
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 483
Title
Deposition of Energy Leading to Learning and Memory Impairment
Short name
Graphical Representation
Point of Contact
Contributors
- Vinita Chauhan
Coaches
- Shihori Tanabe
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.89 | WPHA/WNT Endorsed | Scientific Review | EMM - AOP report: Development of an adverse outcome pathway for deposition of energy leading to learning and memory impairment | OECD Series on AOPs: Deposition of Energy Leading to Learning and Memory Impairment |
This AOP was last modified on July 01, 2025 08:49
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Deposition of Energy | August 23, 2024 09:20 |
| Increase, Oxidative Stress | February 11, 2026 07:05 |
| Tissue resident cell activation | November 15, 2024 10:51 |
| Increase, Pro-Inflammatory Mediators | July 26, 2024 09:01 |
| Increase, Abnormal Neural Remodeling | July 26, 2024 09:32 |
| Increase, DNA strand breaks | December 17, 2024 11:57 |
| Impairment, Learning and memory | April 27, 2026 05:48 |
| Altered Stress Response Signaling | November 15, 2024 10:48 |
| Energy Deposition leads to Increase, Oxidative Stress | August 26, 2024 11:47 |
| Energy Deposition leads to Abnormal Neural Remodeling | July 26, 2024 14:20 |
| Energy Deposition leads to Tissue resident cell activation | July 26, 2024 08:24 |
| Energy Deposition leads to Impairment, Learning and memory | July 26, 2024 14:48 |
| Increase, Pro-Inflammatory Mediators leads to Impairment, Learning and memory | July 26, 2024 15:12 |
| Increase, Oxidative Stress leads to Altered Stress Response Signaling | August 26, 2024 13:05 |
| Increase, Oxidative Stress leads to Tissue resident cell activation | July 26, 2024 09:38 |
| Tissue resident cell activation leads to Increase, Pro-Inflammatory Mediators | July 26, 2024 10:11 |
| Increase, Pro-Inflammatory Mediators leads to Abnormal Neural Remodeling | September 10, 2024 15:13 |
| Abnormal Neural Remodeling leads to Impairment, Learning and memory | July 26, 2024 13:25 |
| Altered Stress Response Signaling leads to Abnormal Neural Remodeling | July 25, 2024 09:57 |
| Increase, DNA strand breaks leads to Abnormal Neural Remodeling | July 29, 2024 08:35 |
| Increase, Oxidative Stress leads to Increase, DNA strand breaks | July 29, 2024 11:21 |
| Energy Deposition leads to Increase, DNA strand breaks | July 29, 2024 09:42 |
| Increase, DNA strand breaks leads to Altered Stress Response Signaling | August 26, 2024 14:05 |
| Ionizing Radiation | May 07, 2019 12:12 |
Abstract
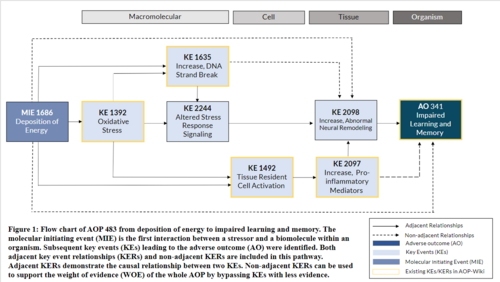
The central nervous system (CNS) is the main processing center of the body and is comprised of the brain and spinal cord. Some diseases of the CNS, including neurodegenerative diseases such as cognitive decline and dementia, are associated with advancing age. However, unwanted exposure to stressors may promote or accelerate these disorders. For example, ionizing radiation is an exposure which can disrupt cellular homeostasis in the brain. Herein, an adverse outcome pathway (AOP) is described from the molecular initiating event (MIE) of deposition of energy to the adverse outcome (AO) of learning and memory impairment. This AOP uses well-understood mechanistic events that encompass oxidative stress, DNA damage, tissue resident cell activation, altered stress response signaling, neuroinflammation, and their interactions, leading to eventual abnormal neural remodeling. The empirical evidence to support this AOP is primarily derived from studies that utilize ionizing radiation stressors relevant to space travel and radiotherapy treatments. Following deposition of energy (MIE, KE#1686), the adjacent key events are oxidative stress (KE#1392), tissue resident cell activation (KE#1492) and increased DNA strand breaks (KE#1635). Uncontrolled radical production within the cell has an adjacent connection with increased DNA strand breaks (KE#1635), altered stress response signaling (KE#2244) and tissue resident cell activation (KE#1492). Tissue resident cell activation has an adjacent connection to increased proinflammatory mediators (KE#1493). Prolonged neuroinflammation and altered stress response signaling have adjacent connections with abnormal neural remodeling (KE#2098) and subsequently learning and memory impairment (AO, KE#341). The AOP also includes multiple non-adjacent connections between key events. The overall weight of evidence for this AOP is moderate. Despite multiple knowledge gaps that are present, the evidence demonstrates a high-level of biological plausibility. The quantitative understanding is low as there is high uncertainty in the quantitative predictions between the KEs. This AOP has wide applicability and is particularly relevant to exposures from long-duration space flight and medical exposures using radiation therapy.
AOP Development Strategy
Context
Understanding the impact of ionizing radiation on CNS is essential as there are many possibilities for humans to be exposed to ionizing radiation including from medical procedures, accidental or wartime exposures, and occupational exposures, such as industrial radiographers or astronaut crewmembers. Various studies have reported cognitive deficits after high-doses of radiation exposure from radiotherapy, though there is a reported individual variability in human cohorts (Greene-Schloesser et al., 2012; Katsura et al., 2021; Turnquist et al., 2020). In preclinical animal models, studies suggest that even low-to-moderate doses of ionizing radiation from heavy ions can cause structural and functional impairments to the CNS including reductions in neurogenesis, changes in dendritic properties, activation of glial cells, leading to neural remodeling (Cekanaviciute et al., 2018; Kiffer et al., 2019b). However, how key changes in structural and functional properties of the CNS from ionizing radiation exposure are related to changes in cognitive function have yet to be delineated. Furthermore, preclinical studies also suggest that ionizing radiation may impact two major cognitive processes: learning and memory. Learning is the ability to create new associative or non-associative relationships and memory is the ability to recall sensory, short-term or long-term information (Desai et al., 2022, Kiffer et al., 2019b). Both learning and memory involve multiple brain areas including the hippocampal region, as well as the amygdala, the prefrontal cortex and the basal ganglia (Cucinotta et al., 2014; Desai et al., 2022; NCRP Commentary, 2016). The link between learning and memory is complexly intertwined, as their processes share mechanisms such as changes in neuronal plasticity, alternations in neurotransmitter release and reuptake and alterations in gray and white matter structure (Toricelli et al., 2021). Thus far, direct pathways linking ionizing radiation to key cellular and molecular events leading to an AO of impaired learning and memory have not been established.
This AOP can serve as a foundational pathway for expansion to other cognitive disorders and CNS diseases from an MIE of deposition of energy. The strength of this AOP is in its rigorous and systematic collection and evaluation of evidence with moderate to high levels of qualitative evidence supporting the KERs.
Strategy
The development strategy for this AOP has been described by Kozbenko et al., 2022. In brief, a structured literature search was conducted that included screening and prioritization of the references. Initial searches involved study inclusion through key words relevant to the MIE and AO, followed by focused searches for each of the KEs and KERs. Studies at all levels of biological organization, regardless of the species, life stage, or sex, were considered. References were excluded using a Population, Exposure, Outcome, Endpont (PEOE) statement. Studies were included if they met definitions of a population (human, mouse, rat, etc.), exposure (i.e., radiation), and/or mention of one of the key events (KEs) or outcome (AO) of interest. Studies were excluded if they lacked full text, and/or were not a peer-reviewed manuscript (i.e., thesis/dissertations, presentations, posters or conference abstracts). Non-English studies were included provided the data could be identified within the abstract. Relevant studies were identified in the context of the modified Bradford Hill criteria, which contain biological plausibility, temporal-, dose-, incidence-concordance, and essentiality.
The research strategy for this AOP has been described by Kozbenko et al., 2022. In brief, all data went through a systematic screening process for its collection and inclusion into the AOP. This included structured literature searches as well as additional screenings and prioritization of the references. Initial searches included key words for the MIE and AO, followed by focused searches for each of the keywords involved in the KEs and KERs. Studies at all levels of biological organization, regardless of the species, life stage, or sex, were considered. References were further screened using a Population, Exposure, Endpoint, and Outcome (PEOE) statement. Studies were excluded if they did not contain definitions of a population (human, mouse, rat, etc.), exposure (i.e., ionizing radiation), and/or mention of one of the key events (KEs) or outcome (AO) of interest. Studies were also excluded if they lacked full text, and/or were not a peer-reviewed manuscript (i.e., thesis/dissertations, presentations, posters or conference abstracts). Non-English studies were included provided the data could be identified within the abstract. Relevant studies were identified in the context of the modified Bradford Hill criteria, which contain biological plausibility, temporal-, dose-, incidence-concordance, and essentiality.
Pre-screening was completed using SWIFT Review (http://www.sciome.com/swift-review/version 1.43). In SWIFT, software generated tags were created based on study abstracts that helped group references and create lists to aid in prioritizing relevant studies. Reviewers could include or exclude references based on the tags and abstracts. DistillerSR (Evidence Partners. www.evidencepartners.com/products/distillersr-systematic-reviewsoftware released 12.06.2020 version 2.34.0R) was then used in a three-level screening exercise: Title and Abstract (Level 1), Full-Text (Level 2), and Data Extraction (Level 3). Screeners used the PEOE statement to assess relevance for inclusion or exclusion. At the data extraction level, screeners extracted data supporting the Bradford Hill criteria, taxonomic applicability, and life-stage applicability. A final screening of all studies was done manually to ensure data was relevant to the KERs in the pathway in the context of the Bradford Hill criteria. Note that not all types of stressors and associated exposure parameters support each KER. Since KERs are independent units from the rest of the AOP and can support multiple AOs, some macromolecular KERs may include studies from cell types (e.g., lens cells) and stressors (e.g., UV) not directly relevant to the AO.
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1686 | Deposition of Energy | Energy Deposition |
| KE | 1392 | Increase, Oxidative Stress | Increase, Oxidative Stress |
| KE | 2244 | Altered Stress Response Signaling | Altered Stress Response Signaling |
| KE | 1492 | Tissue resident cell activation | Tissue resident cell activation |
| KE | 2097 | Increase, Pro-Inflammatory Mediators | Increase, Pro-Inflammatory Mediators |
| KE | 2098 | Increase, Abnormal Neural Remodeling | Abnormal Neural Remodeling |
| KE | 1635 | Increase, DNA strand breaks | Increase, DNA strand breaks |
| AO | 341 | Impairment, Learning and memory | Impairment, Learning and memory |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Energy Deposition leads to Abnormal Neural Remodeling | non-adjacent | Moderate | Low |
| Energy Deposition leads to Impairment, Learning and memory | non-adjacent | Moderate | Low |
| Increase, Pro-Inflammatory Mediators leads to Impairment, Learning and memory | non-adjacent | Moderate | Low |
Network View
Prototypical Stressors
| Name |
|---|
| Ionizing Radiation |
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Unspecific | High |
| Male | Moderate |
| Female | Low |
Overall Assessment of the AOP
This AOP was derived from data that investigates the CNS of humans, animals and cellular models following predominantly exposure to ionizing radiation. The AOP is qualitative in nature and not intended to be specific to any particular exposure parameter. The exposure parameters informing the AOP include doses of moderate-high (>1 Gy) and both high and low-LET radiation qualities. However, the extent to which cognitive deficits exist at low-to-moderate ionizing radiation doses (0.1 Gy - 1 Gy) across all the KEs in the AOP remains incompletely understood as limited empirical evidence was retrieved to support this understanding. Since KERs are independent units from the rest of the AOP and can support multiple AOs, some macromolecular KERs may include studies from cell types (e.g., lens cells) and stressors (e.g., UV) not relevant to the AO.
The goal of this AOP is to identify the qualitative biological perturbations of the MIE of deposition of energy through the molecular, cellular and tissue-level KEs that lead to abnormal neural remodeling and the AO of impaired learning and memory. While neural remodeling is a natural process that allows the brain to continue to adapt, long-term exposure to stressors such as the space environment (e.g., microgravity and space radiation) may lead to chronic inflammation and possible changes in structure and function of neural cells ultimately resulting in cognitive deficits. The progression of KEs along the proposed hypothetical AOP is driven by persistent oxidative stress and chronic release of pro-inflammatory markers, creating an environment of neuroinflammation. The KEs chosen for this AOP had adequate empirical evidence, however, other KEs may be added later to incorporate new mechanisms and AOs into its broader network. Since the AOP is stressor agnostic, this pathway is applicable to impaired learning and memory in the context of multiple stressors of deposition of energy including ionizing radiation exposure from space travel and radiotherapy.
Biological Plausibility
The overall biological plausibility in this AOP is high. The KERs in the AOP have either moderate or high evidence for the mechanistic understanding of the relationships between the upstream and downstream KEs. The KEs are well-studied, and an understanding of the structural and functional linkages are well-established.
This AOP is initiated with deposition of energy. Deposition of energy can damage DNA via direct mechanisms, by which the electrons ionize DNA molecules themselves, or via indirect mechanisms, by which the ionization of water produces hydroxyl radicals that can damage DNA bases causing DNA strand breaks (Nikjoo et al., 2016; Wilkinson et al., 2023) or directly upregulating enzymes involved in reactive oxygen and nitrogen species (RONS) production (i.e., catalase) (de Jager, Cockrell and Du Plessis, 2017). Both reactive oxygen species (ROS) as well as reactive nitrogen species (RNS) (Ahmadi et al., 2022; Karimi et al., 2017; Slezak et al., 2015; Tahimic & Globus, 2017; Wang et al., 2019a) may be produced after deposition of energy. If RONS cannot be eliminated quickly and efficiently by the cell’s defense system, oxidative stress ensues (Balasubramanian, 2000; Ganea & Harding, 2006; Karimi et al., 2017). Within the brain, oxidative stress can lead to the activation of microglial cells (Fishman et al., 2009; Schnegg et al., 2012; Zhang et al., 2017) and astrocytes (Daverey & Agrawal, 2016; Wang et al., 2017). These cells then release pro-inflammatory mediators and initiate antioxidant defenses (Lee, Cha & Lee, 2021; Simpson & Oliver, 2020). However, if the antioxidant capacity is overwhelmed, chronic inflammation may result.
Oxidative stress can also lead to altered stress response signaling. Directly, ROS causes oxidation of amino acid residues resulting in conformational changes, protein expansion, and protein degradation. This can cause changes in the activity and level of signaling proteins (Ping et al., 2020; Li et al., 2013). Oxidation of key functional amino acids can also alter the activity of signaling proteins, resulting in downstream alterations in signaling pathways (Ping et al., 2020; Schmidt-Ullrich et al., 2000; Valerie et al., 2007; Lehtinen & Bonni, 2006; Ramalingam & Kim, 2012). DNA strand breaks from oxidative damage can activate DNA damage response signaling and modify the expression of other signaling proteins (Ping et al., 2020; Nagane et al., 2021; Schmidt-Ullrich et al., 2000; Valerie et al., 2007).
Both increased pro-inflammatory mediators and altered stress response signaling can lead to abnormal neural remodeling. Various pro-inflammatory cytokines can affect neural activity/function, the most common being IL-1β, TNF-α, IL-6 and IFN-γ. During an inflammatory response, these cytokines act on different receptors to initiate several signaling pathways to induce neuronal degeneration, apoptosis or to propagate further pro-inflammatory responses (Mousa & Bakhiet, 2013; Prieto & Cotman, 2018). These signaling pathways include, but are not limited to PI3K/Akt pathways, MAPK pathways, senescence signaling, and apoptosis pathways. The PI3K/Akt and MAPK pathways are involved in many processes in neurons, including cell survival, morphology, proliferation, differentiation, and synaptic activity (Davis and Laroche, 2006; Falcicchia et al., 2020; Long et al., 2021; Mazzucchelli and Brambilla, 2000; Mielke and Herdegen, 2000; Nebreda and Porras, 2000; Rai et al., 2019; Rodgers and Theibert, 2002; Sherrin, Blank, and Todorovic, 2011). The apoptosis pathway decreases cell number, while senescence signaling can influence the regenerative potential of the cell and therefore, neurogenesis (Betlazar et al., 2016; McHugh and Gil, 2018; Mielke and Herdegen, 2000). Disruptions to components of these pathways will lead to abnormal neural remodeling, which includes alterations in both morphological properties and functional properties of the neurons (Betlazar et al., 2016; Davis and Laroche, 2006; Mazzucchelli and Brambilla, 2000; Nebreda and Porras, 2000). However, the biological changes that follow perturbation of these pathways is not understood in every context and cell type, making the biological plausibility for this relationship moderate (Nebreda and Porras, 2000). Decreased morphological properties of neurons, including reductions in dendritic complexities and spine densities, as well as altered functional properties of neurons including altered synaptic signaling and neurogenesis, has been associated with learning and memory impairment (Bálentová & Adamkov, 2020; Hladik & Tapio, 2016; Monje & Palmer, 2003; Romanella et al., 2020; Tomé et al., 2015).
Empirical Support (Temporal, Dose, and Incidence Concordance)
This AOP demonstrates moderate empirical evidence to support the modified Bradford Hill criteria. Overall, many studies demonstrated that upstream KEs occurred at lower or the same doses and at earlier or the same times as downstream KEs. There were some inconsistencies where the KEs were only measured at one dose or time. The evidence collected was gathered from various studies using in vitro and in vivo rat, mice, rabbit, squirrel, bovine and human models. Various stressors were applied, including UV, UVB, UVA, gamma ray, X-ray, protons, alpha particle, neutron, and heavy ion irradiation.
Regarding time concordance, deposition of energy occurs immediately following irradiation, and downstream events will always occur at a later time-point. DNA damage occurs within nanoseconds of deposition of energy with DNA strand breaks measured from seconds to minutes later and altered signaling measured minutes to days later (Acharya et al., 2010; Antonelli et al., 2015; Mosconi et al., 2011; Rogakou et al., 1999; Rothkamm and Lo, 2003; Sabirzhanov et al., 2020; Zhang et al., 2017). Rapid increases in ROS (Limoli et al., 2004; Giedzinski et al., 2005; Suman et al., 2013) and activation of microglia and astrocytes have been observed within hours of irradiation and can persist for 12 months (Kyrkanides et al., 1999; Hwang et al., 2006; Suman et al., 2013). For tissue resident cell activation and increase in pro-inflammatory mediators, studies generally show that these events occur at a similar time frame (Parihar et al., 2018; Liu et al., 2010; Dong et al., 2015; Lee et al., 2010; Zhou et al., 2017). The alteration of signaling pathways is a molecular-level KE like oxidative stress, and both can occur concurrently (Xu et al., 2019), although increased ROS levels can be initiated significantly before altered stress response signaling (Suman et al., 2013). Abnormal neural remodeling has been observed at various time points from hours to months after exposure to a stressor, and its upstream KEs (altered signaling and increased pro-inflammatory mediators) generally appear earlier (Kanzawa et al., 2006; Limoli et al., 2004; Pius-Sadowska et al., 2016) or at similar times, respectively (Zonis et al., 2015; Wong et al., 2004, Green et al., 2012; Ryan et al., 2013; Vallieres et al., 2002). In response to irradiation, impaired learning and memory is typically observed at similar time-points as markers of abnormal neural remodeling due to the timing of measurements (Raber et al., 2004; Parihar et al., 2016; Madsen et al., 2003; Winocur et al., 2006; Rola et al., 2004).
Regarding dose concordance, multiple studies also demonstrate that the upstream KEs occur at lower or the same doses as downstream KEs as energy is deposited immediately at any dose of radiation. Some studies report a linear-dose-dependent increases in DNA strand breaks for a large range of doses (Antonelli et al., 2015; Hamada et al, 2006; Rübe et al., 2008). In addition, neural precursor cells irradiated with protons at 1, 2, 5 and 10 Gy showed a dose-dependent increase in ROS levels (Giedzinski et al., 2005). In another study, activation of microglia and astrocytes were seen at doses as low as 5 cGy that persisted to 30 cGy (Parihar et al., 2018). However, dose concordance is not consistently observed across studies, which can be attributed to differences in experimental design. Some studies also only measured the KEs at one dose, which presented further inconsistencies.
Few studies showed incidence concordance where the upstream KE demonstrated a greater change than the downstream KE following a stressor. Not all KERs displayed an incident-concordant relationship, but for those that did, only a small proportion of the empirical evidence supported this relationship. For example, mice exposed to 2 Gy of gamma irradiation showed increases of pro- apoptotic markers p53 and BAX by 8.4- and 2.3-fold, respectively. A 0.6-fold decrease in Bcl-2 (anti-apoptotic marker) was also observed, and gamma rays cause a decrease in cortical thickness by 0.9-fold (Suman et al., 2013).
Uncertainties, Inconsistencies, and Data Gaps
There are a few inconsistencies in this AOP. Some studies show sex-specific changes in the KEs. For example, two studies reported that tissue resident cell activation was not affected in female mice after 0.3 and 0.5 Gy of radiation (Krukowski et al., 2018a; Parihar et al., 2020) while a separate study showed that only female mice had activated cells after 2 Gy (Raber et al., 2019). Another study reported a greater radiation-induced reduction in neurogenesis in male mice compared with female mice (Kalm et al., 2013). More research is necessary to identify if these results are sex-specific or due to other modulating factors.
There have been some inconsistencies reported in the KER Deposition of Energy (KE#1686) to Increase DNA Strand Breaks (KE#1635). For example, dose-rates and radiation quality may influence dose-response relationships (Brooks et al., 2016, Sutherland et al., 2000; Nikjoo et al., 2001; Jorge et al., 2012). More research is necessary to understand the impact low-doses of ionizing radiation exposure on DNA damage as some studies report low-dose exposures may invoke protection against spontaneous genomic damage (as reviewed by ICRP (2007) and UNSCEAR (2008)).
Anatomical location of change in the KEs may impact its response. For example, in response to ionizing radiation, changes occurred in hippocampal dendritic spines CA1 subregion of hippocampus but not in the dorsal dentate gyrus (Kiffer et al., 2019a).
Changes in KEs and the AO may be dose and stressor specific when assessed using animal models. For example, cue feared conditioning, a measure of learning and memory had different responses in mice at 0.2 Gy vs. 1 Gy of 28Si exposure (Whoolery et al., 2017). Also in mice, object memory was impaired after 0.1 or 0.25 Gy 16O exposure and social novelty learning was impaired after 0.25 Gy 16O exposure, but neither dose impaired short-term spatial memory (Kiffer et al., 2019a).
Changes in signaling pathways may provide inconsistent outcomes related to abnormal neural remodeling. For example, the p38 pathway is involved in many, often opposing, biological processes (Nebreda and Porras, 2000). Furthermore, the MAPK pathways can exhibit varied responses after exposure to oxidative stress (Azimzadeh et al., 2015).
Many studies do not report direct measures of oxidative stress. As free radicals are quickly scavenged, the quantitative understanding of this relationship can be inconsistent, due to varied response of antioxidant enzymes across experimental conditions and time measurements. This has led to some inconsistencies within the KERs. For example, in contrast to other studies demonstrating an increase in oxidative stress following deposition of energy, neutron radiation decreased malondialdehyde, a product of oxidative stress (Chen et al., 2021).
The scope of several KEs in this AOP is broad and this reflects a level of uncertainty in exact endpoints that specifically link to the AO, therefore several KEs (e.g. abnormal neural remodeling and signaling pathways) are defined by multiple structural and functional measurements.
Inflammatory markers exhibit a dual role, with the capacity for both anti-inflammatory and pro-inflammatory actions. Variables such as concentration, timing, and the specific microenvironment play pivotal roles in determining whether a mediator acts in a pro-inflammatory or anti-inflammatory manner (Lawrence & Gilroy, 2007; Nathan & Ding, 2010).
The use of different assays to assess the KEs may lead to variations in the quantitative understanding of observations across studies.
Limited data is available to support an understanding of oxidative stress and pro-inflammatory mediators at low doses < 0.1 Gy.
Finally, many of the KERs do not include studies in humans. More research could be needed to observe these relationships in human models.
There were multiple challenges present in the development of this AOP which identified numerous gaps in the data. The majority of the evidence for this AOP is extracted from preclinical animal and cellular models. Therefore, the low availability of human studies presents a challenge as translation of the animal and cellular models to humans is difficult due to differences in physiology, methods and measurements. In addition, although both age and sex are listed as modulating factors, there is more research necessary to elucidate the interaction between age and sex on the KEs, particularly how these factors may modulate the causal connectivity of the relationships and the AO. Direct comparisons between studies were also difficult due to differences in model, radiation quality, dose, dose rate and endpoint which led to some inconsistencies. Many studies reported limited dose ranges or time-points and often measured a single KE, limiting evidence for direct KERs. The current AOP has low quantitative evidence supporting the KERs, however, this AOP can be expanded with experiments that further exemplify the level of dose- and time- concordance across multiple endpoints. This will improve the quantitative understanding of the relationships which can then support the development of risk models and tools for mitigating risk.
Domain of Applicability
This AOP is relevant to vertebrates, such as humans, mice, rats. The taxonomic evidence supporting the AOP comes from the use of human (Homo sapiens), human-derived cell line, beagle dog (Canis lupus familiaris), rat (Rattus orvegicus), and mouse (Mus musculus) studies. Across all species, most available data was derived from adult and adolescent models with a moderate to high level of evidence compared to less available data from preadolescent models. Many of the KEs demonstrated moderate to high evidence for males and low evidence for females. In multiple KEs, sex was unspecified.
Essentiality of the Key Events
Overall, the KEs in this AOP demonstrate moderate essentiality. Essentiality is demonstrated when upstream KEs are blocked or inhibited eliciting a change in the downstream KE.
Essentiality of the Deposition of Energy (MIE, KE#1686)
-
Deposition of energy is difficult to test for essentiality as deposition of energy is a physical stressor and cannot be blocked/decreased using chemicals. Since deposited energy initiates events immediately, the removal of deposited energy, a physical stressor, also supports the essentiality of the key event. Studies that do not deposit energy are observed to have no downstream effects.
Essentiality of Oxidative Stress (KE#1392)
-
The effect of antioxidants on altered stress response signaling (KE#2244)
-
Antioxidants including Melandrii Herba extract, N-acetyl-L-cysteine (NAC), gallocatechin gallate/epigallocatechin-3-gallate, Cornus officinalis (CC) and fermented CC (FCC), L-165041, fucoxanthin, and edaravone were shown to decrease phosphorylation of MAPKs such as ERK1/2, JNK1/2 and p38 after exposure to radiation, H2O2 or lipopolysaccharide (LPS) (Lee et al., 2017; Deng et al., 2012; Park et al., 2021; Tian et al., 2020; Schnegg et al., 2012; Zhao et al., 2017; Zhao et al., 2013; El-Missiry et al., 2018).
-
The effects of antioxidants on tissue resident cell activation (KE#1492)
-
Antioxidants including Kukoamine A (KuA) and curcumin were found to reduce levels of microglia and astrocyte activation (Zhang et al. 2017; Daverey & Agrawal, 2016; Wang et al., 2017).
-
The effect of knocking out a ROS-producing enzyme
-
A knockout model of mitochondrial superoxide dismutase 2 (SOD2) resulted in an increase in reactivity of microglial cells (Fishman et. al 2009).
Essentiality of Increase, DNA Strand Breaks (KE#1635)
-
The effects of blocking DNA strand breaks on altered stress response signaling (KE#2244)
-
Treatment with mesenchymal stem cell-conditioned medium (MSC-CM) reduced γ-H2AX, decreased the levels of p53, Bax, cleaved caspase 3 and increased the levels of Bcl-2 in HT22 cells irradiated with 10 Gy of X-rays (Huang et al., 2021).
-
The inhibition of microRNA (miR)-711 decreased levels of DNA damage markers, p-ATM, p-ATR and γ-H2AX, and decreased signaling molecules including p-p53, p21 and cleaved caspase 3 (Sabirzhanov et al., 2020).
-
The effects of blocking DNA strand breaks on abnormal neural remodeling (KE#2098)
-
Treatment of HT22 hippocampal neuronal cells with minocycline inhibited the expression of γ-H2AX and the p-ATM/ATM ratio as well as reduced apoptosis following X-ray exposure (Zhang et al., 2017). Similarly, MSC-CM reduced the expression of γ- H2AX and reduced apoptosis, reversing the changes induced by X-ray radiation (Huang et al., 2021).
-
Lithium chloride was also shown to reduce γ-H2AX levels and increase proliferation in neural stem cells irradiated with 60Co gamma rays (Zanni et al., 2015).
Essentiality of Altered Stress Response Signaling (KE#2244)
-
The effects of modulating cell signaling on abnormal neural remodeling (KE#2098)
-
Knockout models of key molecules in the MAPK pathways and apoptotic pathway reduced apoptotic activity and restored neuron numbers induced by simulated ischemic stroke or radiation (Tian et al., 2020; Chow, Li and Wong, 2000; Limoli et al., 2004).
-
Inhibition of key signaling molecules involved in the MAPK pathways and the PI3K/Akt pathway restored neural stem cell numbers, neuronal differentiation, and neuronal structure induced by radiation (Eom et al., 2016; Kanzawa et al., 2006; Zhang et al. 2018)
Essentiality of Tissue Resident Cell Activation (KE#1492)
-
The effects of modulating cell activation on pro-inflammatory mediators (KE#1493)
-
Drugs including tamoxifen, retinoic acid, N-acetyl-L-cysteine (NAC), SP 600125 (SP), a specific c-jun kinase inhibitor, and NS- 398, a microglial activator attenuated the activation of tissue-resident cells and consequently reduced the levels of pro- inflammatory mediators (Liu et al., 2010; van Neerven et al., 2010; Komatsu et al., 2017; Ramanan, 2008; Kyrkanides et al., 2002).
Essentiality of Pro-Inflammatory Mediators (KE#1493)
-
The effects of modulating pro-inflammatory mediators on abnormal neural remodeling (KE#2098)
-
Treatments including MW-151, a selective inhibitor of pro-inflammatory cytokine production, KuA, and histamine restored neurogenic signaling, hippocampal apoptosis, and neuronal complexity (Jenrow et al., 2013; Zhang et al., 2017; Saraiva et al., 2019).
-
Multiple studies use cytokine receptor antagonists or knock-out key receptors to block the effects of IL-1β, TNF-α, and CCL2, which preserves neuron survival (Green et al., 2012; Ryan et al., 2013; Wu et al., 2012; Chen and Palmer, 2013).
-
Complement component 3 (C3) knockout models also caused increased synaptic number, reduced neuron loss and ameliorated synaptic morphology impairment (Shi et al., 2017).
-
The effects of modulating pro-inflammatory mediators on learning and memory impairment (AO, KE#341)
-
Anti-inflammatory drugs or hormones including MW-151, a selective inhibitor of pro-inflammatory cytokine production, lidocaine, an anesthetic with anti-inflammatory properties, ethyl-eicosapentaenoate (E-EPA) and 1-[(4-nitrophenyl)sulfonyl]-4- phenylpiperazine (NSPP), both of which are anti-inflammatory drugs and α-Melanocyte stimulating hormone (α-MSH), which antagonizes the effects of pro-inflammatory cytokines, have rescued the impairments seen in learning and memory (Bhat et al., 2020; Gonzalez et al., 2009; Jenrow et al., 2013; Taepavarapruk & Song, 2010; Tan et al., 2014).
Essentiality of Abnormal Neural Remodeling (KE#2098)
No identified studies describe essentiality of abnormal neural remodeling as it cannot be blocked / decreased using chemicals
Evidence Assessment
Biological Plausibility
|
1. Support for Biological Plausibility of KERs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
Extensive understanding of the KER based on extensive previous documentation and broad acceptance; Established mechanistic basis |
KER is plausible based on analogy to accepted biological relationships, but scientific understanding is not completely established |
There is empirical support for statistical association between KEs, but the structural or functional relationship between them is not understood |
|
|
Deposition of Energy (MIE, KE#1686) → Oxidative Stress (KE#1392) |
High There is high evidence surrounding the biological plausibility of deposition of energy leading to increased oxidative stress. When energy reaches a cell, it reacts with water and organic materials to produce ROS. Oxidative stress occurs when antioxidant systems cannot eliminate ROS. |
|||
|
Deposition of Energy (MIE, KE#1686) → Tissue Resident Cell Activation (KE#1492) |
High There is high evidence surrounding biological plausibility of deposition of energy leading to tissue resident cell activation. It is well understood that deposition of radiation energy leads to a recruitment of immune cells within the local tissue which can induce an immune and inflammatory response, characterized by the recruitment and activation of local macrophages in the brain. |
|||
|
Oxidative Stress (KE#1392) → Increase, DNA Strand Breaks (KE#1635) |
High There is high evidence surrounding biological plausibility of oxidative stress leading to DNA strand breaks. Oxidative stress can induce DNA damage by oxidizing or deleting DNA bases leading to strand breaks. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Altered Stress Response Signaling (KE#2244) |
High There is high evidence surrounding biological plausibility of increased DNA strand breaks to altered stress response signaling. DNA strand breaks induce DNA damage responses which result in the induction of various stress response signaling. |
|||
|
Oxidative Stress (KE#1392) → Tissue Resident Cell Activation (KE#1492) |
Moderate There is moderate evidence surrounding biological plausibility of increased oxidative stress leading to tissue resident cell activation. Increases in oxidative stress elicits activation of microglial cells and astrocytes in the brain. Activated microglia and astrocytes release pro-inflammatory mediators and promote antioxidant defenses. Feedforward and feedback loops of RONS and inflammatory pathways make the direct link between oxidative stress and microglial cell or astrocyte activation difficult to discern. |
|||
|
Oxidative Stress (KE#1392) → Altered Stress Response Signaling (KE#2244) |
High There is high evidence surrounding the biological plausibility of increased oxidative stress to altered stress response signaling. Oxidative stress can lead to altered stress response signaling both directly and indirectly. Directly, oxidative stress conditions can lead to oxidation of amino acid residues. This causes conformational changes, protein expansion, and protein degradation, leading to changes in the activity and level of signaling proteins that result in downstream alterations in signaling pathways. Indirectly, oxidative stress can damage DNA causing changes in the expression of signaling proteins as well as the activation of DNA damage response signaling. |
|||
|
Altered Stress Response Signaling (KE#2244) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate There is moderate evidence surrounding biological plausibility of altered stress response signaling to abnormal neural remodeling. Abnormal neural remodeling is controlled by signaling pathways in the brain, including PI3K/Akt pathway, MAPK pathways, senescence pathways, and apoptosis pathways. The PI3K/Akt and MAPK pathways are involved in many processes in neurons, including cell survival, morphology, proliferation, differentiation, and synaptic activity. The apoptosis pathway influences cell numbers, while the senescence pathway can influence neurogenesis. Disruptions to components of these pathways will lead to abnormal neural remodeling in a relationship that is structurally well-understood. However, the biological changes that follow perturbation of these pathways is not understood in every context and cell type. |
|||
|
Tissue Resident Cell Activation (KE#1492) → Increase, Pro-inflammatory Mediators (KE#2097) |
High There is high evidence surrounding biological plausibility of tissue resident activation to increase in pro-inflammatory mediators. In the brain, activated astrocytes and microglia undergo gliosis and proliferate, releasing pro-inflammatory mediators and production of cytokines. This response is normal after exposure to pathogens, but prolonged activation can prolong the inflammatory response. Cytokines and chemokines can also increase the permeability of the blood-brain barrier, further increasing pro-inflammatory mediator levels. |
|||
|
Increase, Pro-inflammatory Mediators (KE#2097) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate There is moderate evidence surrounding the biological plausibility of increased pro-inflammatory mediators to abnormal neural remodeling. There are various pro-inflammatory cytokines that can affect neuronal integrity an inflammatory response and these cytokines act on different receptors to initiate several signaling pathways to induce neuronal degeneration, apoptosis or to propagate pro-inflammatory responses. However, the exact mechanistic relationship remains to be elucidated due to the complexity of cytokine cascading events. |
|||
|
Increase, Abnormal Neural Remodeling (KE#2098) → Impairment, Learning and Memory (AO, KE#341) |
Moderate There is moderate evidence surrounding biological plausibility of abnormal neural remodeling leading to impaired learning and memory. Evidence of abnormal neural remodeling, such as reductions in spine density, reduced adult neurogenesis and impaired neuronal networks are associated with cognitive impairments, as evident from studies in multiple different species |
|||
|
Deposition of Energy (MIE, KE# 1686) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate There is moderate evidence surrounding biological plausibility of deposition of energy to abnormal neural remodeling. Irradiation induces oxidative stress and neuroinflammation, which alter neuronal integrity. Many reviews examine the radiation-induced neural damage and identify correlation with oxidative stress and neuroinflammatory mechanisms. |
|||
|
Deposition of Energy (MIE, KE#1686) → Impairment, Learning and Memory (AO, KE#341) |
High There is high evidence surrounding biological plausibility of deposition of energy to impaired learning and memory. Energy deposition in the form of ionizing radiation can result in behavioral changes and impairments in learning and memory. Under normal conditions, diminished cognitive functions is influenced by aging or can occur if there is a predisposition to neurodegenerative diseases such as Alzheimer’s, however, exposure to ionizing radiation may accelerate risk for age-related cognitive decline. |
|||
|
Deposition of Energy (MIE, KE#1686) → Increase, DNA Strand Breaks (KE#1635) |
High There is high evidence surrounding biological plausibility of deposition of energy to DNA strand breaks. Direct DNA damage can occur after deposition of energy by direct oxidation of the DNA. Indirect DNA damage from deposition of energy can also occur via generation of ROS that can subsequently oxidize and damage DNA. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate There is moderate evidence surrounding biological plausibility of increased DNA strand breaks to increase, abnormal neural remodeling. DNA strand breaks may initiate apoptotic signaling and impact synaptic activity, neural plasticity, differentiation, and proliferation. |
|||
|
Pro-inflammatory Mediators (KE#2097) → Impairment, Learning and Memory (AO, KE#341) |
Moderate There is moderate support for the biological plausibility of the key event relationship between pro-inflammatory mediators to impaired learning and memory. In a neuroinflammatory response, pro-inflammatory mediators including cytokines induce physiological and/or structural changes within the brain that can ultimately lead to impaired learning and memory. The exact mechanistic relationship is still unclear due to the complexity of cytokine cascading events. |
|||
Empirical Support
|
Review of the Empirical support for each KER |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Does KEupstream occur at lower doses and earlier time points than KEdownstream; is the incidence or frequency of KEupstream greater than that for KEdownstream for the same dose of tested stressor? |
There is a dependent change in both events following exposure to a wide range of specific stressors (extensive evidence for temporal, dose-response and incidence concordance) and no or few data gaps or conflicting data. |
There is demonstrated dependent change in both events following exposure to a small number of specific stressors and some evidence inconsistent with the expected pattern that can be explained by factors such as experimental design, technical considerations, differences among laboratories, etc |
There are limited or no studies reporting dependent change in both events following exposure to a specific stressor (i.e., endpoints never measured in the same study or not at all), and/or lacking evidence of temporal or dose-response concordance, or identification of significant inconsistencies in empirical support across taxa and species that don’t align with the expected pattern for the hypothesised AOP |
|
|
Deposition of Energy (MIE, KE#1686) → Oxidative Stress (KE#1392) |
High Ample evidence from in vitro and in vivo rat, mice, rabbit, squirrel, bovine and human models support time and dose response effects related to deposition of energy from various ionizing radiation sources leading to an increase in oxidative stress. |
|||
|
Deposition of Energy (MIE, KE#1686) → Tissue Resident Cell Activation (KE#1492) |
Moderate With increasing dose of ionizing radiation, there are increasing amounts of resident tissue activation in both astrocytes and microglial cells. Multiple studies show dose-response and time-response effects with both high and low dose studies, as well as time ranges from hours to months, though additional studies at low-doses would improve empirical support. |
|||
|
Oxidative Stress (KE#1392) → Increase, DNA Strand Breaks (KE#1635) |
Moderate Empirical evidence from in vivo and in vitro studies demonstrates increased DNA strand breaks from oxidative stress. Multiple studies show dose-response effects, though time response effects are difficult to monitor for both KEs. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Altered Stress Response Signaling (KE#2244) |
Moderate A few studies demonstrate dose-concordance, and multiple studies demonstrate time-concordance for this relationship. DNA strand breaks were observed prior to altered stress response signaling. |
|||
|
Oxidative Stress (KE#1392) → Tissue Resident Cell Activation (KE#1492) |
Moderate The literature demonstrates that an increase in the level of stressor related to oxidative stress results in an increase in cellular activation of microglial cells or astrocytes and this relationship is consistent between studies. However, dose and time concordance are unclear as there is limited data that describes oxidative stress occurring at lower doses or before tissue resident cell activation. |
|||
|
Oxidative Stress (KE#1392) → Altered Stress Response Signaling (KE#2244) |
Moderate Many studies demonstrate dose-concordance, and few demonstrate time-concordance for this relationship. Oxidative stress was often observed at lower, or the same doses as altered signaling and sometimes also at earlier times as altered signaling. However, only a few specific stressors are used in this KER and inconsistencies are present, likely due to different experimental designs. |
|||
|
Altered Stress Response Signaling (KE#2244) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate Many studies demonstrate dose-concordance in multiple signaling pathways. Studies have also shown that signaling pathways are altered before abnormal neural remodeling is observed. However, inconsistent changes in signaling pathways may be due to the context-dependence of signaling pathways as they can have different biological processes. |
|||
|
Tissue Resident Cell Activation (KE#1492) → Increase, Pro-inflammatory Mediators (KE#2097) |
Moderate Studies consistently observed changes in astrocyte and microglial activation at lower or the same dose as increased pro-inflammatory mediators and many studies also found changes in astrocyte and microglial activation earlier or at the same time as increased pro-inflammatory mediators. However, inconsistencies could be due to differences in experimental conditions. |
|||
|
Increase, Pro-inflammatory Mediators (KE#2097) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate There are multiple studies that show time-concordance, though studies on dose-concordance are lacking. Studies suggest that pro-inflammatory mediators are increased before abnormal neural remodeling occurs, reporting changes as early as 3 hours and persisting as long as 3 months. However, additional studies describing dose-concordance would improve empirical support. |
|||
|
Increase, Abnormal Neural Remodeling (KE#2098) → Impairment, Learning and Memory (AO, KE#341) |
Moderate Multiple studies suggest dose- and time-response effects of deposited energy leading to abnormal neural remodeling and impaired learning and memory. However, additional studies at low doses would improve empirical support. Also, discrepancies in the data may be due to experimental set up and type of exposure from the stressor. |
|||
|
Deposition of Energy (MIE, KE#1686) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate Multiple studies suggest dose- and time-response effects of deposition of energy to abnormal neural remodeling. Studies report changes at very low doses. However, responses may be dependent on exposure type. Also, additional studies describing time-concordance would improve empirical support. |
|||
|
Deposition of Energy (MIE, KE#1686) → Impairment, Learning and Memory (AO, KE#341) |
Moderate Various studies show that ionizing radiation can lead to impairments in learning and memory in a dose and time dependent manner. Although the impairment to learning and memory is well-studied across various doses and over multiple time points, studies often do not show impaired learning and memory with every cognitive test used, contributing to inconsistency in the relationship. |
|||
|
Deposition of Energy (MIE, KE#1686) → Increase, DNA Strand Breaks (KE#1635) |
High There is ample empirical evidence demonstrating the relationship between deposition of energy and increase, DNA strand breaks. Multiple studies in various models show both dose-concordance and time-concordance. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Increase, Abnormal Neural Remodeling (KE#2098) |
Moderate Multiple studies demonstrate that increased DNA strand breaks lead to increased abnormal neural remodeling. However, additional studies describing both dose-concordance and time-concordance would improve empirical support. |
|||
|
Increase, Pro-inflammatory Mediators (KE#2097) → Impairment, Learning and Memory (AO, KE#341) |
Moderate Evidence shows that pro-inflammatory mediators increase at lower or the same stressor doses than impaired learning. Also, pro-inflammatory mediators increase before impaired learning and memory is observed. Significant inconsistencies in empirical support across taxa and species that do not align with the expected pattern have not been identified. |
|||
Essentiality
|
Support for Essentiality of KEs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
|
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from specifically designed experimental studies illustrating essentiality for at least one of the important KEs |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE |
No or contradictory experimental evidence of the essentiality of any of the KEs |
||
|
MIE, KE#1686: Deposition of energy |
Moderate Deposition of energy is difficult to test for essentiality as deposition of energy is a physical stressor and cannot be blocked/decreased using chemicals. In the absence of energy deposition or presence of shielding as demonstrated there should be no alterations to the relevant downstream KE. |
||||
|
KE#1392: Oxidative stress |
Moderate Treatments with antioxidants, which reduce oxidative stress, attenuate downstream microglial activation and DNA strand breaks. |
||||
|
KE#1635: Increase, DNA Strand Breaks |
Moderate Prevention of DNA strand breaks, for example treatment with mesenchymal stem cell-conditioned medium or minocycline, has restored altered signaling and prevented abnormal neural remodeling. |
||||
|
KE#2244: Altered Stress Response Signaling |
Moderate Knockout models or inhibition of key signaling molecules, have all been shown to influence the effects of altered stress response signaling on abnormal neural remodeling through the attenuation of stressor-induced changes in neuronal morphology and growth. The KE has also been shown to be modulated by sex and exercise. |
||||
|
KE#1492: Tissue Resident Cell Activation |
Moderate For example, the attenuation of the activation of tissue-resident cells and consequent reduction in pro- inflammatory mediators has been reported using multiple drugs |
||||
|
KE#2097: Increase, Pro-inflammatory Mediators |
Moderate Treatments with anti-inflammatory drugs, antioxidants or hormones have influenced the effects of pro- inflammatory mediators and improved neuronal structure and function. Anti-inflammatory drugs have also influenced the effects of pro-inflammatory mediators and rescued the impairments seen in learning and memory. |
||||
|
KE#2098: Increase, Abnormal Neural Remodeling |
Moderate No identified studies describe essentiality of neural remodeling as it cannot be blocked / decreased using chemicals.
|
||||
Known Modulating Factors
Multiple factors can modulate this AOP, most of which are listed in the table below. Other modulating factors that influence the AOP are knockout models or receptor antagonists (Tian et al., 2020; Chow, Li and Wong, 2000; Limoli et al., 2004; Eom et al., 2016; Kanzawa et al., 2006; Zhang et al. 2018; Green et al., 2012; Ryan et al., 2013; Wu et al., 2012; Chen and Palmer, 2013; Shi et al., 2017).
|
Modulating Factor |
MF details |
Effects on the KER |
References |
|
Antioxidants |
Catalase, glutathione peroxidase, superoxide dismutase, peroxiredoxins, vitamins E, C, carotene, lutein, zeaxanthin, selenium, zinc, alpha-lipoic acid, melatonin, gingko biloba leaf, fermented gingo biloba leaf, Nigella sativa oil, thymoquinone, ferulic acid, Kukoamine A, curcumin, high antioxidant diet, α-tocopherol, α-lipoic acid |
Adding or withholding antioxidants will decrease or increase the level of oxidative stress respectively |
Zigman et al., 1995; Belkacémi et al., 2001; Chitchumroonchokchai et al., 2004; Fatma et al., 2005; Jiang et al., 2006; Fletcher, 2010; Karimi et al., 2017; El-Mesallamy et al., 2018; Hua et al., 2019; Kang et al., 2020; Yang et al., 2020; Manda et al., 2008; Limoli et al., 2007; Manda et al., 2007a; Ismail et al., 2016; Demir et al., 2019; Chen et al., 2021, Zhang et al., 2017, Wang et al., 2017; Daverey & Agrawal, 2016, Ávila-Escalante et al., 2020, Hladik & Tapio, 2016, Manda et al., 2007b |
|
Drugs |
Modulators of tissue resident cell activation (e.g. tamoxifen, retinoic acid, NAC, SP 600125, NS-398), pro-inflammatory mediators (e.g. MW-151, lidocaine, E-EPA, NSPP, α-MSH), and drugs that inhibit DNA damage (lithium chloride, minocycline). |
Several drugs have attenuated the activation of tissue-resident cells and reduced the levels of pro-inflammatory mediators to consequently ameliorate the downstream KE. Drugs that inhibit DNA damage reduced the expression of γ-H2AX and reduced cellular apoptosis. |
Liu et al., 2010; van Neerven et al., 2010; Komatsu et al., 2017; Ramanan, 2008; Kyrkanides et al., 2002; Bhat et al., 2020; Gonzalez et al., 2009; Jenrow et al., 2013; Taepavarapruk & Song, 2010; Tan et al., 2014; Zhang et al., 2017; Zanni et al., 2015 |
|
Age |
Age of organism |
Aging can impact multiple KEs. For example, older organisms have lower levels of antioxidants and an increased likelihood of oxidative stress. Older age is also associated with greater tissue resident cell activation and aging tissue becomes more sensitive to immune signals and increases inflammation. Age is associated with reduced hippocampal neurogenesis and greater radiation-related decrements in learning and memory. |
Liguori et al., 2018; Hanslik, Marino & Ulland, 2021; Casciati et al., 2016; Patterson, 2015; Barrientos et al., 2009; Barrientos et al., 2012. |
|
Sex |
Sex of organism |
The sex of the organism studied can impact several KEs. For example, male mice typically showed an increase in microglia activation, while female mice showed no significant changes. Female mice were also protected from radiation-induced impairments in learning and memory. However, not all studies found this trend. |
Krukowski et al., 2018a; Parihar et al., 2020; Raber et al., 2019 |
|
Prior Disease |
Neurodegenerative diseases like Alzheimer’s and Parkinson’s |
Generates an environment of increased oxidative stress and promotes the activation of glial cells. |
Hanslik, Marino & Ulland, 2021 |
|
Genetics |
Polymorphism that increases the expression of the APOE4 gene increases the risk of developing Alzheimer’s diseases, which generally consists of a decline in memory, thinking and language. MicroRNA expression such as miR-711. |
In homozygous human APOE4 knock-in mice, a dramatic increase in pro-inflammatory cytokines TNF-alpha, IL-1β and IL-6 was seen after LPS injection compared to the APOE2 and APOE3 alleles, suggesting that APOE4 is implicated in a greater inflammatory response. Inhibition of miR-711 reduced DNA damage responses and signaling molecules. |
Hunsberger et al., 2019; Zhu et al., 2012; Sabirzhanov et al., 2020
|
|
Exercise |
Forced running in 30-min intervals twice per day, 5 times per week for 3 weeks. |
Forced running after irradiation completely restored the levels of the signaling molecules in the BDNF-pCREB pathway and slightly restored neurogenesis. |
Ji et al., 2014 |
Quantitative Understanding
Overall quantitative understanding for the KERs in the AOP is low. Despite evidence supporting the KERs, there is limited understanding of the trends of the relationships between KEs. In the KERs of this AOP, there are positive relationships between the KEs (i.e., an increase in the upstream KE elicits a change in the downstream KE); however, the trends and shapes of the relationships are not well established due to differences in experimental parameters, such as model, radiation type, doses, dose rate, and time of measurements. The measures of the KEs cannot be precisely predicted based on relevant measures of the other KEs in the KER and the quantitative descriptions does not account for all known modulating factors and feedback or feedforward mechanisms.
|
Review of the Quantitative Understanding for each KER |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
To what extent can a change in a KEdownstream can be predicted for a KEupstream? With what precision the uncertainty in the prediction of the KEdownstream can be quantified? To what extent are the known modulating factors of feedback mechanisms accounted for? To what extent to which the relationships described can be reliably generalized across the applicability domain of the KER? |
Change in KEdownstream can be precisely predicted based on a relevant measure of KEupstream; Uncertainty in the quantitative prediction can be precisely estimated from the variability in the relevant KEupstream measure; Known modulating factors and feedback/ feedforward mechanisms are accounted for in the quantitative description; Evidence that the quantitative relationship between the KEs generalizes across the relevant applicability domain of the KER |
Change in KEdownstream can be precisely predicted based on relevant measure of KEupstream; Uncertainty in the quantitative prediction is influenced by factors other than the variability in the relevant KEupstream measure; Quantitative description does not account for all known modulating factors and/or known feedback/ feedforward mechanisms; Quantitative relationship has only been demonstrated for a subset of the overall applicability domain of the KER |
Only a qualitative or semi-quantitative prediction of the change in KEdownstream can be determined from a measure of KEupstream; Known modulating factors and feedback/ feedforward mechanisms are not accounted for; Quantitative relationship has only been demonstrated for a narrow subset of the overall applicability domain of the KER |
|
|
Deposition of Energy (MIE, KE#1686) → Oxidative Stress (KE#1392) |
Moderate The evidence for this KER suggests that increased deposition of energy elicits increased oxidative stress, and this is supported by measurements from a large range of doses and dose rates. Despite a large amount of available evidence, the shape of the relationship is not clear, and may depend on the endpoints used to measure oxidative stress. |
|||
|
Deposition of Energy (MIE, KE#1686) → Tissue Resident Cell Activation (KE#1492) |
Moderate Despite a moderate amount of evidence supporting this KER such that increased deposition of energy leads to increased tissue resident cell activation, no clear trends have been established to allow for prediction of the precise amount of tissue resident cell activation based on the deposition of energy. It is likely that the abundance of tissue resident cell activation depends on the biological model, radiation type, radiation dose range and radiation dose rate. |
|||
|
Oxidative Stress (KE#1392) → Increase, DNA Strand Breaks (KE#1635) |
Moderate There are models available that predict DNA strand breaks after deposition of energy; however the exact contribution of indirect DNA damage from oxidative stress is unknown. The quantitative understanding of the relationship depends on the biological target, the radiation quality, and the dose. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Altered Stress Response Signaling (KE#2244) |
Low There are no trends or mathematical models that describe this relationship. Quantitative relationships between these two KEs depend on experimental model and dose. |
|||
|
Oxidative Stress (KE#1392) → Tissue Resident Cell Activation (KE#1492) |
Low Overall, the evidence suggests that there is a positive relationship between increased oxidative stress and tissue resident cell activation. However, studies often report a single dose or time-point, making it difficult to quantitatively predict the amount of tissue resident cell activation after an increase in oxidative stress. |
|||
|
Oxidative Stress (KE#1392) → Altered Stress Response Signaling (KE#2244) |
Low A precise quantitative relationship between oxidative stress and altered stress response signalingis difficult to determine because each study uses a different experimental design. The exact changes to signaling pathways due to oxidative stress will depend on the cell type and species. In addition, modulating factors are often not indicated or accounted for in studies. |
|||
|
Altered Stress Response Signaling (KE#2244) → Increase, Abnormal Neural Remodeling (KE#2098) |
Low The complexity of the many synergistic and antagonistic signaling pathways influenced by a stressor will lead to a cumulative change not representative of the change to a single or a few signaling pathways. Therefore, no trend or mathematical model has been established to quantitatively determine the relationship between altered stress response signaling and endpoints related to abnormal neural remodeling. |
|||
|
Tissue Resident Cell Activation (KE#1492) → Increase, Pro-inflammatory Mediators (KE#2097) |
Low Despite evidence to show that tissue-resident cell activation leads to an increase in pro-inflammatory mediators, it is difficult to compare results and identify a trend as each study uses different models, stressors, doses and time scales. No trend or mathematical model has been established to quantitatively determine the increase in pro-inflammatory mediators after glial cell activation. |
|||
|
Increase, Pro-inflammatory Mediators (KE#2097) → Increase, Neural Remodeling (KE#2098) |
Low Despite studies showing time concordance, no trends or mathematical models have been established that can describe the relationship between increased pro-inflammatory mediators and abnormal neural remodeling. |
|||
|
Increase, Abnormal Neural Remodeling (KE#2098) → Impairment, Learning and Memory (AO, KE#341) |
Low Although a linear relationship was found between reductions in spine density and impairment on a test of learning and memory, no further trends have been established. There may be sex-, dose-, time- and radiation type-dependent differences in the effects of ionizing radiation on abnormal neural remodeling to impairment in learning and memory. |
|||
|
Deposition of Energy (MIE, KE#1686) → Increase, Abnormal Neural Remodeling (KE#2098) |
Low Evidence in support of this KER suggests that deposition of energy elicits a dose-dependent change in neural remodeling. However, the evidence base for the KER has some inconsistencies. In addition, the age of the subjects may impact the trend of the relationship. Ultimately the shape of the trend between the two KEs is unclear and may depend on the biological model used, age of the subjects, type of radiation and radiation dose/dose-rate. |
|||
|
Deposition of Energy (MIE, KE#1686) → Impairment, Learning and Memory (AO, KE#341) |
Low Deposition of energy from ionizing radiation is consistently shown to impair learning and memory. However, no trend or mathematical model has been established to accurately describe this relationship. Much of the evidence for this KER comes from different experimental model species, exposures, time scales and cognitive function tests to assess the AO, making comparisons between studies and quantitative model development difficult. |
|||
|
Deposition of Energy (MIE, KE#1686) → Increase, DNA Strand Breaks (KE#1635) |
High There are models available that predict DNA strand breaks after deposition of energy due to exposure to ionizing radiation. The quantitative understanding of the relationship depends on the biological target, the radiation quality, and the dose and these have been well studied. |
|||
|
Increase, DNA Strand Breaks (KE#1635) → Increase, Abnormal Neural Remodeling (KE#2098) |
Low There are multiple studies that report abnormal neural remodeling in response to DNA strand breaks. However, no trend or mathematical model has been established to accurately describe this relationship. |
|||
|
Pro-inflammatory Mediators (KE#2097) → Impairment, Learning and Memory (AO, KE#341) |
Low Most studies report that an increase in pro-inflammatory mediators to impaired learning and memory; however, semi-quantitative measurements have been described for either upstream or downstream KE. Some, but not all, known modulating factors have been accounted for, mainly consisting of anti-inflammatory treatments. No trends or quantitative models have described this relationship. |
|||
Considerations for Potential Applications of the AOP (optional)
This AOP was developed to bring together mechanistic knowledge in the area of impairments in learning and memory from exposure to radiation. It includes studies from multiple species at multiple life stages and radiation exposures that contain different doses, dose-rates, and radiation qualities. Relevant studies have been selected, consolidated, and reported using the framework.
There are multiple considerations for potential applications of the AOP. Since exposure to radiation can occur in humans from multiple events, including occupational settings, accidental exposures, nuclear events, radiotherapy treatment and space travel, understanding its impact on CNS structure and function is essential. This AOP outlines a biological framework for the connection between the MIE and AO. It can be expanded to other pathophysiologies of the CNS. The qualitative information presented within each KER can be used to inform on risk-model strategies, countermeasure development, and identification of gaps in the evidence base where more research is necessary. Importantly, this AOP is a dynamic document so it can be modified as new evidence emerges.
References
Acharya, M. M. et al. (2010), "Consequences of ionizing radiation-induced damage in human neural stem cells", Free Radical Biology and Medicine, Vol. 49/12, Pergamon, https://doi.org/10.1016/j.freeradbiomed.2010.08.021.
Ahmadi, M. et al. (2022), “Early responses to low-dose ionizing radiation in cellular lens epithelial models”, Radiation research, Vol. 197/1, Radiation Research Society, Bozeman, https://doi.org/10.1667/RADE-20-00284.1
Antonelli, A.F. et al. (2015), "Induction and Repair of DNA DSB as Revealed by H2AX Phosphorylation Foci in Human Fibroblasts Exposed to Low- and High-LET Radiation: Relationship with Early and Delayed Reproductive Cell Death", Radiation Research, Vol 183/4, BioOne, Washington, httrps://doi.org/10.1667/RR13855.1.
Azimzadeh, O. et al. (2015), "Integrative proteomics and targeted transcriptomics analyses in cardiac endothelial cells unravel mechanisms of long-term radiation-induced vascular dysfunction", Journal of Proteome Research, Vol. 14/2, American Chemical Society, Washington, https://doi.org/10.1021/pr501141b
Balasubramanian, D (2000), “Ultraviolet radiation and cataract”, Journal of ocular pharmacology and therapeutics, Vol. 16/3, Mary Ann Liebert Inc., Larchmont, https://doi.org/10.1089/jop.2000.16.285.
Bálentová, S. and M. Adamkov. (2020), "Pathological changes in the central nervous system following exposure to ionizing radiation", Physiological Research, Czech Academy of Sciences, https://doi.org/10.33549/PHYSIOLRES.934309.
Barrientos, R. M. et al. (2009), "Time course of hippocampal IL-1 β and memory consolidation impairments in aging rats following peripheral infection", Brain, Behavior, and Immunity, Vol. 23/1, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbi.2008.07.002.
Barrientos, R. M. et al. (2012), "Aging-related changes in neuroimmune-endocrine function: Implications for hippocampal-dependent cognition", Hormones and Behavior, Vol. 62/3, Elsevier, Amsterdam, https://doi.org/10.1016/j.yhbeh.2012.02.010.
Belkacémi, Y. et al. (2001), “Lens epithelial cell protection by aminothiol WR-1065 and anetholedithiolethione from ionizing radiation”, International journal of cancer, Vol. 96, John Wiley & Sons, Ltd., Hoboken, https://doi.org/10.1002/ijc.10346.
Betlazar, C. et al. (2016), "The impact of high and low dose ionising radiation on the central nervous system", Redox Biology, Vol. 9, Elsevier, Amsterdam, https://doi.org/10.1016/j.redox.2016.08.002.
Bhat, K. et al. (2020), "1-[(4-Nitrophenyl)sulfonyl]-4-phenylpiperazine treatment after brain irradiation preserves cognitive function in mice", Neuro-Oncology, Vol. 22/10, Oxford University Press, Oxford, https://doi.org/10.1093/neuonc/noaa095.
Brooks, A.L., D.G. Hoel & R.J. Preston (2016), "The role of dose rate in radiation cancer risk: evaluating the effect of dose rate at the molecular, cellular and tissue levels using key events in critical pathways following exposure to low LET radiation.", International Journal of Radiation Biology, Vol. 92/8, Taylor & Francis, London, doi:10.1080/09553002.2016.1186301.
Casciati, A. et al. (2016), "Age-related effects of X-ray irradiation on mouse hippocampus", Oncotarget, Vol. 7/19, https://doi.org/10.18632/oncotarget.8575.
Cekanaviciute, E., S. Rosi and S. V. Costes. (2018), "Central nervous system responses to simulated galactic cosmic rays", International Journal of Molecular Sciences, Multidisciplinary Digital Publishing Institute (MDPI) AG, Basel, https://doi.org/10.3390/ijms19113669.
Chen, Z. and T. D. Palmer. (2013), "Differential roles of TNFR1 and TNFR2 signaling in adult hippocampal neurogenesis", Brain, Behavior, and Immunity, Vol. 30, Elsevier Inc., Amsterdam, https://doi.org/10.1016/j.bbi.2013.01.083.
Chen, Y. et al. (2021), “Effects of neutron radiation on Nrf2-regulated antioxidant defense systems in rat lens”, Experimental and therapeutic medicine, Vol. 21/4, Spandidos Publishing Ltd, Athens, https://doi.org/10.3892/etm.2021.9765.
Chitchumroonchokchai, C. et al. (2004), “Xanthophylls and α-tocopherol decrease UVB-induced lipid peroxidation and stress signaling in human lens epithelial cells”, The Journal of Nutrition, Vol. 134/12, American Society for Nutritional Sciences, Bethesda, https://doi.org/10.1093/jn/134.12.3225.
Chow, B. M., Y.-Q. Li and C. S. Wong. (2000), "Radiation-induced apoptosis in the adult central nervous system is p53-dependent", Cell Death & Differentiation, Vol. 7/8, Springer Nature, https://doi.org/10.1038/sj.cdd.4400704.
Cucinotta, F. A. et al. (2014), "Space radiation risks to the central nervous system", Life Sciences in Space Research, Vol. 2, Elsevier Ltd, Amsterdam, https://doi.org/10.1016/j.lssr.2014.06.003.
Daverey, A. and S. K. Agrawal. (2016), "Curcumin alleviates oxidative stress and mitochondrial dysfunction in astrocytes", Neuroscience, Vol. 333, https://doi.org/10.1016/j.neuroscience.2016.07.012.
Davis, S. and S. Laroche. (2006), "Mitogen-activated protein kinase/extracellular regulated kinase signalling and memory stabilization: a review", Genes, Brain and Behavior, Vol. 5, Wiley, https://doi.org/10.1111/j.1601-183X.2006.00230.x.
de Jager, T. L., A. E. Cockrell, S. S. Du Plessis (2017), “Ultraviolet Light Induced Generation of Reactive Oxygen Species”, in: Ultraviolet Light in Human Health, Diseases and Environment, vol 996. Springer Cham, https://doi.org/10.1007/978-3-319-56017-5_2.
Demir, E. et al. (2020), “Nigella sativa oil and thymoquinone reduce oxidative stress in the brain tissue of rats exposed to total head irradiation”, International journal of radiation biology, Vol. 96/2, Informa, London, https://doi.org/10.1080/09553002.2020.1683636.
Deng, Z. et al. (2012), "Radiation-Induced c-Jun Activation Depends on MEK1-ERK1/2 Signaling Pathway in Microglial Cells", (I. Ulasov, Ed.) PLoS ONE, Vol. 7/5, https://doi.org/10.1371/journal.pone.0036739.
Desai, R. I. et al. (2022), "Impact of spaceflight stressors on behavior and cognition: A molecular, neurochemical, and neurobiological perspective", Neuroscience & Biobehavioral Reviews, Vol. 138, Elsevier, Amsterdam, https://doi.org/10.1016/j.neubiorev.2022.104676.
Dong, X. et al. (2015), "Relationship between irradiation-induced neuro-inflammatory environments and impaired cognitive function in the developing brain of mice", International Journal of Radiation Biology, Vol. 91/3, Informa Healthcare, London, https://doi.org/10.3109/09553002.2014.988895.
El-Missiry, M. A. et al. (2018), "Neuroprotective effect of epigallocatechin-3-gallate (EGCG) on radiation-induced damage and apoptosis in the rat hippocampus", International Journal of Radiation Biology, Vol. 94/9, https://doi.org/10.1080/09553002.2018.1492755.
Eom, H. S. et al. (2015), "Ionizing radiation induces neuronal differentiation of Neuro-2a cells via PI3-kinase and p53-dependent pathways", International Journal of Radiation Biology, Vol. 91/7, Informa, London, https://doi.org/10.3109/09553002.2015.1029595.
Falcicchia, C. et al. (2020), "Involvement of p38 MAPK in Synaptic Function and Dysfunction", International Journal of Molecular Sciences, Vol. 21/16, MDPI, Basel, https://doi.org/10.3390/ijms21165624.
Fatma, N. et al. (2005), “Impaired homeostasis and phenotypic abnormalities in Prdx6-/- mice lens epithelial cells by reactive oxygen species: Increased expression and activation of TGFβ”, Cell death and differentiation, Vol. 12, Nature Portfolio, London, https://doi.org/10.1038/sj.cdd.4401597.
Fishman, K. et al. (2009), "Radiation-induced reductions in neurogenesis are ameliorated in mice deficient in CuZnSOD or MnSOD", Free Radical Biology and Medicine, Vol. 47/10, https://doi.org/10.1016/j.freeradbiomed.2009.08.016.
Fletcher, A. E (2010), “Free radicals, antioxidants and eye diseases: evidence from epidemiological studies on cataract and age-related macular degeneration”, Ophthalmic Research, Vol. 44, Karger International, Basel, https://doi.org/10.1159/000316476.
Ganea, E. and J. J. Harding (2006), “Glutathione-related enzymes and the eye”, Current eye research, Vol. 31/1, Informa, London, https://doi.org/10.1080/02713680500477347.
Giedzinski, E. et al. (2005), “Efficient production of reactive oxygen species in neural precursor cells after exposure to 250 MeV protons”, Radiation research, Vol. 164/4, Radiation Research Society, Bozeman, https://doi.org/10.1667/rr3369.1.
Gonzalez, P. V. et al. (2009), "Memory impairment induced by IL-1β is reversed by α-MSH through central melanocortin-4 receptors", Brain, Behavior, and Immunity, Vol. 23/6, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbi.2009.03.001.
Green, H. F. et al. (2012), "A role for interleukin-1β in determining the lineage fate of embryonic rat hippocampal neural precursor cells", Molecular and Cellular Neuroscience, Vol. 49/3, Elsevier Inc., Amsterdam, https://doi.org/10.1016/j.mcn.2012.01.001.
Greene-Schloesser, D. et al. (2012), "Radiation-induced brain injury: A review", Frontiers in Oncology, Vol. 2, Frontiers, Lausanne, https://doi.org/10.3389/fonc.2012.00073.
Hanslik, K. L., K. M. Marino and T. K. Ulland. (2021), "Modulation of Glial Function in Health, Aging, and Neurodegenerative Disease", Frontiers in Cellular Neuroscience, Vol. 15, https://doi.org/10.3389/fncel.2021.718324.
Hamada, N. et al. (2006), “Histone H2AX phosphorylation in normal human cells irradiated with focused ultrasoft X rays: evidence for chromatin movement during repair”, Radiation Research, Vol. 166/1, Radiation Research Society, United States, https://doi.org/10.1667/RR3577.1
Hladik, D. and S. Tapio. (2016), "Effects of ionizing radiation on the mammalian brain", Mutation Research - Reviews in Mutation Research, Vol. 770, Elsevier B.V., Amsterdam, https://doi.org/10.1016/j.mrrev.2016.08.003.
Huang, Y. et al. (2021), "Mesenchymal Stem Cell-Conditioned Medium Protects Hippocampal Neurons From Radiation Damage by Suppressing Oxidative Stress and Apoptosis", Dose-Response, Vol. 19/1, SAGE publications, https://doi.org/10.1177/1559325820984944.
Hua, H. et al. (2019), “Protective effects of lanosterol synthase up-regulation in UV-B-induced oxidative stress”, Frontiers in pharmacology, Vol. 10, Frontiers Media SA, Lausanne, https://doi.org/10.3389/fphar.2019.00947.
Hunsberger, H. C. et al. (2019), "The role of APOE4 in Alzheimer’s disease: strategies for future therapeutic interventions", Neuronal Signaling, Vol. 3/2, Portland Press, London, https://doi.org/10.1042/NS20180203.
Hwang, S. Y. et al. (2006), "Ionizing radiation induces astrocyte gliosis through microglia activation", Neurobiology of Disease, Vol. 21/3, Academic Press, https://doi.org/10.1016/j.nbd.2005.08.006.
International commission on Radiological Protection (ICRP). (2007), “The 2007 recommendations of the International Commission on Radiological Protection.”, Ann ICRP 37, ICRP Publication 103.
Ismail, A. F. and S. M. El-Sonbaty (2016), “Fermentation enhances Ginkgo biloba protective role on γ-irradiation induced neuroinflammatory gene expression and stress hormones in rat brain”, Journal of photochemistry and photobiology. B, Biology, Vol. 158, Elsevier, Amsterdam, https://doi.org/10.1016/j.jphotobiol.2016.02.039.
Jenrow, K. A. et al. (2013), "Selective Inhibition of Microglia-Mediated Neuroinflammation Mitigates Radiation-Induced Cognitive Impairment", Radiation Research, Vol. 179/5, BioOne, https://doi.org/10.1667/RR3026.1.
Ji, J. et al. (2014), "Forced running exercise attenuates hippocampal neurogenesis impairment and the neurocognitive deficits induced by whole-brain irradiation via the BDNF-mediated pathway", Biochemical and Biophysical Research Communications, Vol. 443/2, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbrc.2013.12.031.
Jiang, Q. et al. (2006), “UV radiation down-regulates Dsg-2 via Rac/NADPH oxidase-mediated generation of ROS in human lens epithelial cells”, International Journal of Molecular Medicine, Vol. 18/2, Spandidos Publishing Ltd, Athens, https://doi.org/10.3892/ijmm.18.2.381.
Serment-Guerrero, J. et al. (2012), "Evidence of DNA double strand breaks formation in Escherichia coli bacteria exposed to alpha particles of different LET assessed by the SOS response", Applied Radiation and Isotopes, Vol. 71, Elsevier, Amsterdam, https://doi.org/10.1016/j.apradiso.2012.05.007.
Kalm, M., K. Roughton and K. Blomgren. (2013), "Lipopolysaccharide sensitized male and female juvenile brains to ionizing radiation", Cell Death & Disease, Vol. 4/12, https://doi.org/10.1038/cddis.2013.482.
Kang, L. et al. (2020), “Ganoderic acid A protects lens epithelial cells from UVB irradiation and delays lens opacity”, Chinese journal of natural medicines, Vol. 18/12, Elsevier, Amsterdam, https://doi.org/10.1016/S1875-5364(20)60037-1.
Kanzawa, T. et al. (2006), "Ionizing radiation induces apoptosis and inhibits neuronal differentiation in rat neural stem cells via the c-Jun NH2-terminal kinase (JNK) pathway", Oncogene, Vol. 25/26, Springer Nature, https://doi.org/10.1038/sj.onc.1209414.
Karimi, N. et al. (2017), “Radioprotective effect of hesperidin on reducing oxidative stress in the lens tissue of rats”, International Journal of Pharmaceutical Investigation, Vol. 7/3, Phcog Net, Bengaluru, https://doi.org/10.4103/jphi.JPHI_60_17.
Katsura, M. et al. (2021), "Recognizing Radiation-induced Changes in the Central Nervous System: Where to Look and What to Look For", RadioGraphics, Vol. 41/1, https://doi.org/10.1148/rg.2021200064.
Kiffer, F. et al. (2019a), "Late Effects of 16O-Particle Radiation on Female Social and Cognitive Behavior and Hippocampal Physiology", Radiation Research, Vol. 191/3, BioOne, Washington, https://doi.org/10.1667/RR15092.1.
Kiffer, F., M. Boerma and A. Allen. (2019b), "Behavioral effects of space radiation: A comprehensive review of animal studies", Life Sciences in Space Research, Vol. 21, Elsevier, Amsterdam, https://doi.org/10.1016/j.lssr.2019.02.004.
Komatsu, W. et al. (2017), “Nasunin inhibits the lipopolysaccharide-induced pro-inflammatory mediator production in RAW264 mouse macrophages by suppressing ROS-mediated activation of PI3 K/Akt/NF-κB and p38 signaling pathways”, Bioscience, Biotechnology, and Biochemistry, Vol. 81/10, Elsevier, https://doi.org/10.1080/09168451.2017.1362973
Kozbenko, T. et al. (2022), "Deploying elements of scoping review methods for adverse outcome pathway development: a space travel case example", International Journal of Radiation Biology, Vol. 98/12, https://doi.org/10.1080/09553002.2022.2110306.
Krukowski, K. et al. (2018a), "Female mice are protected from space radiation-induced maladaptive responses", Brain, Behavior, and Immunity, Vol. 74, Academic Press Inc., https://doi.org/10.1016/j.bbi.2018.08.008.
Kyrkanides, S. et al. (1999), "TNFα and IL-1β mediate intercellular adhesion molecule-1 induction via microglia-astrocyte interaction in CNS radiation injury", Journal of Neuroimmunology, Vol. 95/1–2, Elsevier, Amsterdam, https://doi.org/10.1016/S0165-5728(98)00270-7.
Kyrkanides, S. et al. (2002), "Cyclooxygenase-2 modulates brain inflammation-related gene expression in central nervous system radiation injury", Molecular Brain Research, Vol. 104/2, Elsevier, https://doi.org/10.1016/S0169-328X(02)00353-4.
Lawrence, T. & Gilroy D. W. (2007) “Chronic inflammation: a failure of resolution?” International journal of experimental pathology, Vol. 88/2, John Wiley & Sons, Hoboken, https://doi.org/10.1111/j.1365-2613.2006.00507.x.
Lee, W. H. et al. (2010), "Irradiation induces regionally specific alterations in pro-inflammatory environments in rat brain", International Journal of Radiation Biology, Vol. 86/2, Informa, London, https://doi.org/10.3109/09553000903419346.
Lee, K., A. Lee and I. Choi. (2017), "Melandrii Herba Extract Attenuates H2O2-Induced Neurotoxicity in Human Neuroblastoma SH-SY5Y Cells and Scopolamine-Induced Memory Impairment in Mice", Molecules, Vol. 22/10, MDPI, Basel, https://doi.org/10.3390/molecules22101646.
Lee, K. H., M. Cha and B. H. Lee. (2021), "Crosstalk between Neuron and Glial Cells in Oxidative Injury and Neuroprotection", International Journal of Molecular Sciences, Vol. 22/24, https://doi.org/10.3390/ijms222413315.
Lehtinen, M. and A. Bonni. (2006), "Modeling Oxidative Stress in the Central Nervous System", Current Molecular Medicine, Vol. 6/8, https://doi.org/10.2174/156652406779010786.
Li, J. et al. (2013), "Oxidative Stress and Neurodegenerative Disorders", International Journal of Molecular Sciences, Vol. 14/12, https://doi.org/10.3390/ijms141224438.
Liguori, I. et al. (2018), "Oxidative stress, aging, and diseases", Clinical Interventions in Aging, Vol.13, https://doi.org/10.2147/CIA.S158513.
Limoli, C. L. et al. (2004), “Radiation response of neural precursor cells: linking cellular sensitivity to cell cycle checkpoints, apoptosis and oxidative stress”, Radiation research, Vol. 161/1, Radiation Research Society, Bozeman, https://doi.org/10.1667/rr3112.
Limoli, C. L. et al. (2007), “Redox changes induced in hippocampal precursor cells by heavy ion irradiation”, Radiation and environmental biophysics, Vol. 46/2, Springer, London, https://doi.org/10.1007/s00411-006-0077-9.
Liu, J. L. et al. (2010), "Tamoxifen alleviates irradiation-induced brain injury by attenuating microglial inflammatory response in vitro and in vivo", Brain Research, Vol. 1316, Elsevier B.V., https://doi.org/10.1016/j.brainres.2009.12.055.
Long, H.-Z. et al. (2021), "PI3K/AKT Signal Pathway: A Target of Natural Products in the Prevention and Treatment of Alzheimer’s Disease and Parkinson’s Disease", Frontiers in Pharmacology, Vol. 12, Frontiers, https://doi.org/10.3389/fphar.2021.648636.
Madsen, T. M. et al. (2003), "Arrested neuronal proliferation and impaired hippocampal function following fractionated brain irradiation in the adult rat", Neuroscience, Vol. 119/3, Elsevier Ltd, https://doi.org/10.1016/S0306-4522(03)00199-4.
Manda, K. et al. (2007a), “Melatonin attenuates radiation-induced learning deficit and brain oxidative stress in mice”, Acta neurobiologiae experimentalis, Vol. 67/1, Nencki Institute of Experimental Biology, Warsaw, pp. 63 –70.
Manda, K. et al. (2007b), "Radiation-induced cognitive dysfunction and cerebellar oxidative stress in mice: Protective effect of α-lipoic acid", Behavioural Brain Research, Vol. 177/1, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbr.2006.11.013.
Manda, K., M. Ueno and K. Anzai (2008), “Memory impairment, oxidative damage and apoptosis induced by space radiation: ameliorative potential of alpha-lipoic acid”, Behavioural brain research, Vol. 187/2, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbr.2007.09.033.
Mazzucchelli, C. and R. Brambilla. (2000), "Ras-related and MAPK signalling in neuronal plasticity and memory formation", Cellular and Molecular Life Sciences, Vol. 57/4, Springer Nature, https://doi.org/10.1007/PL00000722.
McHugh, D. and J. Gil. (2018), "Senescence and aging: Causes, consequences, and therapeutic avenues", Journal of Cell Biology, Vol. 217/1, Rockefeller University Press, New York, https://doi.org/10.1083/jcb.201708092.
Mielke, K. and T. Herdegen. (2000), "JNK and p38 stresskinases — degenerative effectors of signal-transduction-cascades in the nervous system", Progress in Neurobiology, Vol. 61/1, Elsevier, Amsterdam, https://doi.org/10.1016/S0301-0082(99)00042-8.
Mosconi, M. et al. (2011), "53BP1 and MDC1 foci formation in HT-1080 cells for low- and high-LET microbeam irradiations", Radiation and Environmental Biophysics, Vol. 50/3, Springer Nature, Berlin, https://doi.org/10.1007/s00411-011-0366-9.
Monje, M. L. and T. Palmer. (2003), "Radiation injury and neurogenesis", Current Opinion in Neurology, Vol. 16/2, Ovid Technologies (Wolters Kluwer Health), https://doi.org/10.1097/01.wco.0000063772.81810.b7.
Mousa, A. and M. Bakhiet. (2013), "Role of Cytokine Signaling during Nervous System Development", International Journal of Molecular Sciences, Vol. 14/7, MDPI, Basel, https://doi.org/10.3390/ijms140713931.
Nagane, M. et al. (2021), "DNA damage response in vascular endothelial senescence: Implication for radiation-induced cardiovascular diseases", Journal of Radiation Research, Vol. 62/4, Oxford University Press, Oxford, https://doi.org/10.1093/jrr/rrab032
Nathan, C. and A. Ding. (2010) “Nonresolving inflammation.” Cell, Vol. 140/6,Cell press, Cambridge, https://doi.org/10.1016/j.cell.2010.02.029
National Council on Radiation Protection and Measures (NCRP). (2016). Commentary No. 25 – Potential for central nervous system effects from radiation exposure during space activities phase I: Overview.
Nikjoo, H. et al. (2001), "Computational approach for determining the spectrum of DNA damage induced by ionizing radiation.", Radiation Research, Vol. 156/5 Pt 2, BioOne, Washington, https://doi.org/10.1667/0033-7587(2001)156[0577:cafdts]2.0.co;2
Nikjoo, H. et al. (2016), "Radiation track, DNA damage and response—a review", Reports on Progress in Physics, Vol. 79/11, IOP Publishing, Bristol, https://doi.org/10.1088/0034-4885/79/11/116601.
Nebreda, A. R. and A. Porras. (2000), "p38 MAP kinases: beyond the stress response", Trends in Biochemical Sciences, Vol. 25/6, Elsevier, Amsterdam, https://doi.org/10.1016/S0968-0004(00)01595-4.
Parihar, V. K. et al. (2016), "Cosmic radiation exposure and persistent cognitive dysfunction", Scientific Reports, Vol. 6/1, Nature Publishing Group, https://doi.org/10.1038/srep34774.
Parihar, V. K. et al. (2018), "Persistent nature of alterations in cognition and neuronal circuit excitability after exposure to simulated cosmic radiation in mice", Experimental Neurology, Vol. 305, Elsevier B.V., https://doi.org/10.1016/j.expneurol.2018.03.009.
Parihar, V. K. et al. (2020), "Sex-Specific Cognitive Deficits Following Space Radiation Exposure", Frontiers in behavioral neuroscience, Vol. 14, Frontiers, https://doi.org/10.3389/fnbeh.2020.535885.
Park, D. H. et al. (2021), "Neuroprotective Effect of Gallocatechin Gallate on Glutamate-Induced Oxidative Stress in Hippocampal HT22 Cells", Molecules, Vol. 26/5, MDPI, Basel, https://doi.org/10.3390/molecules26051387.
Patterson, S. L. (2015), "Immune dysregulation and cognitive vulnerability in the aging brain: Interactions of microglia, IL-1β, BDNF and synaptic plasticity", Neuropharmacology, Vol. 96, Elsevier B.V., https://doi.org/10.1016/j.neuropharm.2014.12.020.
Ping, Z. et al. (2020), "Oxidative Stress in Radiation-Induced Cardiotoxicity", Oxidative Medicine and Cellular Longevity, Vol. 2020, Hindawi, London, https://doi.org/10.1155/2020/3579143
Pius-Sadowska, E. et al. (2016), "Alteration of Selected Neurotrophic Factors and their Receptor Expression in Mouse Brain Response to Whole-Brain Irradiation", Radiation Research, Vol. 186/5, BioOne, https://doi.org/10.1667/RR14457.1.
Prieto, G. A. and C. W. Cotman. (2017), "Cytokines and cytokine networks target neurons to modulate long-term potentiation", Cytokine & Growth Factor Reviews, Vol. 34, Elsevier, Amsterdam, https://doi.org/10.1016/j.cytogfr.2017.03.005.
Raber, J. et al. (2004), "Radiation-induced cognitive impairments are associated with changes in indicators of hippocampal neurogenesis", Radiation Research, Vol. 162/1, Allen Press, https://doi.org/10.1667/RR3206.
Raber, J. et al. (2019), "Combined Effects of Three High-Energy Charged Particle Beams Important for Space Flight on Brain, Behavioral and Cognitive Endpoints in B6D2F1 Female and Male Mice", Frontiers in physiology, Vol. 10, Frontiers, https://doi.org/10.3389/fphys.2019.00179.
Rai, S. N. et al. (2019), "The Role of PI3K/Akt and ERK in Neurodegenerative Disorders", Neurotoxicity Research, Vol. 35/3, Elsevier, Amsterdam, https://doi.org/10.1007/s12640-019-0003-y.
Ramalingam, M. and S.-J. Kim. (2012), "Reactive oxygen/nitrogen species and their functional correlations in neurodegenerative diseases", Journal of Neural Transmission, Vol. 119/8, Springer Nature, Berlin, https://doi.org/10.1007/s00702-011-0758-7.
Ramanan, S. et al. (2008), "PPARα ligands inhibit radiation-induced microglial inflammatory responses by negatively regulating NF-κB and AP-1 pathways", Free Radical Biology and Medicine, Vol. 45/12, Elsevier B.V., https://doi.org/10.1016/j.freeradbiomed.2008.09.002.
Rodgers, E. E. and A. B. Theibert. (2002), "Functions of PI 3‐kinase in development of the nervous system", International Journal of Developmental Neuroscience, Vol. 20/3–5, Wiley, https://doi.org/10.1016/S0736-5748(02)00047-3.
Rogakou, E. P. et al. (1999), "Megabase Chromatin Domains Involved in DNA Double-Strand Breaks in Vivo", Journal of Cell Biology, Vol. 146/5, Rockefeller University Press, New York, https://doi.org/10.1083/jcb.146.5.905.
Rola, R. et al. (2004), "Radiation-induced impairment of hippocampal neurogenesis is associated with cognitive deficits in young mice", Experimental Neurology, Vol. 188/2, Academic Press Inc., https://doi.org/10.1016/j.expneurol.2004.05.005.
Romanella, S. M. et al. (2020), "Noninvasive Brain Stimulation & Space Exploration: Opportunities and Challenges", Neuroscience & Biobehavioral Reviews, Vol. 119, https://doi.org/10.1016/j.neubiorev.2020.09.005.
Rothkamm, K. and M. Löbrich. (2003), "Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses", Proceedings of the National Academy of Sciences, Vol. 100/9, National Academy of Sciences, https://doi.org/0.1073/pnas.0830918100.
Rübe, C. E. et al. (2008), "DNA Double-Strand Break Repair of Blood Lymphocytes and Normal Tissues Analysed in a Preclinical Mouse Model: Implications for Radiosensitivity Testing", Clinical Cancer Research, Vol. 14/20, American Association for Cancer Research, Washington, https://doi.org/10.1158/1078-0432.CCR-07-5147.
Ryan, S. M. et al. (2013), "Negative regulation of TLX by IL-1β correlates with an inhibition of adult hippocampal neural precursor cell proliferation", Brain, Behavior, and Immunity, Vol. 33, Elsevier, Amsterdam, https://doi.org/10.1016/j.bbi.2013.03.005.
Sabirzhanov, B. et al. (2020), "Irradiation-Induced Upregulation of miR-711 Inhibits DNA Repair and Promotes Neurodegeneration Pathways", International Journal of Molecular Sciences, Vol. 21/15, Multidisciplinary Digital Publishing Institute (MDPI) AG, Basel, https://doi.org/10.3390/ijms21155239.
Saraiva, C. et al. (2019), "Histamine modulates hippocampal inflammation and neurogenesis in adult mice", Scientific Reports, Vol. 9/1, Springer Nature, Berlin, https://doi.org/10.1038/s41598-019-44816-w.
Schmidt-Ullrich, R. K. et al. (2000), "Signal transduction and cellular radiation responses.", Radiation research, Vol. 153/3, BioOne, https://doi.org/10.1667/0033-7587(2000)153[0245:stacrr]2.0.co;2
Schnegg, C. I. et al. (2012), "PPARδ prevents radiation-induced proinflammatory responses in microglia via transrepression of NF-κB and inhibition of the PKCα/MEK1/2/ERK1/2/AP-1 pathway", Free Radical Biology and Medicine, Vol. 52/9, Pergamon, https://doi.org/10.1016/J.FREERADBIOMED.2012.02.032.
Sherrin, T., T. Blank and C. Todorovic. (2011), "c-Jun N-terminal kinases in memory and synaptic plasticity", Reviews in the Neurosciences, Vol. 22/4, De Gruyter, https://doi.org/10.1515/rns.2011.032.
Shi, Q. et al. (2017), "Complement C3 deficiency protects against neurodegeneration in aged plaque-rich APP/PS1 mice", Science Translational Medicine, Vol. 9/392, American Association for the Advancement of Science, Washington, https://doi.org/10.1126/scitranslmed.aaf6295.
Simpson, D. S. A. and P. L. Oliver. (2020), "ROS Generation in Microglia: Understanding Oxidative Stress and Inflammation in Neurodegenerative Disease", Antioxidants, Vol. 9/8, https://doi.org/10.3390/antiox9080743.
Slezak, J. et al. (2017), “Potential markers and metabolic processes involved in the mechanism of radiation-induced heart injury”, Canadian journal of physiology and pharmacology, Vol. 95/10, Canadian Science Publishing, Ottawa, https://doi.org/10.1139/cjpp-2017-0121.
Sutherland, B. M. et al. (2000), "Clustered DNA damages induced in isolated DNA and in human cells by low doses of ionizing radiation", Proceedings of the National Academy of Sciences, Vol. 97/1, National Academy of Sciences, https://doi.org/10.1073/pnas.97.1.103.
Suman, S. et al. (2013), “Therapeutic and space radiation exposure of mouse brain causes impaired DNA repair response and premature senescence by chronic oxidant production”, Aging, Vol. 5/8, Impact Journals, Orchard Park, https://doi.org/10.18632/aging.100587.
Taepavarapruk, P. and C. Song. (2010), "Reductions of acetylcholine release and nerve growth factor expression are correlated with memory impairment induced by interleukin-1β administrations: effects of omega-3 fatty acid EPA treatment", Journal of Neurochemistry, Vol. 112/4, Wiley https://doi.org/10.1111/j.1471-4159.2009.06524.x.
Tan, H. et al. (2014), "Critical role of inflammatory cytokines in impairing biochemical processes for learning and memory after surgery in rats", Journal of Neuroinflammation, Vol. 11/1, Springer Nature, https://doi.org/10.1186/1742-2094-11-93.
Tian, R. et al. (2020), "miR-137 prevents inflammatory response, oxidative stress, neuronal injury and cognitive impairment via blockade of Src-mediated MAPK signaling pathway in ischemic stroke", Aging, Vol. 12/11, https://doi.org/10.18632/aging.103301.
Tomé, W. A. et al. (2015), "Hippocampal-dependent neurocognitive impairment following cranial irradiation observed in pre-clinical models: current knowledge and possible future directions", The British Journal of Radiobiology, Vol. 89/1057, British Institute of Radiology, https://doi.org/10.1259/bjr.20150762.
Toricelli, M., Pereira, A. A. R., Souza Abrao, G., Malerba, H. N., Maia, J., Buck, H. S., & Viel, T. A. (2021). Mechanisms of neuroplasticity and brain degeneration: strategies for protection during the aging process. Neural regeneration research, 16(1), 58–67. https://doi.org/10.4103/1673-5374.286952.
Turnquist, C., B. T. Harris and C. C. Harris. (2020), "Radiation-induced brain injury: current concepts and therapeutic strategies targeting neuroinflammation", Neuro-Oncology Advances, Vol. 2/1, Oxford University Press, Oxford, https://doi.org/10.1093/noajnl/vdaa057.
United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). (2008), “Sources and effects of ionizing radiation”, UNSCEAR 2008 Report, Vol 1, UN Publications.
Valerie, K. et al. (2007), "Radiation-induced cell signaling: inside-out and outside-in", Molecular Cancer Therapeutics, Vol. 6/3, American Association for Cancer Research, https://doi.org/10.1158/1535-7163.MCT-06-0596
Valliéres, L. et al. (2002), "Reduced hippocampal neurogenesis in adult transgenic mice with chronic astrocytic production of interleukin-6", Journal of Neuroscience, Vol. 22/2, Society for Neuroscience, Washington, https://doi.org/10.1523/jneurosci.22-02-00486.2002.
van Neerven, S. et al. (2010), "Inflammatory cytokine release of astrocytes in vitro is reduced by all-trans retinoic acid", Journal of Neuroimmunology, Vol. 229/1–2, Elsevier B.V., https://doi.org/10.1016/j.jneuroim.2010.08.005.
Wang, Y. L. et al. (2017), "Protective Effect of Curcumin Against Oxidative Stress-Induced Injury in Rats with Parkinson’s Disease Through the Wnt/ β-Catenin Signaling Pathway", Cellular Physiology and Biochemistry, Vol. 43/6, https://doi.org/10.1159/000484302.
Wang, H. et al. (2019a), “Radiation-induced heart disease: a review of classification, mechanism and prevention”, International Journal of Biological Sciences, Vol. 15/10, Ivyspring International Publisher, Sydney, https://doi.org/10.7150/ijbs.35460.
Whoolery, C. W. et al. (2017), "Whole-body exposure to 28Si-radiation dose-dependently disrupts dentate gyrus neurogenesis and proliferation in the short term and new neuron survival and contextual fear conditioning in the long term", Radiation Research, Vol. 188/5, Radiation Research Society, https://doi.org/10.1667/RR14797.1.
Wilkinson, B., Hill, M.A., and Parsons, J.L. (2023), “The Cellular Response to Complex DNA Damage Induced by Ionising Radiation” International Journal of Molecular Sciences Vol. 24/4920, Multidisciplinary Digital Publishing Institute (MDPI) AG, Basel, http://doi.org/10.3390/ijms24054920.
Winocur, G. et al. (2006), "Inhibition of neurogenesis interferes with hippocampus-dependent memory function", Hippocampus, Vol. 16/3, https://doi.org/10.1002/HIPO.20163.
Wong, G., Y. Goldshmit and A. M. Turnley. (2004), "Interferon-γ but not TNFα promotes neuronal differentiation and neurite outgrowth of murine adult neural stem cells", Experimental Neurology, Vol. 187/1, Elsevier, Amsterdam, https://doi.org/10.1016/j.expneurol.2004.01.009.
Wu, M. D. et al. (2012), "Adult murine hippocampal neurogenesis is inhibited by sustained IL-1β and not rescued by voluntary running", Brain, Behavior, and Immunity, Vol. 26/2, Elsevier Inc., Amsterdam, https://doi.org/10.1016/j.bbi.2011.09.012.
Xu, B. et al. (2019), "Oxidation Stress-Mediated MAPK Signaling Pathway Activation Induces Neuronal Loss in the CA1 and CA3 Regions of the Hippocampus of Mice Following Chronic Cold Exposure", Brain Sciences, Vol. 9/10, MDPI, Basel, https://doi.org/10.3390/brainsci9100273.
Yang, H. et al. (2020), “Cytoprotective role of humanin in lens epithelial cell oxidative stress-induced injury”, Molecular medicine reports, Vol. 22/2, Spandidos Publishing Ltd, Athens, https://doi.org/10.3892/mmr.2020.11202.
Zanni, G. et al. (2015), "Lithium increases proliferation of hippocampal neural stem/progenitor cells and rescues irradiation-induced cell cycle arrest in vitro", Oncotarget, Vol. 6/35, https://doi.org/10.18632/oncotarget.5191.
Zhang, L. et al. (2017), "The inhibitory effect of minocycline on radiation-induced neuronal apoptosis via AMPKα1 signaling-mediated autophagy", Scientific Reports, Vol. 7/1, Springer Nature, Berlin, https://doi.org/10.1038/s41598-017-16693-8.
Zhang, Y. et al. (2017), "Kukoamine A Prevents Radiation-Induced Neuroinflammation and Preserves Hippocampal Neurogenesis in Rats by Inhibiting Activation of NF-κB and AP-1", Neurotoxicity Research, Vol. 31/2, https://doi.org/10.1007/s12640-016-9679-4.
Zhang, Q. et al. (2018), "The effect of brain-derived neurotrophic factor on radiation-induced neuron architecture impairment is associated with the NFATc4/3 pathway", Brain Research, Vol. 1681, Elsevier, Amsterdam, https://doi.org/10.1016/j.brainres.2017.12.032.
Zhao, Z.-Y. et al. (2013), "Edaravone Protects HT22 Neurons from H 2 O 2 -induced Apoptosis by Inhibiting the MAPK Signaling Pathway", CNS Neuroscience & Therapeutics, Vol. 19/3, John Wiley & Sons, Hoboken, https://doi.org/10.1111/cns.12044.
Zhao, D. et al. (2017), "Anti-Neuroinflammatory Effects of Fucoxanthin via Inhibition of Akt/NF-κB and MAPKs/AP-1 Pathways and Activation of PKA/CREB Pathway in Lipopolysaccharide-Activated BV-2 Microglial Cells", Neurochemical Research, Vol. 42/2, Springer Nature, Berlin, https://doi.org/10.1007/s11064-016-2123-6.
Zhou, K. et al. (2017), "Radiation induces progenitor cell death, microglia activation, and blood-brain barrier damage in the juvenile rat cerebellum", Scientific Reports, Vol. 7, Springer Nature, London, https://doi.org/10.1038/srep46181.
Zhu, Y. et al. (2012), "APOE genotype alters glial activation and loss of synaptic markers in mice", Glia, Vol. 60/4, John Wiley & Sons, Inc., Hoboken, https://doi.org/10.1002/glia.22289.
Zigman, S. et al. (1995), “Damage to cultured lens epithelial cells of squirrels and rabbits by UV-A (99.9%) plus UV-B (0.1%) radiation and alpha tocopherol protection”, Molecular and cellular biochemistry, Vol. 143, Springer, London, https://doi.org/10.1007/BF00925924.
Zonis, S. et al. (2015), "Chronic intestinal inflammation alters hippocampal neurogenesis", Journal of Neuroinflammation, Vol. 12/1, Springer Nature, Berlin, https://doi.org/10.1186/s12974-015-0281-0.