
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 451
Title
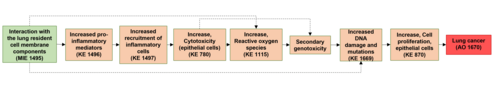
Interaction with lung resident cell membrane components leads to lung cancer
Short name
Graphical Representation
Point of Contact
Contributors
- Penny Nymark
- Sabina Halappanavar
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 29, 2023 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Substance interaction with the lung resident cell membrane components | May 17, 2023 15:10 |
| Increased, secretion of proinflammatory mediators | May 17, 2023 15:18 |
| Increased, recruitment of inflammatory cells | May 12, 2023 17:03 |
| Increase, Cytotoxicity (epithelial cells) | September 16, 2017 10:16 |
| Increase, Reactive oxygen species | June 12, 2025 01:27 |
| Secondary genotoxicity | May 25, 2022 05:25 |
| Increased, DNA damage and mutation | August 13, 2019 05:41 |
| Increase, Cell Proliferation | December 10, 2024 15:01 |
| Lung cancer | August 13, 2019 05:34 |
| Interaction with the lung cell membrane leads to Increased proinflammatory mediators | February 20, 2025 07:12 |
| Interaction with the lung cell membrane leads to Increase, ROS | May 25, 2022 05:44 |
| Increased proinflammatory mediators leads to Recruitment of inflammatory cells | May 18, 2023 12:46 |
| Interaction with the lung cell membrane leads to Increased, DNA damage and mutation | May 25, 2022 05:47 |
| Recruitment of inflammatory cells leads to Increase, Cytotoxicity (epithelial cells) | May 25, 2022 05:32 |
| Increase, Cytotoxicity (epithelial cells) leads to Secondary genotoxicity | May 25, 2022 05:51 |
| Increase, Cytotoxicity (epithelial cells) leads to Increase, ROS | May 25, 2022 05:35 |
| Increase, ROS leads to Secondary genotoxicity | May 25, 2022 05:35 |
| Secondary genotoxicity leads to Increased, DNA damage and mutation | May 25, 2022 05:35 |
| Increased, DNA damage and mutation leads to Increase, Cell Proliferation | July 03, 2019 11:53 |
| Increase, Cell Proliferation leads to Lung cancer | July 03, 2019 11:53 |
| nanoparticles | December 21, 2016 09:40 |
| welding fumes | May 25, 2022 07:02 |
| Carbon black | May 25, 2022 07:03 |
| Diesel engine exhaust | September 28, 2021 08:55 |
| nanomaterials | April 22, 2021 09:28 |
| Titanium dioxide nanoparticles | August 29, 2022 08:22 |
Abstract
This AOP was developed and first presented in Nymark et al. 2021.
The particulate fraction of diesel exhaust is known to be required for carcinogenesis, since filtered exhaust does not cause lung cancer in rodents (Brightwell et al., 1989). Inhaled, nanosized particles deposit primarily in the alveolar region, where clearance is low, and lead to prolonged particle retention enabling particle-bio interaction (Oberdörster et al., 2005; Gaté et al., 2017). Interaction between particles and lung resident cell membrane components (Figure 1A below in the Background section, MIE) leads to inflammation (KE1A) which is proportional to the total deposited surface area (Schmid and Stoeger, 2016; Danielsen et al., 2020; Kokot et al., 2020). The persistence of particles results in long-lasting inflammation (Hougaard et al., 2010; Chézeau et al., 2018). Metabolic activity of pro-inflammatory cells induces formation of ROS, which may also be augmented by the surface reactivity of particles themselves (Jacobsen et al., 2008b; Bendtsen et al., 2020). The sustained inflammatory signaling and concomitant synthesis of reactive radicals, cause a chronic state of oxidant-antioxidant imbalance and loss of protective mechanisms, potentially resulting in secondary genotoxicity (KE1B) (Evans et al., 2019a). Diesel exhaust consists of nanosized particles of inorganic and organic carbon with associated metal oxides and polyaromatic hydrocarbons (PAHs) (Taxell and Santonen, 2016; Bendtsen et al., 2020). Both the carbon core and solvent-extractable fractions containing PAHs are mutagenic in vivo and several metal (oxides) have been classified as (possibly) carcinogenic (IARC, 2006, 2012b; Hashimoto et al., 2007). It is possible that such genotoxic agents leach from the pulmonary deposited particles leading to activation of alternative AOPs associated with the formation of bulky DNA adducts and resulting in accumulation of mutations (Li and Nel, 2006) as indicated by the alternative path in Figure 1A below in the Background section (in gray) (Sasaki et al., 2020). In addition, the insoluble carbon core generates particle-induced ROS leading to oxidative stress (KE1C) (Bendtsen et al., 2020; Gren et al., 2020). In a recent study of five diesel exhaust particles designed to differ in chemical composition, DNA strand breaks (KE2) in bronchoalveolar lavage cells were found to correlate with the ROS forming capacity of the particles (Bendtsen et al., 2020). Similarly, carbon black generates surface-dependent ROS, causing oxidative DNA damage (KE2) and mutagenicity (KE3) in vivo and in vitro (Jacobsen et al., 2008b). Finally, direct interactions between nanosized particles and DNA or the mitotic spindle are also possible, and the AOP features a direct link between the MIE and mutagenicity, i.e., KE3 (Buliaková et al., 2017; Patel et al., 2017).
AOP Development Strategy
Context
Lung cancer, one of the most common and deadly forms of cancer, is in some cases associated with exposure to certain types of particles. With the rise of nanotechnology, there is concern that some engineered nanoparticles may be among such particles. In the absence of epidemiological evidence, assessment of nanoparticle carcinogenicity is currently performed on a time-consuming case-by-case basis, relying mainly on animal experiments. Non-animal alternatives exist, including a few validated cell-based methods accepted for regulatory risk assessment of nanoparticles. Furthermore, new approach methodologies (NAMs), focused on carcinogenic mechanisms and capable of handling the increasing numbers of nanoparticles, have been developed. However, such alternative methods are mainly applied as weight-of-evidence linked to generally required animal data, since challenges remain regarding interpretation of the results. These challenges may be more easily overcome by the novel Adverse Outcome Pathway (AOP) framework, which provides a basis for validation and uptake of alternative mechanism-focused methods in risk assessment. Here, we propose an AOP for lung cancer induced by nanosized foreign matter, anchored to a selection of 18 standardized methods and NAMs for in silico- and in vitro-based integrated assessment of lung carcinogenicity (Figure 1). The AOP provides a basis for development of AOP-aligned alternative methods-based integrated testing strategies for assessment of nanoparticle-induced lung cancer.
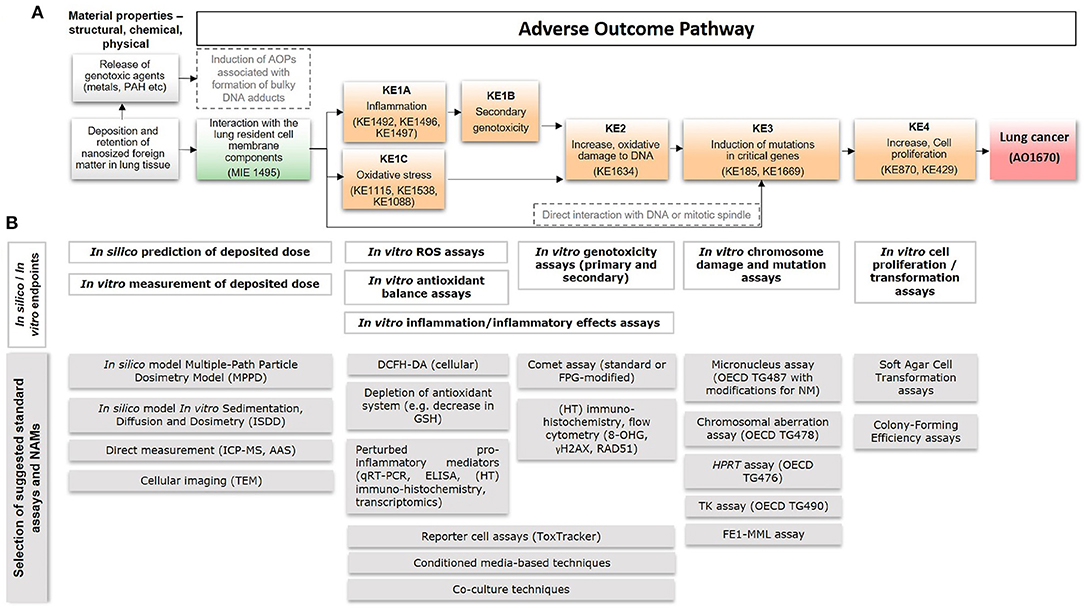
Figure 1. A putative AOP for pulmonary deposition and retention of nanosized foreign matter leading to lung cancer, including anchored in silico and in vitro methods. (A) A putative AOP developed based on information and knowledge about the process-generated and engineered nanoparticles diesel exhaust, carbon black, and TiO2. Suggested relevant existing KEs in the AOP-Wiki, that could serve for informing development of the proposed AOP, are mentioned within parentheses. (B) The AOP supports integrated application of in silico- and in vitro-based standard OECD tests with new approach methodologies (NAMs), including models/approaches for prediction of deposited dose, detection of ROS generation, inflammation, DNA damage, mutations, and cell transformation. Examples of specific assays are provided at the bottom. MIE, molecular initiating event; KE, key event; AO, adverse outcome; AOP, adverse outcome pathway; IC-PMS, inductively coupled plasma mass spectrometry; AAS, atomic absorption spectroscopy; TEM, transmission electron microscopy; ROS, reactive oxygen species; DCFH-DA, 2'-7'dichlorofluorescin diacetate; GSH, glutathione; ELISA, enzyme-linked immunosorbent assay; HT, high-throughput; FPG, formamidopyrimidine DNA glycosylase; OECD, Organization for Economic Co-operation and Development; HPRT, hypoxanthine phosphorybosyl transferase; TK, thymidine kinase; FE1-MML, FE1-MutaMouse lung epithelial cells.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1495 | Substance interaction with the lung resident cell membrane components | Interaction with the lung cell membrane |
| KE | 1496 | Increased, secretion of proinflammatory mediators | Increased proinflammatory mediators |
| KE | 1497 | Increased, recruitment of inflammatory cells | Recruitment of inflammatory cells |
| KE | 780 | Increase, Cytotoxicity (epithelial cells) | Increase, Cytotoxicity (epithelial cells) |
| KE | 1115 | Increase, Reactive oxygen species | Increase, ROS |
| KE | 2006 | Secondary genotoxicity | Secondary genotoxicity |
| KE | 1669 | Increased, DNA damage and mutation | Increased, DNA damage and mutation |
| KE | 870 | Increase, Cell Proliferation | Increase, Cell Proliferation |
| AO | 1670 | Lung cancer | Lung cancer |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Interaction with the lung cell membrane leads to Increased proinflammatory mediators | adjacent | Moderate | Moderate |
| Increased proinflammatory mediators leads to Recruitment of inflammatory cells | adjacent | Moderate | Low |
| Recruitment of inflammatory cells leads to Increase, Cytotoxicity (epithelial cells) | adjacent | Moderate | Moderate |
| Increase, Cytotoxicity (epithelial cells) leads to Increase, ROS | adjacent | ||
| Increase, ROS leads to Secondary genotoxicity | adjacent | ||
| Secondary genotoxicity leads to Increased, DNA damage and mutation | adjacent | ||
| Increased, DNA damage and mutation leads to Increase, Cell Proliferation | adjacent | ||
| Increase, Cell Proliferation leads to Lung cancer | adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| human | Homo sapiens | High | NCBI |
Sex Applicability
Overall Assessment of the AOP
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Nymark P, Karlsson HL, Halappanavar S, Vogel U. Adverse Outcome Pathway Development for Assessment of Lung Carcinogenicity by Nanoparticles. Front Toxicol. 2021 Apr 29;3:653386. https://doi.org/10.3389/ftox.2021.653386. eCollection 2021.
Bendtsen, K. M., Gren, L., Malmborg, V. B., Shukla, P. C., Tunér, M., Essig, Y. J., et al. (2020). Particle characterization and toxicity in C57BL/6 mice following instillation of five different diesel exhaust particles designed to differ in physicochemical properties. Part Fibre Toxicol. 17:38. doi: 10.1186/s12989-020-00369-9
Brightwell, J., Fouiliet, X., Cassano-Zoppi, A.-L., Bernstein, D., Crawley, F., Duchosal, F., et al. (1989). Tumours of the respiratory tract in rats and hamsters following chronic inhalation of engine exhaust emissions. J. Appl. Toxicol. 9, 23–31. doi: 10.1002/jat.2550090106
Buliaková, B., Mesárošová, M., Bábelová, A., Šelc, M., Némethová, V., Šebová, L., et al. (2017). Surface-modified magnetite nanoparticles act as aneugen-like spindle poison. Nanomedicine 13, 69–80. doi: 10.1016/j.nano.2016.08.027
Chézeau, L., Sébillaud, S., Safar, R., Seidel, C., Dembélé, D., Lorcin, M., et al. (2018). Short- and long-term gene expression profiles induced by inhaled TiO2 nanostructured aerosol in rat lung. Toxicol. Appl. Pharmacol. 356, 54–64. doi: 10.1016/j.taap.2018.07.013
Danielsen, P. H., Knudsen, K. B., Štrancar, J., Umek, P., Koklič, T., Garvas, M., et al. (2020). Effects of physicochemical properties of TiO2 nanomaterials for pulmonary inflammation, acute phase response and alveolar proteinosis in intratracheally exposed mice. Toxicol. Appl. Pharmacol. 386:114830. doi: 10.1016/j.taap.2019.114830
Evans, S. J., Clift, M. J. D., Singh, N., Wills, J. W., Hondow, N., Wilkinson, T. S., et al. (2019a). In vitro detection of in vitro secondary mechanisms of genotoxicity induced by engineered nanomaterials. Part. Fibre Toxicol. 16:8. doi: 10.1186/s12989-019-0291-7
Gaté, L., Disdier, C., Cosnier, F., Gagnaire, F., Devoy, J., Saba, W., et al. (2017). Biopersistence and translocation to extrapulmonary organs of titanium dioxide nanoparticles after subacute inhalation exposure to aerosol in adult and elderly rats. Toxicol. Lett. 265, 61–69. doi: 10.1016/j.toxlet.2016.11.009
Gren, L., Malmborg, V. B., Jacobsen, N. R., Shukla, P. C., Bendtsen, K. M., Eriksson, A. C., et al. (2020). Effect of renewable fuels and intake O2 concentration on diesel engine emission characteristics and Reactive Oxygen Species (ROS) formation. Atmosphere 11:641. https://www.mdpi.com/2073-4433/11/6/641/htm 10.3390/atmos11060641
Hashimoto, A. H., Amanuma, K., Hiyoshi, K., Sugawara, Y., Goto, S., Yanagisawa, R., et al. (2007). Mutations in the lungs of gpt delta transgenic mice following inhalation of diesel exhaust. Environ. Mol. Mutagen 48, 682–693. doi: 10.1002/em.20335
Hougaard, K. S., Jackson, P., Jensen, K. A., Sloth, J. J., Löschner, K., Larsen, E. H., et al. (2010). Effects of prenatal exposure to surface-coated nanosized titanium dioxide (UV-Titan). A study in mice. Part Fibre Toxicol. 7:16. doi: 10.1186/1743-8977-7-16
IARC (2006). Cobalt in Hard Metals and Cobalt Sulfate, Gallium Arsenide, Indium Phosphideand Vanadium Pentoxide. Monograph 86. Available online at: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono86.pdf
IARC (2010). Carbon Black, Titanium Dioxide, and Talc.Monograph 93. Available online at: https://publications.iarc.fr/111
IARC (2012a). Diesel and Gasoline Engine Exhausts and Some Nitroarenes. Monograph 105. Available online at: https://publications.iarc.fr/129
IARC (2012b). Review of Human Carcinogens (Package of 6 Volumes: A,B,C,D,E,F). Monograph 100. Available online at:: https://monographs.iarc.fr/wp-content/ uploads/2018/06/mono100C-10.pdf
IARC (2014). Some Nanomaterials and Some Fibres. Monograph 111. Available online at:: https://publications.iarc.fr/552
Jacobsen, N. R., Pojana, G., White, P., Møller, P., Cohn, C. A., Smith Korsholm, K., et al. (2008b). Genotoxicity, cytotoxicity, and reactive oxygen species induced by single-walled carbon nanotubes and C60 fullerenes in the FE1-Muta™Mouse lung epithelial cells. Environ. Mol. Mutagen 49, 476–487. doi: 10.1002/em.20406
Kokot, H., Kokot, B., Sebastijanović, A., Voss, C., Podlipec, R., Zawilska, P., et al. (2020). Prediction of chronic inflammation for inhaled particles: the impact of material cycling and quarantining in the lung epithelium. Adv. Mater. 32:2003913. doi: 10.1002/adma.202003913
Li, N., and Nel, A. E. (2006). The cellular impacts of diesel exhaust particles: beyond inflammation and death. Eur. Respir. J. 27, 667–668. doi: 10.1183/09031936.06.00025006
Oberdörster, G., Oberdörster, E., and Oberdörster, J. (2005). Nanotoxicology: an emerging discipline evolving from studies of ultrafine particles. Environ. Health Perspect. 113, 823–839. doi: 10.1289/ehp.7339
Patel, S., Patel, P., and Bakshi, S. R. (2017). Titanium dioxide nanoparticles: an in vitro study of DNA binding, chromosome aberration assay, and comet assay. Cytotechnology 69, 245–263. doi: 10.1007/s10616-016-0054-3
Sasaki, J. C., Allemang, A., Bryce, S. M., Custer, L., Dearfield, K. L., Dietz, Y., et al. (2020). Application of the adverse outcome pathway framework to genotoxic modes of action. Environ. Mol. Mutagen. 61, 114–134. doi: 10.1002/em.22339
Schmid, O., and Stoeger, T. (2016). Surface area is the biologically most effective dose metric for acute nanoparticle toxicity in the lung. J. Aerosol. Sci. 99, 133–143. doi: 10.1016/j.jaerosci.2015.12.006
Taxell, P., and Santonen, T. (2016). Diesel Engine Exhaust. Occupational and Environmental Medicine. The Nordic Expert Group for Criteria Documentation of Health Risks from Chemicals and the Dutch Expert Committee on Occupational Safety. Available online at: https://gupea.ub.gu.se/bitstream/2077/44340/1/gupea_2077_44340_1.pdf