
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 396
Title
Deposition of ionizing energy leads to population decline via impaired meiosis
Short name
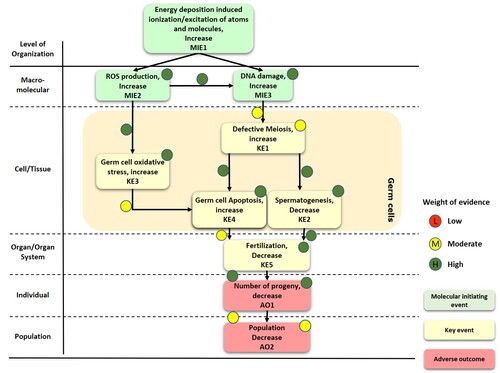
Graphical Representation
Point of Contact
Contributors
- Erica Maremonti
- Dag Anders Brede
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on February 25, 2025 04:18
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Deposition of Ionizing Energy | December 13, 2021 08:03 |
| Increase, Reactive oxygen species | June 12, 2025 01:27 |
| Increase, DNA damage | May 08, 2019 12:28 |
| Altered, Meiotic chromosome dynamics | May 27, 2019 14:17 |
| Increase, Oocyte apoptosis | April 30, 2020 16:41 |
| Decreased spermatogenesis | February 09, 2021 08:36 |
| decreased, Fertility | December 17, 2024 15:46 |
| Decrease of egg production and cummulative fecundity | October 03, 2019 11:13 |
| Decreased, Population size | December 03, 2016 16:37 |
| Decrease, Reproduction | April 11, 2021 17:38 |
| Increase, Cell cycle disruption | May 19, 2026 02:04 |
| Decrease, Oogenesis | April 30, 2020 16:41 |
| Increase, Oxidative Stress | February 11, 2026 07:05 |
| Ionizing Energy leads to Increase, Oxidative Stress | May 31, 2024 17:10 |
| Ionizing Energy leads to Increase, Oocyte apoptosis | May 14, 2021 04:49 |
| Increase, Oocyte apoptosis leads to Decrease of egg production and cummulative fecundity | May 18, 2021 07:30 |
| Ionizing Energy leads to Increase, DNA Damage | May 12, 2021 08:40 |
| Ionizing Energy leads to Altered, Meiotic chromosome dynamics | May 14, 2021 04:46 |
| Ionizing Energy leads to Decreased spermatogenesis | May 14, 2021 04:46 |
| Increase, DNA Damage leads to Altered, Meiotic chromosome dynamics | May 12, 2021 08:41 |
| Ionizing Energy leads to decreased, Fertility | May 14, 2021 04:47 |
| Ionizing Energy leads to Decrease of egg production and cummulative fecundity | May 14, 2021 04:48 |
| Increase, DNA Damage leads to Increase, Oocyte apoptosis | May 12, 2021 08:42 |
| Ionizing Energy leads to Decreased, Population size | May 14, 2021 04:48 |
| Altered, Meiotic chromosome dynamics leads to Increase, Oocyte apoptosis | May 12, 2021 08:42 |
| Altered, Meiotic chromosome dynamics leads to Decreased spermatogenesis | May 12, 2021 08:43 |
| Increase, Oxidative Stress leads to Increase, Oocyte apoptosis | May 31, 2024 17:09 |
| Increase, DNA Damage leads to Cell cycle disruption | October 25, 2022 03:56 |
| Cell cycle disruption leads to Altered, Meiotic chromosome dynamics | October 25, 2022 03:56 |
| Increase, Oocyte apoptosis leads to Decrease, Oogenesis | April 30, 2020 16:44 |
| Increase, Oocyte apoptosis leads to decreased, Fertility | May 12, 2021 08:46 |
| Decreased spermatogenesis leads to decreased, Fertility | July 13, 2020 04:42 |
| Decreased spermatogenesis leads to Decrease of egg production and cummulative fecundity | May 12, 2021 08:47 |
| decreased, Fertility leads to Decrease of egg production and cummulative fecundity | May 12, 2021 08:47 |
| Decrease of egg production and cummulative fecundity leads to Decreased, Population size | May 12, 2021 08:48 |
| Ionizing Energy leads to Increase, ROS | May 12, 2021 08:39 |
| Increase, ROS leads to Increase, Oxidative Stress | August 02, 2024 15:40 |
| Gamma radiation | April 15, 2017 16:04 |
| Ionizing Radiation | May 07, 2019 12:12 |
Abstract
Despite the tolerance demonstrated under exposure to high acute doses (> 1 kGy) of ionizing radiation in the nematode Caenorhabditis elegans, adverse outcome at the reproductive level have been observed under exposure of early stages of larval development to low-medium chronic doses (≥ 2.8 Gy). L1-L4 larval stages were shown to be the most radiosensitive stages of development due to adverse effects on gamete production. Specifically, significant sperm reduction and dysregulation of genes related to sperm meiosis and maturation were identified as the main key events (KE1, KE2) causing reduced number of progeny (AO1). Adverse effects of ionizing radiation on proliferative cells were also shown by enhanced germ cell apoptosis (KE3, KE4) in F0 nematodes and significant DNA damage in embryonic cells (F1) of irradiated nematodes, which was corroborated by the dysregulation of genes related to cell-cycle checkpoints, DNA repair, embryonic and post-embryonic development. Increased ROS levels (MIE2) and AODs activation were measured in vivo and by gene expression analysis after chronic irradiation of F0 nematodes. This was not accompanied by any adverse effect on somatic cell viability or any visible phenotypical effect, indicating tolerance of somatic tissue compared to the observed adverse effects shown on the germ cells. The observed redox imbalance suggested a significant contribution of indirect effects, including oxidative damage to DNA (MIE3), and represented the molecular initiating event derived from ionization and excitation of atoms and molecules (MIE1) after chronic irradiation.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1550 | Deposition of Ionizing Energy | Ionizing Energy |
| KE | 1392 | Increase, Oxidative Stress | Increase, Oxidative Stress |
| KE | 752 | Altered, Meiotic chromosome dynamics | Altered, Meiotic chromosome dynamics |
| KE | 1775 | Increase, Oocyte apoptosis | Increase, Oocyte apoptosis |
| KE | 1115 | Increase, Reactive oxygen species | Increase, ROS |
| KE | 1798 | Decreased spermatogenesis | Decreased spermatogenesis |
| KE | 1194 | Increase, DNA damage | Increase, DNA Damage |
| KE | 406 | decreased, Fertility | decreased, Fertility |
| KE | 1505 | Increase, Cell cycle disruption | Cell cycle disruption |
| KE | 1366 | Decrease, Oogenesis | Decrease, Oogenesis |
| AO | 1696 | Decrease of egg production and cummulative fecundity | Decrease of egg production and cummulative fecundity |
| AO | 1863 | Decrease, Reproduction | Decrease, Reproduction |
| AO | 997 | Decreased, Population size | Decreased, Population size |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Ionizing Energy leads to Increase, Oxidative Stress | non-adjacent | High | High |
| Ionizing Energy leads to Increase, Oocyte apoptosis | non-adjacent | Moderate | Moderate |
| Increase, Oocyte apoptosis leads to Decrease of egg production and cummulative fecundity | non-adjacent | Moderate | Moderate |
| Ionizing Energy leads to Altered, Meiotic chromosome dynamics | non-adjacent | Moderate | Moderate |
| Ionizing Energy leads to Decreased spermatogenesis | non-adjacent | Moderate | Moderate |
| Ionizing Energy leads to decreased, Fertility | non-adjacent | Moderate | Moderate |
| Ionizing Energy leads to Decrease of egg production and cummulative fecundity | non-adjacent | Moderate | Moderate |
| Ionizing Energy leads to Decreased, Population size | non-adjacent | Moderate | Moderate |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Larval development | High |
Taxonomic Applicability
| Term | Scientific Term | Evidence | Link |
|---|---|---|---|
| Caenorhabditis elegans | Caenorhabditis elegans | High | NCBI |
Sex Applicability
| Sex | Evidence |
|---|---|
| Hermaphrodite | High |
Overall Assessment of the AOP
Domain of Applicability
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Hartman, P. S., & Herman, R. K. (1982). Radiation-sensitive mutants of Caenorhabditis elegans. Genetics, 102(2), 159-178.
Hodgkin, J., & Barnes, T. M. (1991). More is not better: brood size and population growth in a self-fertilizing nematode. Proceedings of the Royal Society of London. Series B: Biological Sciences, 246(1315), 19-24.
Shakes, D. C., Wu, J. C., Sadler, P. L., LaPrade, K., Moore, L. L., Noritake, A., & Chu, D. S. (2009). Spermatogenesis-specific features of the meiotic program in Caenorhabditis elegans. PLoS Genet, 5(8), e1000611.
Reisz, J. A., Bansal, N., Qian, J., Zhao, W., & Furdui, C. M. (2014). Effects of ionizing radiation on biological molecules—mechanisms of damage and emerging methods of detection. Antioxidants & redox signaling, 21(2), 260-292.
Buisset-Goussen, A., Goussen, B., Della-Vedova, C., Galas, S., Adam-Guillermin, C., & Lecomte-Pradines, C. (2014). Effects of chronic gamma irradiation: a multigenerational study using Caenorhabditis elegans. Journal of environmental radioactivity, 137, 190-197.
Engert, C. G., Droste, R., van Oudenaarden, A., & Horvitz, H. R. (2018). A Caenorhabditis elegans protein with a PRDM9-like SET domain localizes to chromatin-associated foci and promotes spermatocyte gene expression, sperm production and fertility. PLoS genetics, 14(4), e1007295.
Maremonti, E., Eide, D. M., Oughton, D. H., Salbu, B., Grammes, F., Kassaye, Y. A., ... & Brede, D. A. (2019). Gamma radiation induces life stage-dependent reprotoxicity in Caenorhabditis elegans via impairment of spermatogenesis. Science of the Total Environment, 695, 133835.
Maremonti, E., Eide, D. M., Rossbach, L. M., Lind, O. C., Salbu, B., & Brede, D. A. (2020). In vivo assessment of reactive oxygen species production and oxidative stress effects induced by chronic exposure to gamma radiation in Caenorhabditis elegans. Free radical biology and medicine, 152, 583-596.
Guédon, R., Maremonti, E., Armant, O., Galas, S., Brede, D. A., & Lecomte-Pradines, C. (2021). A systems biology analysis of reproductive toxicity effects induced by multigenerational exposure to ionizing radiation in C. elegans. Ecotoxicology and Environmental Safety, 225, 112793.