposure Pathway (AEP) to ramify AOPs by setting out an aggregation pathway for a chemical (Teeguarden, J. G., et al., 2016). In this case, the pathway illustrates the route taken by the chemical since its original use to the triggering of the MIE of the AOP. It is then possible to design new structures called AEP-AOP (Hines, D. E., et al., 2018).
The main innovation here is the evolution from AOP towards COP by adding a socio-economic term at the end of the toxicity pathway. On the one hand, the study demonstrated the scalability of the AOP-helpFinder tool, initially created in relation to conventional AOP events (MIEs, KEs, AOs). The use of such AI-based tools could be applied to any type of information (e.g., COs) and no longer be restricted to the PubMed database in such a way that text-mining and artificial intelligence technologies open up new horizons for massive data mining.
On the other hand, the COP framework can be applied to any AOP if there is a connection between an adverse effect and a socio-economic cost. It then reflects the economic burden borne by individuals and the care society has to take care of, that is the consequence of a biological endpoint. The last adverse effect making the AO-CO connection can be different from the decrease of IQ, as ADHD or autism are both associated with neurotoxicological effects and are socio-economic relevance.
More generally, COPs have a wider potential interest because several pathologies associated with chemical exposure can lead to significant economic costs for society, such as cancer or obesity (Luengo-Fernandez, R., et al., 2013; Wang, Y. C., et al., 2022), and many others. Recently, an excellent example has been the covid-19 pandemic which was subject to a colossal number of studies, some of which assessing the cost-of-illness of the disease (Wang, H., et al., 2023).
If AOP and diseases were more systematically extended as COPs, this would allow to describe and have a common understanding of the complex interconnections between exposures, socio-economic consequences. Therefore, COPs appear as a powerful tool to represent the exposome in a more holistic and consistent way, with potential to better design wide-ranging policy interventions to reduce the health and socio-economic impacts of environmental exposure to chemicals.