
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 392
Title
Decreased fibrinolysis and activated bradykinin system leading to hyperinflammation
Short name
Graphical Representation
Point of Contact
Contributors
- Penny Nymark
- Merlin Mei
- Sabina Halappanavar
- Marvin Martens
Coaches
- Cinzia La Rocca
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.96 | Under Development |
This AOP was last modified on April 29, 2023 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Fibrinolysis, decreased | October 19, 2021 11:33 |
| Bradykinin system, hyperactivated | January 24, 2022 11:39 |
| Increased, secretion of proinflammatory mediators | May 17, 2023 15:18 |
| Increased, recruitment of inflammatory cells | May 12, 2023 17:03 |
| Hyperinflammation | December 29, 2021 02:29 |
| Hypofibrinolysis leads to Bradykinin, activated | April 20, 2021 02:45 |
| Hypofibrinolysis leads to Increased proinflammatory mediators | February 07, 2023 23:43 |
| Bradykinin, activated leads to Hypofibrinolysis | January 24, 2022 11:49 |
| Bradykinin, activated leads to Increased proinflammatory mediators | January 14, 2022 15:02 |
| Bradykinin, activated leads to Hyperinflammation | October 19, 2021 16:19 |
| Increased proinflammatory mediators leads to Recruitment of inflammatory cells | May 18, 2023 12:46 |
| Recruitment of inflammatory cells leads to Hyperinflammation | February 08, 2023 09:25 |
| Sars-CoV-2 | September 09, 2022 05:09 |
| nanoparticles | December 21, 2016 09:40 |
| nanomaterials | April 22, 2021 09:28 |
| Titanium dioxide nanoparticles | August 29, 2022 08:22 |
| Iron nanoparticles | August 29, 2022 08:23 |
Abstract
This AOP describes a sequence of molecular, cellular and tissue events associated with immune response, and leading to an uncontrolled, self-perpetuating inflammatory response referred to as hyperinflammation. The AOP is developed with the intent to clarify the complex inflammatory processes associated with the ongoing COVID-19 pandemic caused by the coronavirus SARS-CoV-2. In the broader context, this AOP is applicable to other fields of research including toxicology, with a specific focus on the toxicity and inflammatory elements of nanomaterials. AOP 392 is referred to as a “hub” AOP since it is hypothesized to describe and connect the process of hyperinflammation to other related AOPs, including organ, individual, population, or community-focused AOPs. Such AOPs can include the COVID-19 associated AOPs currently in development (e.g. AOPs 319 and 320) or previously established AOPs (e.g. AOP 173 and 302). In addition, the AOP may be used as a basis for development of new AOPs fit for specific purposes, including e.g. connection to MIEs relevant for SARS-CoV-2 such as the ACE2 binding KE, which leads to the imbalanced fibrinolysis/bradykinin activation MIEs (AOP392).
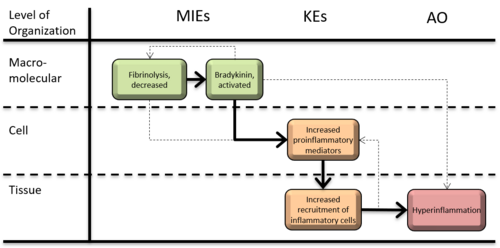
Fibrinolysis (MIE) is the breakdown of fibrin in blood clots which prevents blood clots from growing too large, and hypo-fibrinolysis occurs when the fibrinolysis process becomes impaired. High levels of the SERPINE1 inhibitor and dysregulation of the uPA/uPAR system found in COVID-19 patients are the main weights of evidence found in support of hypo-fibrinolysis(KE1866). Bradykinin is a peptide that plays an important role in blood pressure regulation and inflammation. Activation of the bradykinin system, causing a bradykinin upregulation, is seen in BAL samples of COVID-19 patients, as well as evidence of B1 and B2 receptor binding in COVID-19 patients(KE1867). Decrease of fibrinolysis and bradykinin upregulation causes inflammation, which evolves into hyperinflammation, an uncontrolled and self-perpetuating inflammatory process that results in tissue damage. Further exploration of how inflammation becomes uncontrolled and evolves into hyperinflammation is needed. There are high serum levels of pro-inflammatory cytokines such as IL-6, CXCL8, and TNF, indicating inflammation (KE1496 and 1497) as well as evidence of high D-dimer, CRP, neutrophil, and a lack of lymphocytes, all evidence of hyperinflammation(AO 1868).
The WoE involving nanomaterials, the other stressor associated with the AOP, is that certain nanomaterials directly interact with coagulation factor XII (FXII). Several proteins of the coagulation system, including fibrinogen and kallikrein, bind to TiO2 and α-Fe2O3 nanoparticles (NPs), and induce clot formation triggered by FXII. This leads to fibrinolysis decrease (KE1866). Nanomaterial activation of FXII also activates the bradykinin system and generates increased bradykinin (KE1867). When proteins of the coagulation system bind to TiO2 and α-Fe2O3 NPs, it causes the release of proinflammatory cytokines, leading to inflammation (KE1496 and 1497), and eventually leading to hyperinflammation (AO1868).
COVID-19 has been a global pandemic that the world has faced for over a year, making this AOP relevant to the understanding of the disease. The other applicability of the AOP is through nanomaterials, including examples such as TiO2, which can promote activation of this AOP in the same mechanism. Nanoparticles such as TiO2 are included in many essential items of day to day life, including food and drug treatments, making this AOP applicable to the future of human health.
AOP Development Strategy
Context
AOP 392 is developed as a part of the CIAO project, Modelling the Pathogenesis of COVID-19 Using the Adverse Outcome Pathway (AOP). The overall goal was to organize, consolidate, and understand the vast amount of data that is constantly evolving as a result of the COVID-19 pandemic and identify knowledge gaps that may be missing using the AOP framework. Many AOPs were developed as a part of the CIAO project, each AOP focusing on a specific element of the SARS-COV-2 virus responses in humans. AOP392 focuses on the inflammatory responses.
AOP 392 covers a set of events that act in concert towards perturbing inflammation. Hypo-fibrinolysis represents the first event (KE1866), in which interaction with SARS-COV-2 or nanomaterials causes decreased fibrinolytic response. The activation of the bradykinin system(KE1867) (MIE2), also instigated by SARS-COV-2 or nanomaterial stressors, can overactivate the bradykinin system and increases bradykinin production. As a result of hypo-fibrinolysis (KE1866), and bradykinin activation (KE1867), there is an increase in pro-inflammatory mediators secretion (KE1496), which signals the recruitment of pro-inflammatory cells into the lungs (KE1497). This KE process represents the changes occurring in inflammation. The increase in pro-inflammatory cells levels lead to a higher neutrophil to lymphocyte ratio, increased CRP, and high D-dimer and ferritin levels, all hallmarks of a hyperinflammatory state (KE1868). Hyperinflammation plays a critical role in driving the severity of the COVID-19 disease. Further exploration of the inflammation to hyperinflammation process is needed.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1866 | Fibrinolysis, decreased | Hypofibrinolysis |
| MIE | 1867 | Bradykinin system, hyperactivated | Bradykinin, activated |
| KE | 1496 | Increased, secretion of proinflammatory mediators | Increased proinflammatory mediators |
| KE | 1497 | Increased, recruitment of inflammatory cells | Recruitment of inflammatory cells |
| AO | 1868 | Hyperinflammation | Hyperinflammation |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| humans | Homo sapiens | High | NCBI |