
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 107
Title
Constitutive androstane receptor activation leading to hepatocellular adenomas and carcinomas in the mouse and the rat
Short name
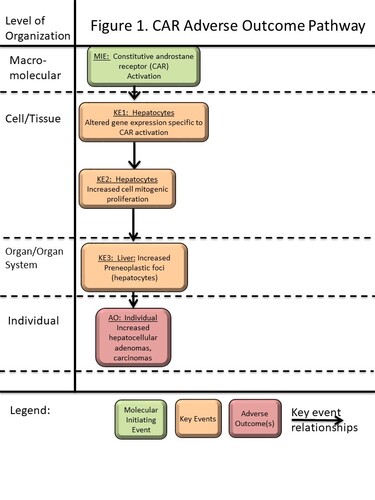
Graphical Representation
Point of Contact
Contributors
- Richard C. Peffer, Ph.D., DABT
- Katie Bailey
- Kristin Lichti-Kaiser
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.17 | Under Review |
This AOP was last modified on April 29, 2023 16:02
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Activation, Constitutive androstane receptor | June 28, 2018 10:28 |
| Increase, cell proliferation (hepatocytes) | January 06, 2021 16:21 |
| Increase, hepatocellular adenomas and carcinomas | December 26, 2020 10:09 |
| Increase, Preneoplastic foci (hepatocytes) | September 16, 2017 10:16 |
| Altered gene expression specific to CAR activation, Hepatocytes | December 19, 2018 15:36 |
| Activation, Constitutive androstane receptor leads to Altered expression of hepatic CAR-dependent genes | December 21, 2018 08:51 |
| Altered expression of hepatic CAR-dependent genes leads to Increase, cell proliferation (hepatocytes) | December 21, 2018 08:42 |
| Increase, cell proliferation (hepatocytes) leads to Increase, Preneoplastic foci (hepatocytes) | February 14, 2017 17:05 |
| Increase, Preneoplastic foci (hepatocytes) leads to Increase, hepatocellular adenomas and carcinomas | February 15, 2017 10:20 |
| Phenobarbital | November 29, 2016 18:42 |
| metofluthrin | December 06, 2016 12:08 |
| TCPOBOP | February 14, 2017 14:22 |
Abstract
The constitutive androstane receptor (CAR; NR1I3) is a mammalian nuclear receptor that is involved in the regulation of various cellular processes following its activation by xenobiotics or endogenous ligands. Activation of CAR in the liver of rats and mice leads to altered gene expression, including genes related to Phase 1 and Phase 2 metabolism, transporters, gluconeogenesis, lipid homeostasis, cell cycle control and apoptosis regulation. This AOP describes the sequence of key events (KEs) that occur in rats and mice following chronic exposure to CAR activators, which leads from activation of CAR to an increased incidence of hepatocellular adenomas and carcinomas. The molecular initiating event (MIE) is activation of the CAR nuclear receptor, either by direct binding of a ligand or by an indirect mechanism (e.g. as with phenobarbital), both of which allow the CAR protein to translocate to the nucleus and alter expression of CAR target genes. In rats and mice, CAR activation alters the expression of certain genes related to cell cycle control, producing changes resulting in a pro-proliferative and anti-apoptotic environment. These gene expression changes (KE1) lead to an increase in mitogenic cell proliferation (KE2). In this proliferative environment, a higher number of spontaneously mutated hepatocytes can form. With longer time intervals, the mutated hepatocytes clonally expand into pre-neoplastic altered foci (KE2). Eventually, under continued CAR activation, the pre-neoplastic foci expand to form hepatocellular adenomas and carcinomas, the adverse outcome (AO). Based on a wide dataset of testing with CAR activators, this AOP is considered to only be operative in mice and rats, and not in other mammalian species including humans. Good dose concordance between the early KEs, associated biological markers of the KEs and the AO have been demonstrated with example molecules. A potential application of this AOP is to guide future risk assessments, where dose-response values for critical early key events, e.g. NOAEL (no observed adverse effect level) or BMDL (Benchmark Dose Lower Limit) values, may be useful endpoints. Another potential application of this AOP is to highlight how methods for assessment of the MIE and/or the early KEs can be reliably used to demonstrate that a CAR mode of action is operative for a particular molecule, avoiding large scale use of animal testing to demonstrate every KE in the proposed pathway.
AOP Development Strategy
Context
In chronic carcinogenicity studies conducted in rats and mice as part of the registration process for drugs, agrochemicals and other xenobiotics, a frequent finding after high dose treatments is hepatocellular adenomas and carcinomas in the liver (Cohen, 2010; Gold et al., 2005). An AOP via activation of the CAR nuclear receptor is one well-understood mechanism by which these tumors can occur (Elcombe et al., 2014). An overall framework for describing a mode of action (Anderson et al., 2014; Meek et al., 2014) has established that a mode of action (or an AOP) can be described based on a series of Key Events (KEs), which are causal and required precursor steps to the AO. Studies comparing the postulated KEs in various species (e.g. rat, mouse, hamster, guinea pig, non-human primate) as well as in human hepatocytes or humans on chronic treatment with CAR activating drugs, have indicated large species differences in the susceptibility to 1) certain key events including KE2 (cell proliferation) and 2) the ability of CAR activators to produce liver tumors. The main purpose of this AOP is to outline the measurable key events for an AOP via CAR activation that leads to liver tumors in rats and mice. However, by summarizing experimental results across a wider range of mammalian species, the AOP outlines species differences relating to the KEs and the AO. By this approach, it is intended that the AOP can help the wider scientific and regulatory community to recognize the measurable KEs that would indicate a xenobiotic produces liver effects via this AOP, and the methods typically employed to demonstrate the thresholds in dose-response, below which no KEs and no tumors have been shown to occur in rats and/or mice with model CAR activators.
Published summaries regarding the CAR mode of action (Elcombe et al., 2014; Peffer et al., 2018a; Peffer et al., 2018b; Lake, 2018) have identified a similar set of KEs, and they also identify associatve events, which are biological processes that are not necessary for the AOP, but can often be used as surrogate markers for a particular KE (Anderson et al., 2014). The associative events that follow CAR activation include: 1) Increased CYP2B, CYP3A enzyme activity and/or protein in hepatocytes; 2) increased hepatocellular hypertrophy; and 3) increased liver weight. Based on the guidance for writing AOPs, this current AOP on CAR activation leading to rodent hepatocellular tumors does not specifically include associative events within the graphical illustration (Figure 1), but instead identifies them within the context of "How the KE is measured" within each individual KE or KER. These associative events are readily measured in the course of many toxicology studies, and provide useful markers for the KEs that are part of the CAR AOP.
CAR (NR1I3) and PXR (pregnane X receptor; NR1I2)) are often cited together regarding potential mode(s) of action (MoA) for a specific chemical agent, because they are from the same family of nuclear receptors and exhibit extensive cross-talk in terms of the set of genes and response elements that they can activate (Stanley et al., 2006). In the published proceedings of a nuclear receptor workshop on the CAR / PXR MoA (Andersen et al., 2014; Elcombe et al., 2014), the authors could not identify a suitable nongenotoxic PXR activator for which carcinogenicity data were available, and hence a MoA was not developed for liver tumor formation by PXR activators. Some agents can activate both CAR and PXR in a particular species (Elcombe et al., 2014). In fact, PXR is activated by a large array of chemical substances, far more than those that activate CAR (Martin et al., 2010; Timsit and Negishi, 2007; Willson and Kliewer, 2002). PXR has been shown to increase liver weight after activation by a number of substrates, but suspected activators of PXR such as pregnenolone-16α-carbonitrile or dexamethasone have not consistently shown increases in assays for cell proliferation in rats and mice (Lake et al., 1998; Shizu et al., 2013; Thatcher and Caldwell, 1994). PXR activation is classically considered to selectively induce increased expression of CYP3A isoforms, with lesser induction of CYP2B isoforms, but again, cross-talk between PXR and CAR receptors upon activation of either nuclear receptor can be part of the altered expression of these Cyp isoforms in vivo. Given the lack of actual tumorigenic key events due to PXR activators alone, the rest of this current AOP will focus on the CAR mode of action by itself.
The AOP submitted here is limited to a definitive set of readily measurable endpoints that encompass the KEs that are critical to the progression from CAR activation to liver tumor formation, and differentiate an agent that works via CAR activation from one that operates by alternative modes of action. Therefore, not all of the possible biochemical steps discussed by Elcombe et al. (2014) or proposed in other publications are shown (Phillips and Goodman, 2008; Moennikes et al., 2000, Huang et al., 2005; Brauening et al., 2016). While suppression of apoptosis, changes in DNA methylation status or inhibition of gap junction intercellular communication (GJIC) have been shown to occur in rodent liver after treatment with CAR activators such as phenobarbital (Huang et al., 2005; Klaunig et al., 1990; Moennikes et al., 2000; Phillips and Goodman, 2008), these were viewed by Elcombe et al. (2014) as associative events, since clear demonstrations of essentiality and/or association with CAR activation have not been established. More importantly regarding the AOP for CAR activation, these possible associative events require specialized techniques to demonstrate them, such as micro-injection of individual hepatocytes with dye in the case of GJIC (Klaunig et al., 1990), or examination of apoptosis within altered foci at later time points in longer-term studies (Kolaja et al., 1996b). Considering the specialized methods needed and the fact that these associative events are not considered essential to demonstrating the overall AOP, they are not included in the set of KEs described in this AOP (Figure 1 and Table 1). As with all of the AOPs on the AOPwiki site, this AOP for rodent liver tumors via CAR activation may be modified in the future to include additional or different KEs and experimental markers of those KEs as the understanding of these toxicological processes evolves.
Table 1 (see below)
In Table 1, the typical data that is generated to demonstrate a molecule produces a liver tumor response in mice or rats via CAR activation AOP is described. However, this should not be viewed as a set of required studies, as other techniques are available that can lead to the same endpoint of demonstrating the KEs in a CAR AOP, or markers for them. A more thorough description of current and emerging methods for identifying a CAR mode of action with a particular molecule is available in Peffer et al., 2018b.
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 715 | Activation, Constitutive androstane receptor | Activation, Constitutive androstane receptor |
| KE | 1214 | Altered gene expression specific to CAR activation, Hepatocytes | Altered expression of hepatic CAR-dependent genes |
| KE | 716 | Increase, cell proliferation (hepatocytes) | Increase, cell proliferation (hepatocytes) |
| KE | 774 | Increase, Preneoplastic foci (hepatocytes) | Increase, Preneoplastic foci (hepatocytes) |
| AO | 719 | Increase, hepatocellular adenomas and carcinomas | Increase, hepatocellular adenomas and carcinomas |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Activation, Constitutive androstane receptor leads to Altered expression of hepatic CAR-dependent genes | adjacent | High | Moderate |
| Altered expression of hepatic CAR-dependent genes leads to Increase, cell proliferation (hepatocytes) | adjacent | High | Moderate |
| Increase, cell proliferation (hepatocytes) leads to Increase, Preneoplastic foci (hepatocytes) | adjacent | High | Moderate |
| Increase, Preneoplastic foci (hepatocytes) leads to Increase, hepatocellular adenomas and carcinomas | adjacent | High | Not Specified |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| All life stages | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
| Female | High |
Overall Assessment of the AOP

1. Concordance of Dose-Response Relationships
Exposure-response relationships for nearly all of the key events and associative events have been established in vitro and/or in vivo in both mice and rats. A large and growing number of xenobiotics have been shown to produce rodent liver tumors via the CAR activation AOP (Deguchi et al., 2009; Geter et al., 2014; Huang et al., 2005; LeBaron et al., 2013; Peffer et al., 2007). For ease of presentation, data are provided for three prototypical CAR activators (phenobarbital, TCPOBOP and metofluthrin). There are several variables that can impact dose-response relationships, including the CAR activating compound and the strain, sex, and species of the test system. Therefore, these variables need to be taken into consideration in order to evaluate the dose–response and time-response relationships in the section below. With this goal in mind, one exemplar species/strain/sex for each of these three model CAR activators are summarized in the dose-response and time concordance tables (Tables 2-5) that are illustrative for this AOP.
Phenobarbital causes an increase in the incidence of liver foci/tumors via the CAR activation AOP in male and female mice, and to a lesser extent in the rat (reviewed in Elcombe et al., 2014). For illustration, Table 2 shows the dose-response and time concordance of liver tumor incidence with phenobarbital exposure in male CD-1 mice. In this sex/strain, phenobarbital treatment for 2 years in the diet produced increased incidence of tumors at 75 and 150 mg/kg/day and no increase in tumor incidence at 10 mg/kg/day (Whysner et al., 1996). As shown in Table 2, a very strong dose concordance was observed for each KE or AE (as markers for in vivo CAR activation), with all of these events showing responses at the tumorigenic dose levels of 75 – 150 mg/kg/day. Certain early KEs or AEs produced effects at non-tumorigenic levels of 1.5 – 15 mg/kg/day, with increased PROD activity, a marker of CYP2B enzyme activity, being the most sensitive overall endpoint in male mice (Geter et al., 2014).
Table 3 provides dose-response data for administration of phenobarbital in male mice of the C57BL/10J strain. A 2-year (99 week) study in this strain of mice produced a clear increase in the incidence of liver adenomas and carcinomas at 1000 ppm in the diet, with no increases in tumor incidence at 200 ppm (Jones et al., 2009). As described in the same reference, preliminary studies were conducted in male and female C57BL/10J mice fed diets containing 0 (control), 100, 200, 400, 700 and 1000 ppm phenobarbital for periods of 3, 8, 15 and 29 days (Jones et al., 2009),. As shown in Table 3, The associative events of hepatocyte hypertrophy and increased relative liver weight were increased at dose levels > 200 ppm after 29 days The key event of cell proliferation (KE2) as indicated by BrdU labeling index was increased in the livers of animals exposed to 700 and 1000 ppm, but not at lower dose levels. In addition, the increase in cell proliferation was transient, with no effects observed after 29 days. Finally, an increase in eosinophilic and clear cell foci was observed after 99 weeks at the tumorigenic dose level (1000 ppm), but not at the non-tumorigenic dose level (200 ppm) (Jones et al., 2009). Overall, short-term and long-term studies in C57BL/10J mice showed good dose concordance between the key events and the tumorigenic dose level of 1000 ppm (113 mg/kg/day).
Another short-term study was conducted with phenobarbital in C57BL/6J mice, which are genetically similar to C57BL/10J mice, and in CAR/PXR-null mice on this same background (Ross et al., 2010). In this study, mice received 4 daily intraperitoneal (ip) doses of 80 mg/kg/day phenobarbital. As shown in Table 3, phenobarbital produced a large induction of markers of CAR activation (MIE), including AE1 (increased pentoxyresorufin O-depentylase (PROD) activity) and KE1 (increased Cyp2b10 mRNA). Other gene expression changes associated with CAR activation (KE1) were also impacted, including increased Ki67 expression and decreased Tsc22 expression, which are markers of a pro-proliferative response. These changes in PROD activity and mRNA expression levels were unaffected in the CAR/PXR-null mice, indicating they were a result of CAR activation (Ross et al., 2010).
TCPOBOP (1,4-bis-[2-(3,5-dichloropyridyloxy)]benzene, 3,3′,5,5′-tetrachloro-1,4-bis(pyridyloxy)benzene) is a very potent direct CAR activator in the mouse (Omiecinski et al., 2011) , and it has been shown to produce tumors in male and female mice of various strains at an experimental dose level of 3 mg/kg/day (ip or oral gavage). For illustration, Table 4 shows time concordance of TCPOBOP effects in male C57BL/6 mice at 3 mg/kg/day, given for 1 day, 3 days, 28 days, or 30 weeks (with sacrifice after 60 weeks), using various dosing methods and frequencies. TCPOBOP is a lipophilic substance that is excreted slowly in mice and is a very potent mouse CAR activator, so dosing regimes often have involved intermittent dosing such as once every 14 days (Huang et al., 2005). Consistent effects on all AEs and KEs were observed across the multiple studies summarized in Table 4 at this dose level, including an increase in the level of Cyp2b10 mRNA (KE1), altered CAR-mediated gene expression marking a pro-proliferative response (e.g. increased expression of Cdc20 and Gadd45b, KE1), increased cell proliferation, increased incidence of altered foci and eventually the appearance of hepatocellular tumors. The potent response of the mouse liver to TCPOBOP also afforded a useful experimental tool to investigate the CAR-mediated nature of the effects, via studies in CAR knockout mice and other similar transgenic models. Lack of effects on the early KEs and AEs (including the gene expression changes), and a lack of any increase in liver tumor incidence in CAR null mice exposed to TCPOBOP demonstrated that these processes and the eventual development of tumors were dependent on CAR activation (Huang et al., 2005; Yamamoto, et al., 2004).
Metofluthrin has produced an increase in liver tumor incidence via the CAR activation AOP in male Wistar rats (900 and 1800 ppm) and female Wistar rats (1800 ppm), but it did not produce liver tumors in CD-1 mice (Yamada et al., 2009). For illustration, Table 5 shows the dose-response and time concordance of liver tumor incidence with metofluthrin in male Wistar rats. Investigative studies were conducted at dose levels ranging from 200 ppm (the tumor NOAEL in male rats) up to 3600 ppm (a dose considered in excess of the Maximum Tolerated Dose [MTD]) (Deguchi et al., 2009; Hirose et al., 2009). The key events of Cyp2b1/2 and/or Cyp3a1 mRNA levels (as markers for CAR activation), increased cell proliferation and increased altered foci were each observed at the tumorigenic dose levels of 900 ppm and above. Associative events of increased PROD activity, increased CYP2B protein levels, hepatocellular hypertrophy and increased liver weight were increased at tumorigenic dose levels, but they were unaffected at the tumor NOAEL (200 ppm). In isolated rat hepatocyte cultures treated with 50 µM metofluthrin, co-administration of siRNA for CAR (a gene silencing technique) caused a knockdown of Car mRNA expression and resulting suppression of the response to metofluthrin in terms of Cyp2b1 mRNA and Car mRNA levels (Deguchi et al., 2009).
2. Temporal Concordance Among the Key Events and Adverse Outcome
As shown for the 3 example molecules (Tables 2-5), empirical evidence shows good temporal concordance between the MIE (CAR activation detected within a few days of starting treatment and continuing throughout), intermediate KEs and AEs (days to weeks), leading eventually (months) to later KEs of increased incidence of altered foci followed by tumors.
3. Strength, Consistency and Specificity of the Association of Adverse Effect and Initiating Event
The scientific evidence linking the MIE (CAR activation) and the AO (liver tumors in rodents) have been presented for multiple compounds, and the same sequence of key events was observed for each compound. Blocking the initial MIE (via testing in transgenic mice lacking the CAR nuclear receptor) was able to block all of the subsequent key events, including CAR-specific gene expression, cell proliferation and ultimately liver tumor formation, thus demonstrating high specificity of this association.
4. Biological Plausibility, Coherence and Consistency of the Experimental Evidence
There is a high biological plausibility of the proposed AOP, as it is consistent with known biology and constitutes a plausible sequence of events for non-genotoxic liver carcinogens. Also, the demonstrated key events were consistent across 3 different compounds and between mice and rats in their responses to a demonstrated CAR activator for that species.
5. Alternative Mechanisms
For the data-rich molecules that were chosen such as phenobarbital and TCPOBOP, whole mouse microarray experiments are available (Currie et al., 2014; Geter et al., 2014; Nesnow et al., 2009; Oshida et al., 2015a; Tojima et al., 2012) that allow an examination for markers of certain alternative liver modes of action for liver tumorigenesis that have been characterized in rodent studies (Cohen, 2010). No evidence of PPARα activation (as marked by Cyp4a induction) was observed, nor was there evidence of estrogenic effects. AhR activation (as marked by high levels of CYP1A / CYP1B induction) was shown not be operative; a minor increase in certain isoforms of CYP1A or CYP1B has been shown to occur upon treatment with CAR activators (Oshida et al., 2015a; Oshida et al., 2015b), but the large induction characteristic of AhR activators is not observed. For each of the three molecules, a substantial dataset indicates that these molecules are not genotoxic, do not produce immediate cytotoxicity and regenerative hyperplasia in the liver, and do not produce histological evidence of liver changes via iron deposition or an infectious process within the liver (Elcombe et al., 2014; Huang et al., 2005; Yamada et al., 2009). Thus, no alternative mechanisms appear to be operative, and these three example molecules can be concluded to work via an AOP initiated by CAR activation.
6. Uncertainties, Inconsistencies and Data Gaps
Across the full database for these three example molecules, very few uncertainties or data gaps exist; any data that are missing in one species/strain/test compound are typically present in the others. For example, phenobarbital treatment of male CD-1 mice has been reported in a review article to cause an increase in adenomas and carcinomas of the liver at 75 and 150 mg/kg/day, with a tumor NOAEL at 10 mg/kg/day (Whysner et al., 1996), but the actual tumor incidence data for this study have not been published. Accordingly, no data for the KE3 of increased altered foci are available in CD-1 mice treated with phenobarbital. However, tumorigenic responses in various strains of mice after phenobarbital treatment have been described in other studies (reviewed in Elcombe et al., 2014), and studies in male C57BL/10J mice have shown a clear increase in both altered foci and liver tumors at 1000 ppm and a NOAEL at 200 ppm (Table 3; Jones et al., 2009).
Domain of Applicability
Species Differences
Studies in various species, or in isolated hepatocytes from various mammalian species including humans, have demonstrated that CAR activators such as phenobarbital, TCPOBOP or metofluthrin produce a cell proliferation response that is seen in mice or rats, but not in hamsters, guinea pigs, or humans (Hirose et al., 2009; James and Roberts, 1996; Parzefall et al., 1991; Yamada et al., 2009; Yamada et al., 2015). In accord with the lack of response for this key event in hamsters, phenobarbital does not produce liver tumors in long term initiation-promotion studies in hamsters (Diwan et al., 1986; Stenback et al., 1986; Tanaka et al., 1987; Elcombe et al., 2014). Consistent with the lack of effects on proliferation and on tumor development, Diwan et al. (1986) also reported that phenobarbital treatment at 500 ppm in the drinking water did not produce any increases in preneoplastic foci of cellular alteration in hamsters compared to groups that received an initiator alone. Substances such as phenobarbital have been shown to activate the CAR receptor in mice, rats, hamsters, non-human primates and humans, resulting in altered gene expression for metabolizing enzymes (a subpart of KE1) and increased CYP2B enzyme activity (AE1), hepatocellular hypertrophy (AE2) and increased relative liver weight (AE3) (summarized in Elcombe et al., 2014). However, while phenobarbital and other CAR activators can produce non-adverse liver changes via these associative events in multiple species, they only produce the KEs of increased cell proliferation and increased foci of alteration in rats and mice. Consistent with this pattern, CAR activators do not produce a cell proliferation response in human primary hepatocyte cultures in vitro (Parzefall et al., 1991; Hirose et al., 2009; Yamada et al., 2015)
More recently, humanized liver chimeric mouse models have become commercially available and have been used to evaluate the in vivo response of human liver tissue within a chimeric mouse where the mouse hepatocytes were selectively destroyed, and transplanted human hepatocytes were allowed to repopulate and replace the mouse hepatocytes (Yamada et al., 2014; Scheer and Wilson, 2016). The specific chimeric model in these studies was uPA/SCID mice (PhoenixBio Co., Ltd.) with hepatocytes from one human donor in a study with phenobarbital (Yamada et al., 2014). Replacement of mouse hepatocytes with human hepatocytes in these experiments was in the range of 73 - 90%. Treatment of these mice was commenced at more than 70 days after transplantation. The chimeric mice were supplemented with human growth hormone (by mini-pump), because the human GH receptor is unresponsive to mouse GH. Human albumin levels in blood were monitored as a marker for the degree of replacement of mouse with human hepatocytes. Sodium phenobarbital was administered in the diet for 7 days and compared to untreated control animals. BrdU was administered to all animals via mini-pump for 7 days prior to termination. In livers from the chimeric mice, regions of human hepatocytes were visualized by light microscopy and subsets of the human liver regions were randomly selected for quantification of the BrdU labelling index, as a measure of cell proliferation. A similar process was followed for wild-type SCID mouse livers to quantify BrdU labelling index in that strain of mouse following treatment with phenobarbital. Additional groups of mice were treated with human epidermal growth factor (hEGF) (150 µg/kg, four times per day, i.p., for 2 days) to assess the responsiveness of the human hepatocytes to a known mitogen (Yamada et al., 2014).
Results of experiments in this chimeric mouse system and a corresponding wild-type strain of mice (SCID) with phenobarbital (Yamada et al., 2014) are shown in Table 6. The results of these experiments in the humanized liver chimeric mouse model confirm in vivo what has been observed across multiple experiments using primary mouse and human hepatocyte cultures in vitro. Treatment with 500, 1000 and 1500 ppm phenobarbital caused increased CYP2B mRNA levels with a smaller increase in CYP3A mRNA levels (KE1), PROD activity (AE1), hepatocyte hypertrophy (AE2) and a slight increase in liver weight (AE3) in the chimeric human livers, but it caused no effects on cell proliferation (KE2) based on the BrdU labelling index. In contrast, the wild-type SCID mice showed a 4.6-fold increase in BrdU labelling index after 7 days in addition to the increases in CYP2B mRNA (with a smaller increase in CYP3A mRNA), PROD activity, hepatocellular hypertrophy and liver weight. Consistent with the lack of a proliferative response, the chimeric livers showed no increases in GADD45B mRNA, a pro-proliferative gene, whereas the wild-type SCID mice showed a 5.2-fold increase in expression. Ki67 mRNA was unaffected in both strains of mice, which may be a reflection of the later sampling time (7 days) compared to results in other phenobarbital experiments (Table 2). Dose levels up to 1500 ppm in the humanized liver chimeric mice met or exceed a maximum tolerated dose (i.e. some deaths occurred), and markedly higher blood concentrations of phenobarbital were observed in these mice compared to the wild-type SCID and CD-1 mice, which likely reflects less induction of metabolism in the humanized livers.
Table 6 - Phenobarbital Effects in Humanized Liver Chimeric Mouse Model
The same study (Yamada et al., 2014) also investigated these endpoints in male CD-1 mice and male Wistar rats at dose level (500 – 2500 ppm) that produced similar plasma levels of NaPB as the 500 – 1000 ppm chimeric mice and 1500 ppm SCID mice (data not shown). The CD-1 mice and Wistar rats had effects similar to the wild-type SCID mice (Table 6). The study also compared the global gene expression responses following NaPB treatment via microarrays for chimeric human livers, and for livers from CD-1 mice and Wistar rats. There was very little overlap in the differentially expressed genes when the chimeric humanized livers were compared to CD-1 mice or Wistar rats, and the few overlapping genes were largely related to metabolism.
Concurrent experiments in which hEGF was administered to humanized liver chimeric mice confirmed that the human hepatocytes were responsive to a mitogenic stimulus, with BrdU labelling indices of approximately 4-fold of control values and Ki67 mRNA expression of 6-fold of the control value. Human hepatocyte cultures in vitro that were derived from livers of the chimeric mice also displayed an increase in BrdU labelling index by 1.5- to 2.3-fold. In addition, partial hepatectomy experiments in the humanized chimeric mice have shown that the human livers respond to this procedure with an increase in proliferation (Yamada et al., 2014). Overall, the humanized liver chimeric mouse model has been shown to be a valuable research tool to confirm that human liver responds differently from mouse or rat liver to CAR activators. This humanized liver chimeric mouse model and other similar models are reviewed in Scheer and Wilson, 2016; and in Peffer et al., 2017 (in press).
Multiple epidemiological studies with phenobarbital and other anticonvulsant drugs have been performed (Friedman et al., 2009; IARC, 2001; Olsen et al., 1989; Olsen et al., 1995; Selby et al., 1989; White et al., 1979; Whysner et al., 1996) and results of these studies have been reviewed by La Vecchia and Negri (2014) and Elcombe et al. (2014). In only one study, patients treated with various anticonvulsant drugs showed a possible increase in liver tumors (Lamminpaa et al., 2002), although an independent review indicated that other factors such as alcohol and smoking may have contributed to the increase in liver tumors and that there was no indication of an excess risk attributable to phenobarbital use (La Vecchia and Negri, 2014). In contrast, multiple earlier epidemiological studies in patients that received phenobarbital have demonstrated that phenobarbital did not increase the incidence of liver tumors or of any other tumor type in humans (Friedman et al., 2009; Olsen et al., 1989, 1995; Selby et al., 1989). In their review of the full range of published studies up through October 2012, La Vecchia and Negri (2014) concluded that there was no evidence for a specific role of phenobarbital in human liver cancer risk. In the studies that showed no evidence of increased liver tumor risk, subjects received phenobarbital over many years at doses that produced plasma concentrations similar to those that are carcinogenic in rodents. For example, phenobarbital administered at 500 ppm in drinking water achieved serum concentrations of 5–29 µg/ml in mice (three different strains), and 20 – 33 µg/mL in Wistar male and female rats. In human patients receiving phenobarbital at therapeutic doses of 3–6 mg/kg/day, serum concentrations ranged from 10–25 µg/ml, which reflects the recommended therapeutic range of this anticonvulsant (Monro 1993).
In summary, human epidemiological studies support a conclusion that the AOP for CAR-mediated liver tumors in mice and rats following phenobarbital treatment is not relevant to humans. The lack of effects on the causal KE of cell proliferation, and on certain gene expression changes (e.g. no altered expression of CAR-modulated genes related to cell cycle control) in human hepatocytes and in humanized liver chimeric mice provide a mechanistic support for this conclusion.
Age and Life Stage Differences
In rats and mice, there is evidence that the long-term hepatic effects of phenobarbital or other CAR activators are greater in old animals compared to weanlings. For male F344 rats or male C3H/He mice treated with 500 ppm phenobarbital in drinking water, increased incidences of foci and hepatocellular tumors were observed if the study was started using older animals (2.4-year old or 1-year old, respectively) compared to younger animals (Ward et al., 1983; Ward et al., 1988). These data suggest that tumors in rodents with phenobarbital result from promotion of spontaneous preneoplastic lesions, which are more numerous in the livers of older rats and mice (Schulte-Hermann et al., 1983). In contrast, there are no known associations of differing susceptibility to liver effects from CAR activators in different life stages for humans.
Essentiality of the Key Events
|
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
|
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from experimental studies illustrating essentiality for at least one of the important KEs. |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE. |
No or contradictory experimental evidence of the essentiality of any of the KEs. |
|
|
MIE: CAR Activation |
Strong |
When activation of CAR was prevented via testing in CAR knockout mice, virtually all downstream key events were prevented, including tumors (Huang et al., 2005; Peffer et al., 2007; Tamura et al., 2015; Tamura et al., 2013; Tojima et al., 2012; Wei et al., 2000; Yamamoto et al., 2004). In Wistar rats administered metofluthrin, suppression of CAR synthesis via siRNA was shown to suppress the expression of Cyp2b1 mRNA, confirming that this associative event (AE1) was CAR-dependent in rats (Deguchi et al., 2009). |
||
|
KE1: Altered gene expression specific to CAR activation |
Strong |
When activation of CAR was prevented via testing in CAR knockout mice, differential expression of critical genes and pathways related to a pro-proliferative response and marker genes of CAR activation (e.g. Cyp2b10 in mice; Cyp2b1/2 in rats) was blocked (Oshida et al., 2015a; Peffer et al., 2007; Tojima et al., 2012). In addition, downstream key events or associative events dependent on altered gene expression were also blocked in the CAR null mice, including CYP2B enzyme activity (AE1), hepatocellular hypertrophy and increased liver weights (AE2, AE3), increased cell proliferation (KE2) and the long-term histopathology changes (KE3; AO) (Huang et al., 2005; Peffer et al., 2007; Tamura et al., 2015; Tamura et al., 2013; Wei et al., 2000; Yamamoto et al., 2004). |
||
|
KE2: Mitogenic cell proliferation (hepatocytes), Increase |
Strong |
When activation of CAR was prevented via testing in CAR knockout mice, cell proliferation in the liver was prevented at the tumorigenic dose levels (Huang et al., 2005; Peffer et al., 2007; Ross et al., 2010; Tamura et al., 2013). |
||
|
KE3: Preneoplastic foci (hepatocytes), Increase |
Strong |
When activation of CAR was prevented via testing in CAR knockout mice, foci of cellular alteration in the liver was prevented in an initiation-promotion model using the CAR activators cyproconazole and fluconazole (Tamura et al., 2015). Also, the incidence of adenomas and carcinomas was similarly decreased (Tamura et al., 2015). With phenobarbital treatment of C3H background mice, absence of the CAR receptor in CAR null mice blocked both the increase in eosinophilic foci and the increase in liver adenomas and carcinomas (Yamamoto et al., 2004). Thus, there is strong evidence for the essentiality of KE3 to the overall progression to liver tumors with CAR activators, and this essentiality is further confirmed by normal biology of the rodent liver (Goldsworthy and Fransson-Steen, 2002; Haschek and Rousseaux, 1998). |
||
|
AO: Adenomas/carcinomas (hepatocellular), Increase |
Strong |
When activation of CAR was prevented via testing in CAR knockout mice, virtually all downstream key events were prevented (Huang et al., 2005; Peffer et al., 2007; Ross et al., 2010; Tamura et al., 2015; Tamura et al., 2013; Tojima et al., 2012; Wei et al., 2000; Yamamoto et al., 2004). In addition, testing in CAR knockout mice prevented the formation of liver tumors in initiation-promotion models or in studies where CAR activator alone was administered (Huang et al., 2005; Tamura et al., 2015; Wei et al., 2000; Yamamoto et al., 2004). |
||
Evidence Assessment
|
Support for Biological Plausibility of KERs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
Extensive understanding of the KER based on previous documentation and broad acceptance. |
KER is plausible based on analogy to accepted biological relationships, but scientific understanding is incomplete. |
Empirical support for association between KEs, but the structural or functional relationship between them is not understood. |
|
|
MIE => KE1 |
Strong |
Activation of the CAR receptor causes changes in expression of CAR-responsive genes such as Cyp2b10, Gadd45b, Cdc20, and Ki67, and overall genomic pathway changes associated with increased progression through the cell cycle (leading to later KE2) (Geter et al., 2014; Oshida et al., 2015a; Tojima et al., 2012). The KER is highly plausible, since specific CAR recognition elements for genes in these pathways have been identified previously (Stanley et al., 2006; Baes et al., 1994; Sueyoshi and Negishi, 2001), and absence of the CAR receptor in CAR-null animals blocks these KE1 gene changes. |
||
|
KE1 => KE2 |
Strong |
The CAR-mediated changes in expression of pro-proliferative genes such as Gadd45b, Ki67 and Cdc20 have been demonstrated to occur with multiple CAR activators (Oshida et al., 2015a; Ozawa et al., 2011; Tojima et al., 2012). It is highly plausible that these gene expression changes then lead to increased cell proliferation signals in hepatocytes, as genes in the Gadd45 family are known to interact with cyclins, cyclin-dependent kinase inhibitors and p53 to alter progression through the cell cycle (Liebermann and Hoffman, 2008). For most CAR activators, the DNA labeling index as a marker of cell proliferation is maximal after 1-7 days of treatment, and then diminishes back to control levels by approximately 28 days. Although hepatocyte-labelling index values (% BrdU stained/total hepatocytes per high-powered field) return to control levels with sustained CAR activation, the number of proliferating cells in treated animals is still enhanced due to the increase in the total number of hepatocytes per animal (Lake, 2009; Cohen, 2010). In addition, cell proliferation within altered foci in initiation-promotion studies in mice or in F344 rats has been shown to be enhanced by CAR activator treatment at 20 to 76 weeks after initiation in a dose-responsive manner (Bursch et al., 2005; Klaunig, 1993; Kolaja et al., 1996b). The increased proliferation due to CAR activator treatment tends to occur to a greater extent within eosinophilic foci, and has been shown to be greater in tumor-prone strains of mice (C3H > B6C3F1) than in the comparatively tumor-resistant C57BL/6 strain of mice (Bursch et al., 2005; Pereira, 1993). |
||
|
KE2 => KE3 |
Strong |
The increased cell replication rate in the liver due to CAR activation (i.e. via mitogenic signaling) is similar to other well-understood modes of action where an increase in cell proliferation leads to an eventual increase in pre-neoplastic foci, such as PPARalpha activating ligands or partial hepatectomy leading to regenerative proliferation of the liver. Similarly with AhR activating ligands, progressive liver damage and regenerative proliferation of hepatocytes leads to an increase in pre-neoplastic foci via clonal expansion of transformed hepatocytes (Becker et al., 2015; Cohen, 2010; Corton et al., 2014). In a normal liver, expansion of hepatocyte numbers via proliferation is in a constant balance with controlled cell death of damaged hepatocytes via apoptosis, to keep an appropriate critical mass of functional liver cells that can be maintained by the oxygen and nutrient supplies within the liver lobule (Goldsworthy and Fransson-Steen, 2002). In mice lacking the CAR receptor, including initiation-promotion assays, the upstream key events (e.g. CAR activation, gene expression changes, cell proliferation), the associative events (e.g. increased Cyp2b/Cyp3a activity, hepatocellular hypertrophy, increased liver weight) and the downstream events (e.g. pre-neoplastic foci) are all blocked, providing strong support for the biological plausibility of this Key Event Relationship (Huang et al., 2005; Tamura et al., 2015; Tamura et al., 2013; Yamamoto, Y. et al., 2004). |
||
|
KE3 => AO: |
Strong |
The development of liver tumors in rodents, whether spontaneously or induced by a non-genotoxic carcinogen, has consistently included the development of altered foci as precursor step to hepatocellular adenomas and carcinomas (Goldsworthy and Fransson-Steen, 2002; Haschek and Rousseaux, 1998). These foci are considered pre-neoplastic lesions, and their ability to progress to form adenomas and/or carcinomas in rodents has been previously recognized. In the case of CAR activators, an increased incidence of pre-neoplastic foci has been consistently shown to precede tumor development, and there is a high biological plausibility for this Key Event Relationship (Elcombe et al., 2014; Goldsworthy and Fransson-Steen, 2002). |
||
Known Modulating Factors
Quantitative Understanding
|
Empirical Support for KERs |
Defining Question |
High (Strong) |
Moderate |
Low (Weak) |
|
Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? Does KEup occur at lower doses, earlier time points, and higher in incidence than KEdown ? Inconsistencies? |
Multiple studies showing dependent change in both events following exposure to a wide range of specific stressors. No or few critical data gaps or conflicting data. |
Demonstrated dependent change in both events following exposure to a small number of stressors. Some inconsistencies with expected pattern that can be explained by various factors. |
Limited or no studies reporting dependent change in both events following exposure to a specific stressor; and/or significant inconsistencies in empirical support across taxa and species |
|
|
MIE => KE1: |
Strong |
Strong support for this linkage comes from in vivo and in vitro studies with model CAR activators, and via the absence of the same effects including specific gene expression changes in CAR knockout mice lacking the CAR receptor. In a study with TCPOBOP (a direct activator of mouse CAR), these CAR-dependent changes included Gadd45b (14-fold increase), Cdk1 (11-fold increase), Cdc20 (37-fold increase) and additional cytokines, and these genes were unaffected in CAR knockout mice (Tojima et al., 2012). Gene pathways (by Ingenuity Pathway Analysis, IPA) that were altered by phenobarbital in a dose-responsive manner in CD-1 mice included “Cell cycle of chromosomal replication” and “Cell cycle: G2/M DNA damage checkpoint regulation”; these were only significantly altered above a NOTEL of 15 mg/kg/day (Geter et al., 2014). Finally, Oshida et al. (2015a) have studied microarray data for a wide range of compounds in the NextBio database, including their own studies in WT and CAR null mice, and demonstrated that a CAR biomarker gene expression signature exists which is statistically significantly enriched for known CAR activators, and is non-significant for model compounds that produce liver effects via alternative mechanisms. |
||
|
KE1 => KE2: |
Strong |
Strong support for this linkage comes from in vivo and in vitro studies with model CAR activators, and via the absence of the same effects including specific gene expression changes and cell proliferation changes (by BrdU, PCNA or Ki67 labeling index) in CAR knockout mice lacking the CAR receptor. In a study with TCPOBOP (a direct activator of mouse CAR) in mice, increases in gene expression of Cyp2b10 and Mdm2 were associated with increases in cell proliferation after 3 days or 30 weeks, and both effects were absent in CAR knockout mice (Huang et al., 2005; Wei et al., 2000). Certain compounds such as metofluthrin and pyrethrins have been shown to produce liver tumors in rats, but not mice, and these long-term effects are preceded by appropriate CAR-mediated gene changes and cell proliferation in the liver (Osimitz and Lake, 2009; Yamada et al., 2009). |
||
|
KE2, => KE3: |
Strong |
The observed increase in numbers of preneoplastic foci, usually with eosinophilic staining properties, is observed with great regularity in mode of action work of CAR activating xenobiotics where histopathology at later times has been examined. This increase in foci (mixed or eosinophilic) after 2 years was observed at tumorigenic dose levels with metofluthrin in male rats (Deguchi et al., 2009), and at tumorigenic dose levels in mice treated with phenobarbital (Jones et al., 2009). With TCPOBOP in mice, multiple eosinophilic foci were reported to co-occur along with an increased incidence of eosinophilic adenomas and carcinomas after 60 weeks of treatment (Diwan et al., 1992). With well-studied CAR activators such as phenobarbital, TCPOBOP, pyrethrins, cyproconazole and metofluthrin, increased cell proliferation has been detected at similar dose levels where increased altered foci are seen (Geter et al., 2014; Huang et al., 2005; Kolaja et al., 1996a; Kolaja et al., 1996b; Osimitz and Lake, 2009; Peffer et al., 2007; Tamura et al., 2015; Yamada et al., 2009). In addition, studies in WT and CAR null mice have shown that in the absence of CAR receptor, neither cell proliferation (KE2) nor the development of increased altered foci (KE3) and liver tumors (AO) occurs (Huang et al., 2005; Tamura et al., 2015; Tamura et al., 2013; Yamamoto, Y. et al., 2004). Therefore, there is strong support for the linkage of this earlier key event with CAR activators leading to an increase in pre-neoplastic foci. |
||
|
KE3 => AO: |
Strong |
There is a strong empirical data demonstrating that this sequence of events occurs with CAR activators. An increase in numbers of preneoplastic foci, usually with eosinophilic staining properties, is observed with great regularity in mode of action work of CAR activating xenobiotics where histopathology at later times has been examined (Elcombe et al., 2014). This increase in foci (mixed or eosinophilic) after 2 years was observed at tumorigenic dose levels with metofluthrin in male rats (Deguchi et al., 2009), and at tumorigenic dose levels in mice treated with phenobarbital (Jones et al., 2009). With TCPOBOP in mice, multiple eosinophilic foci were reported to co-occur along with an increased incidence of eosinophilic adenomas and carcinomas after 60 weeks of treatment (Diwan et al., 1992). |
||
There is a rich dataset that demonstrates the quantitative relationships between exposure and the key events and/or associative events in this AOP. These quantitative comparisons are summarized in Tables 2-5 for the example molecules phenobarbital, TCPOBOP and metofluthrin, and are described in the accompanying text and footnotes.
The Guidance for describing AOPs in the AOPwiki emphasizes that the AOP and its KERs should be evaluated with empirical data that is available in a Table format that shows: 1) Dose-response; 2) Temporality; and 3) Incidence/strength of response. Because the AOP for rodent liver tumors includes early key events (Immediate and for several days), intermediate key events (days to months) and long-term key events/AO (pre-neoplastic foci and tumors after 18-24 months), obtaining one complete set of data for one molecule at consistent dose levels in a defined sex, strain and species is highly unlikely. However, a very detailed dose-response relationship of early key events for phenobarbital, which produces tumors in male CD-1 mice, has been published by Geter et al. (2014). Therefore, Table 2 assesses phenobarbital in the manner requested by the AOPwiki Guidance, after incorporating data from other longer-term studies that provide suitable dose-response data for additional key events or associative events. The reader is referred to the original paper by Geter et al. (2014), which has calculated Benchmark Dose (BMD) values and 95% lower confidence limit (BMDL) values for a number of measured early endpoints for both male and female CD-1 mice treated with phenobarbital, which may be suitable for risk assessment applications. In addition, Table 3 provides dose-response and time concordance data for the key events and associative events following phenobarbital treatment in a different strain of mice (C57BL/10J), and this data set includes published data showing the long-term effects of increased altered foci (KE3) and increased liver adenomas and carcinomas (AO) (Jones et al., 2009).
Examining the dose-response and temporal concordance data in Table 2, the empirical data on dose-response and temporality is Moderate or Strong for each Key Event Relationship. For most early events, an increased response was seen with increasing dose, and at the tumorigenic dose levels (75 and 150 mg/kg/day), all of the preceding key events (if measured at that dose) were observed. For KE1 (altered gene expression specific to CAR activation), the gene pathways in Geter et al. (2014; by Ingenuity Pathway Analysis, IPA) that were altered by phenobarbital in a dose-responsive manner in CD-1 mice included “Cell cycle of chromosomal replication” and “Cell cycle: G2/M DNA damage checkpoint regulation”; these were only significantly altered above a NOTEL (no observed transcriptional effect level) of 15 mg/kg/day (Geter et al., 2014). Examining some specific genes that are part of these cell replication pathways, Ki-67 (a marker of cell proliferation) was increased in a dose-responsive manner at 75 and 150 mg/kg/day on Day 2; this gene signal was back to baseline on Day 7 in male mice. Tsc22 was also differentially expressed only on Day 2, at dose levels ≥15 mg/kg/day; suppression of this gene is a marker of increased cell proliferation and suppressed apoptosis. The CAR marker gene Cyp2b10 was greatly increased in its expression by phenobarbital at ≥15 mg/kg/day, and this metabolizing enzyme signal tends to stay elevated as long as treatment continues with a CAR activating compound (Osimitz and Lake, 2009; Peffer et al., 2007). Other genes related to cell cycle control that were differentially expressed in the profiles of TCPOBOP in mice (Table 4) were Gadd45b, Cdc20 and Cdk1 (Tojima et al., 2012). Comparing the genes related to cell proliferation and cell cycle control with phenobarbital and with TCPOBOP, it is clear that individual genes may be differentially expressed in one species/strain/test compound, but not in exactly the same manner with another. As a possible approach to identify a reliable set of gene expression changes that define a CAR activating compound in mice or rats, a pathway-based approach to characterize suspected CAR activators in rodents has advantages over the reliance on a small number of genes. Oshida et al. (2015a) have developed a CAR biomarker gene expression signature that is statistically significantly enriched for known CAR activators, and is non-significant for model compounds that produce liver effects via alternative mechanisms such as AhR activation or PPARα activation (Oshida et al., 2015b; Oshida et al., 2015c).
The key event of increased cell proliferation (KE2) was assessed in the experiment of Geter et al. (2014), both by BrdU labeling index and based on the incidence of “increased mitotic figures”. These two different approaches gave similar dose-response results, with lesser or equivocal increases at 15 mg/kg/day, and more robust changes at the tumorigenic dose levels of 75 and 150 mg/kg/day.
For KE3 (increases in altered foci), an increased incidence was observed in studies in C57BL/10J mice after 99 weeks of phenobarbital treatment (Table 3) at the tumorigenic dose level of 113 mg/kg/day, but not at the tumor NOEL of 22 mg/kg/day (Jones et al., 2009). Overall, the compiled data displayed in Tables 2 and 3 for phenobarbital demonstrate good dose concordance of the key events with the adverse outcome (AO). They also support a logical temporal relationship, with earlier key events preceding later ones in a manner consistent with the known biology.
Data for metofluthrin (Table 5) provides a similar dose-concordance assessment in male rats, and data for TCPOBOP (Table 4) provides a compact summary of multiple key event measurements in male mice with a very potent mouse CAR activator. Specific aspects of these results are described in the assessment of each Key Event Relationship, where appropriate.
Considerations for Potential Applications of the AOP (optional)
This AOP outlines a set of key events that have strong empirical data with several different molecules supporting the dose-concordance, temporal relationships and essentiality of the key events to causing the Adverse Outcome. Accordingly, this AOP provides a mechanistic basis for the development of Integrated Approaches to Testing and Assessment (IATA; Tollefsen et al., 2014). More specifically, the good dose concordance between the early key events and the final AO (rodent liver tumors) can support future risk assessments by demonstrating that derived endpoints (e.g. BMDL values or NOAELs) based on critical early key events are well correlated to later, downstream effects. See Geter et al. (2014) for examples of BMDL values of transcriptomic changes as well as early apical key events/associative events in mice treated with phenobarbital.
Another area of potential applications of this AOP is to illustrate the extent of data needed to establish that the AOP is operative for a regulated chemical, and to assess the lack of human relevance of liver tumors that were produced by that chemical. Table 1 illustrates the MIE, KEs, AEs and AO for this AOP, plus some examples of the typical data that can be generated based on today’s (2017) technology to demonstrate these key events and associative events. This listing in Table 1 should not be viewed as an absolute set of requirements, but as a helpful guide. In particular, as technological advances come into common usage and as our understanding of this AOP progress with time, the typical set of studies and collected data will undoubtedly be refined. A more thorough review of current methods and emerging methods for demonstrating a CAR mode of action is available in recent published literature (Peffer et al., 2017 in press).
Strong data are provided that indicate the critical key events have been shown to occur in rats and mice, but not in other mammalian species including hamsters, guinea pigs and humans. As regulatory and scientific acceptance of the lack of human relevance for multiple agents that produce rodent liver tumors via this AOP increases with time, the opportunity to reduce animal use by only assessing a limited, critical set of key events (perhaps via in vitro methods) is a potential future application of the AOP. Such approaches could avoid the need for expensive, large-scale mode of action research studies. Some specific key events that may lend themselves to these alternative approaches are:
- CAR activation assays: possibly via CAR transactivation assays, which primarily are able to detect direct CAR activating ligands for the different species of interest (see Currie et al., 2014; Omiecinski et al., 2011)
- Excluding alternative modes of action: via commercially available PPARα, AhR and similar nuclear receptor transactivation assays across species
- Transcriptomic approaches to demonstrating a CAR signature, and deriving benchmark doses from these endpoints (see Geter et al., 2014; Oshida et al., 2015a; Oshida et al., 2015b; Oshida et al., 2015c)
- Measuring additional key events as an add-on to existing subchronic and chronic rodent studies, by analysis of formalin-fixed paraffin embedded tissues for important markers such as Ki67 (a marker of cell proliferation that can be quantified via immunohistochemistry), or transcriptomic changes in key genes or pathways by use of laser-capture microdissection (LCM) followed by microarray or RT-PCR analysis of gene expression profiles (Coudry et al., 2007; Muskhelishvili et al., 2003).
References
Andersen, M.E., et al. (2014) Dose-response approaches for nuclear receptor-mediated modes of action for liver carcinogenicity: Results of a workshop. Crit Rev Toxicol, 44(1), 50-63.
Bachowski, S., et al. (1998) Role of oxidative stress in the selective toxicity of dieldrin in the mouse liver. Toxicol Appl Pharmacol, 150(2), 301-9.
Baes, M., Gulick, T., Choi, H. S., Martinoli, M. G., Simha, D., and Moore, D. D. (1994). A new orphan member of the nuclear hormone receptor superfamily that interacts with a subset of retinoic acid response elements. Mol. Cell. Biol. 14, 1544–1551.
Becker, R.A., et al. (2015) The adverse outcome pathway for rodent liver tumor promotion by sustained activation of the aryl hydrocarbon receptor. Regul Toxicol Pharmacol, 73(1), 172-90.
Benet, L.Z.and Shiner, L.B. (1985) Pharmacodynamics: the dynamics of drug absorption, distribution and elimination, in Goodman-Gilman, A., L.S. Goodman, T.W. Ralland F. Murad (eds), The Pharmacological Basis of Therapeutics, 7th edition. New York: MacMillan Publishers.
Bursch, W., et al. (2005) Apoptosis in stages of mouse hepatocarcinogenesis: failure to counterbalance cell proliferation and to account for strain differences in tumor susceptibility. Toxicol Sci, 85(1), 515-29.
Cohen, S.M. (2010) Evaluation of possible carcinogenic risk to humans based on liver tumors in rodent assays: the two-year bioassay is no longer necessary. Toxicol Pathol, 38(3), 487-501.
Corton, J.C., et al. (2014) Mode of action framework analysis for receptor-mediated toxicity: The peroxisome proliferator-activated receptor alpha (PPARalpha) as a case study. Crit Rev Toxicol, 44(1), 1-49.
Coudry, R.A., et al. (2007) Successful application of microarray technology to microdissected formalin-fixed, paraffin-embedded tissue. J Mol Diagn, 9(1), 70-9.
Currie, R.A., et al. (2014) Phenobarbital and propiconazole toxicogenomic profiles in mice show major similarities consistent with the key role that constitutive androstane receptor (CAR) activation plays in their mode of action. Toxicology, 321, 80-8.
de Boer, J.G., et al. (1998) Spontaneous mutation in lacI transgenic mice: a comparison of tissues. Mutagenesis, 13(2), 109-14.
Deguchi, Y., et al. (2009) Mode of action analysis for the synthetic pyrethroid metofluthrin-induced rat liver tumors: evidence for hepatic CYP2B induction and hepatocyte proliferation. Toxicol Sci, 108(1), 69-80.
Diwan, B.A., et al. (1992) Tumor-promoting and hepatocarcinogenic effects of 1,4-bis[2-(3,5-dichloropyridyloxy)]benzene (TCPOBOP) in DBA/2NCr and C57BL/6NCr mice and an apparent promoting effect on nasal cavity tumors but not on hepatocellular tumors in F344/NCr rats initiated with N-nitrosodiethylamine. Carcinogenesis, 13(10), 1893-901.
Diwan, B.A., et al. (1986) Lack of effect of phenobarbital on hepatocellular carcinogenesis initiated by N-nitrosodiethylamine or methylazoxymethanol acetate in male Syrian golden hamsters. Toxicol Appl Pharmacol, 86(2), 298-307.
Elcombe, C.R., et al. (2014) Mode of action and human relevance analysis for nuclear receptor-mediated liver toxicity: A case study with phenobarbital as a model constitutive androstane receptor (CAR) activator. Crit Rev Toxicol, 44(1), 64-82.
Friedman, G.D., et al. (2009) Epidemiologic evaluation of pharmaceuticals with limited evidence of carcinogenicity. Int J Cancer, 125(9), 2173-8.
Geter, D.R., et al. (2014) Dose-response modeling of early molecular and cellular key events in the CAR-mediated hepatocarcinogenesis pathway. Toxicol Sci, 138(2), 425-45.
Gold, L.S., et al. (2005) Supplement to the Carcinogenic Potency Database (CPDB): results of animal bioassays published in the general literature through 1997 and by the National Toxicology Program in 1997-1998. Toxicol Sci, 85(2), 747-808.
Goldsworthy, T.L.and Fransson-Steen, R. (2002) Quantitation of the cancer process in C57BL/6J, B6C3F1 and C3H/HeJ mice. Toxicol Pathol, 30(1), 97-105.
Haschek, W.M.and Rousseaux, C. (1998) Fundamentals of Toxicologic Pathology. San Diego, CA: Academic Press.
Hasmall, S.C.and Roberts, R.A. (1999) The perturbation of apoptosis and mitosis by drugs and xenobiotics. Pharmacol Ther, 82(1), 63-70.
Hino, S., et al. (2002) Cytoplasmic TSC-22 (transforming growth factor-beta-stimulated clone-22) markedly enhances the radiation sensitivity of salivary gland cancer cells. Biochem Biophys Res Commun, 292(4), 957-63.
Hirose, Y., et al. (2009) Comparison of the effects of the synthetic pyrethroid Metofluthrin and phenobarbital on CYP2B form induction and replicative DNA synthesis in cultured rat and human hepatocytes. Toxicology, 258(1), 64-9.
Huang, W., et al. (2005) Xenobiotic stress induces hepatomegaly and liver tumors via the nuclear receptor constitutive androstane receptor. Mol Endocrinol, 19(6), 1646-53.
IARC (2001) Phenobarbital and its sodium salts. Lyon, France: IARC Press, 2001.
Iida, M., et al. (2005) Unique patterns of gene expression changes in liver after treatment of mice for 2 weeks with different known carcinogens and non-carcinogens. Carcinogenesis, 26(3), 689-99.
James, N.H.and Roberts, R.A. (1996) Species differences in response to peroxisome proliferators correlate in vitro with induction of DNA synthesis rather than suppression of apoptosis. Carcinogenesis, 17(8), 1623-32.
Jones, H.B., Orton, T.C.and Lake, B.G. (2009) Effect of chronic phenobarbitone administration on liver tumour formation in the C57BL/10J mouse. Food Chem Toxicol, 47(6), 1333-40.
Klaunig, J.E. (1993) Selective induction of DNA synthesis in mouse preneoplastic and neoplastic hepatic lesions after exposure to phenobarbital. Environ Health Perspect, 101 Suppl 5, 235-9.
Klaunig, J.E., Ruch, R.J.and Weghorst, C.M. (1990) Comparative effects of phenobarbital, DDT, and lindane on mouse hepatocyte gap junctional intercellular communication. Toxicol Appl Pharmacol, 102(3), 553-63.
Kolaja, K.L., et al. (1996a) Subchronic effects of dieldrin and phenobarbital on hepatic DNA synthesis in mice and rats. Fundam Appl Toxicol, 29(2), 219-28.
Kolaja, K.L., et al. (1996b) Dose dependence of phenobarbital promotion of preneoplastic hepatic lesions in F344 rats and B6C3F1 mice: effects on DNA synthesis and apoptosis. Carcinogenesis, 17(5), 947-54.
La Vecchia, C.and Negri, E. (2014) A review of epidemiological data on epilepsy, phenobarbital, and risk of liver cancer. Eur J Cancer Prev, 23(1), 1-7.
Lamminpaa, A., et al. (2002) Cancer incidence among patients using antiepileptic drugs: a long-term follow-up of 28,000 patients. Eur J Clin Pharmacol, 58(2), 137-41.
LeBaron, M.J., et al. (2013) An integrated approach for prospectively investigating a mode-of-action for rodent liver effects. Toxicol Appl Pharmacol, 270(2), 164-73.
LeBaron, M.J. et al. (2014). Characterization of Nuclear Receptor-Mediated Murine Hepatocarcinogenesis of the Herbicide Pronamide and Its Human Relevance. Toxicol Sci 142(1), 74-92.
Liebermann, D.A.and Hoffman, B. (2008) Gadd45 in stress signaling. J Mol Signal, 3, 15.
Liebermann, D.A., et al. (2011) Gadd45 stress sensors in malignancy and leukemia. Crit Rev Oncog, 16(1-2), 129-40.
Martin, P., et al. (2010) Effect of prototypical inducers on ligand activated nuclear receptor regulated drug disposition genes in rodent hepatic and intestinal cells. Acta Pharmacol Sin, 31(1), 51-65.
Meek, M. E., et al., (2014). New developments in the evolution and application of the WHO/IPCS framework on mode of action/species concordance analysis. J Appl Toxicol. 34, 1-18.
Moennikes, O., et al. (2000) Lack of phenobarbital-mediated promotion of hepatocarcinogenesis in connexin32-null mice. Cancer Res, 60(18), 5087-91.
Monro, A. (1993) The paradoxical lack of interspecies correlation between plasma concentrations and chemical carcinogenicity. Regul. Toxicol. Pharmacol. 18, 115-135
Moore, S.R., et al. (1999) Spontaneous mutation frequency and pattern in Big Blue mice fed a vitamin E-supplemented diet. Environ Mol Mutagen, 34(2-3), 195-200.
Muskhelishvili, L., et al. (2003) Evaluation of cell proliferation in rat tissues with BrdU, PCNA, Ki-67(MIB-5) immunohistochemistry and in situ hybridization for histone mRNA. J Histochem Cytochem, 51(12), 1681-8.
Mutoh, S., et al., 2013. Phenobarbital indirectly activates the constitutive active androstane receptor (CAR) by inhibition of epidermal growth factor receptor signaling. Sci Signal. 6, ra31, 1-7.
Nesnow, S., et al. (2009) Discrimination of tumorigenic triazole conazoles from phenobarbital by transcriptional analyses of mouse liver gene expression. Toxicol Sci, 110(1), 68-83.
Olsen, J.H., et al. (1989) Cancer among epileptic patients exposed to anticonvulsant drugs. J Natl Cancer Inst, 81(10), 803-8.
Olsen, J.H., et al. (1995) Antiepileptic treatment and risk for hepatobiliary cancer and malignant lymphoma. Cancer Res, 55(2), 294-7.
Omiecinski, C.J., et al. (2011) Multi-species analyses of direct activators of the constitutive androstane receptor. Toxicol Sci, 123(2), 550-62.
Oshida, K., et al. (2015a) Identification of chemical modulators of the constitutive activated receptor (CAR) in a gene expression compendium. Nucl Recept Signal, 13, e002.
Oshida, K., et al. (2015b) Screening a mouse liver gene expression compendium identifies modulators of the aryl hydrocarbon receptor (AhR). Toxicology, 336, 99-112.
Oshida, K., et al. (2015c) Identification of modulators of the nuclear receptor peroxisome proliferator-activated receptor alpha (PPARalpha) in a mouse liver gene expression compendium. PLoS One, 10(2), e0112655.
Osimitz, T.G.and Lake, B.G. (2009) Mode-of-action analysis for induction of rat liver tumors by pyrethrins: relevance to human cancer risk. Crit Rev Toxicol, 39(6), 501-11.
Ozawa, S., et al. (2011) Altered expression of GADD45 genes during the development of chemical-mediated liver hypertrophy and liver tumor promotion in rats. J Toxicol Sci, 36(5), 613-23.
Parke, D.V.and Ioannides, C. (1990) Role of Cytochrome P-450 in Mouse Liver Tumor Production, Mouse Liver Carcinogenesis: Mechanisms and Species ComparisonsAlan R. Liss, Inc., 215–230.
Parzefall W, Erber E, Sedivy R, Schulte-Hermann R. (1991). Testing for induction of DNA synthesis in human hepatocyte primary cultures by rat liver tumor promoters. Cancer Res, 51, 1143–7.
Peffer, R.C., et al. (2007) Mouse liver effects of cyproconazole, a triazole fungicide: role of the constitutive androstane receptor. Toxicol Sci, 99(1), 315-25.
Peffer, R. C. et al., (2017). Minimum Datasets to Establish a CAR-Mediated Mode of Action for Rodent Liver Tumors: An EU Crop Protection Perspective. Regul. Toxicol. Pharmacol., submitted for publication.
Pereira, M.A. (1993) Comparison in C3H and C3B6F1 mice of the sensitivity to diethylnitrosamine-initiation and phenobarbital-promotion to the extent of cell proliferation. Carcinogenesis, 14(2), 299-302.
Phillips, J.M.and Goodman, J.I. (2008) Identification of genes that may play critical roles in phenobarbital (PB)-induced liver tumorigenesis due to altered DNA methylation. Toxicol Sci, 104(1), 86-99.
Poole, T.M.and Drinkwater, N.R. (1995) Hormonal and genetic interactions in murine hepatocarcinogenesis. Prog Clin Biol Res, 391, 187-94.
Ross, J., et al. (2010) Human constitutive androstane receptor (CAR) and pregnane X receptor (PXR) support the hypertrophic but not the hyperplastic response to the murine nongenotoxic hepatocarcinogens phenobarbital and chlordane in vivo. Toxicol Sci, 116(2), 452-66.
Rossi L, Ravera M, Repetti G, Santi L. (1977). Long-term administration of DDT or phenobarbital-Na in Wistar rats. Int J Cancer, 19, 179–85.
Scheer, N. and Wilson, I. D., (2016). A comparison between genetically humanized and chimeric liver humanized mouse models for studies in drug metabolism and toxicity. Drug Discov Today. 21, 250-63.
Schulte-Hermann, R., et al. (1990) DNA synthesis, apoptosis, and phenotypic expression as determinants of growth of altered foci in rat liver during phenobarbital promotion. Cancer Res, 50(16), 5127-35.
Selby, J.V., Friedman, G.D.and Fireman, B.H. (1989) Screening prescription drugs for possible carcinogenicity: eleven to fifteen years of follow-up. Cancer Res, 49(20), 5736-47.
Shizu, R., et al. (2013) Xenobiotic-induced hepatocyte proliferation associated with constitutive active/androstane receptor (CAR) or peroxisome proliferator-activated receptor alpha (PPARalpha) is enhanced by pregnane X receptor (PXR) activation in mice. PLoS One, 8(4), e61802.
Stanley, L.A., et al. (2006) PXR and CAR: nuclear receptors which play a pivotal role in drug disposition and chemical toxicity. Drug Metab Rev, 38(3), 515-97.
Stenbäck et al. (1986), J. Natl. Cancer Inst. 75, 327-333
Sueyoshi, T., and Negishi, M. (2001). Phenobarbital response elements of cytochrome p450 genes and nuclear receptors. Annu. Rev. Pharmacol. Toxicol. 41, 123–143.
Tamura, K., et al. (2015) Involvement of constitutive androstane receptor in liver hypertrophy and liver tumor development induced by triazole fungicides. Food Chem Toxicol, 78, 86-95.
Tamura, K., et al. (2013) Dose-response involvement of constitutive androstane receptor in mouse liver hypertrophy induced by triazole fungicides. Toxicol Lett, 221(1), 47-56.
Tanaka, T., Mori, H. and Williams, G. (1987). Enhancement of dimethylnitrosamine-initiated hepatocarcinogenesis in hamsters by subsequent administration of carbon tetrachloride but not phenobarbital or p,p′-dichlorodiphenyltrichloroethane. Carcinogenesis. 8 (9): 1171-1178
Thatcher, N.J.and Caldwell, J. (1994) Origins of hepatomegaly produced by dexamethasone (DEX), pregnenolone 16 alpha-carbonitrile (PCN) and phenobarbitone (PB) in female Sprague-Dawley rats. Biochem Soc Trans, 22(2), 132S.
Timsit, Y.E.and Negishi, M. (2007) CAR and PXR: the xenobiotic-sensing receptors. Steroids, 72(3), 231-46.
Tojima, H., et al. (2012) Ligand dependent hepatic gene expression profiles of nuclear receptors CAR and PXR. Toxicol Lett, 212(3), 288-97.
Tollefsen, K.E., et al. (2014) Applying Adverse Outcome Pathways (AOPs) to support Integrated Approaches to Testing and Assessment (IATA). Regul Toxicol Pharmacol, 70(3), 629-40.
Wei, P., et al. (2000) The nuclear receptor CAR mediates specific xenobiotic induction of drug metabolism. Nature, 407(6806), 920-3.
White, S.J., McLean, A.E.and Howland, C. (1979) Anticonvulsant drugs and cancer. A cohort study in patients with severe epilepsy. Lancet, 2(8140), 458-61.
Whysner, J., Ross, P.M.and Williams, G.M. (1996) Phenobarbital mechanistic data and risk assessment: enzyme induction, enhanced cell proliferation, and tumor promotion. Pharmacol Ther, 71(1-2), 153-91.
Willson, T.M.and Kliewer, S.A. (2002) PXR, CAR and drug metabolism. Nat Rev Drug Discov, 1(4), 259-66.
Yamada, T., et al. (2009) Case study: an evaluation of the human relevance of the synthetic pyrethroid metofluthrin-induced liver tumors in rats based on mode of action. Toxicol Sci, 108(1), 59-68.
Yamada T, Okuda Y, Kushida M, Sumida K, Takeuchi H, Nagahori H, Fukuda T, Lake BG, Cohen SM, Kawamura S. (2014). Human hepatocytes support the hypertrophic but not the hyperplastic response to the murine nongenotoxic hepatocarcinogen sodium phenobarbital in an in vivo study using a chimeric mouse with humanized liver. Toxicol Sci. 142(1):137-57.
Yamada, T., Kikumoto, H., Lake, B. and Kawamura, S. (2015). Lack of effect of metofluthrin and sodium phenobarbital on replicative DNA synthesis and Ki-67 mRNA expression in cultured human hepatocytes. Toxicol. Res. 4, 901-913.
Yamamoto, K., Murae, M.and Yasuda, M. (1991) RGD-containing peptides inhibit experimental peritoneal seeding of human ovarian cancer cells. Nihon Sanka Fujinka Gakkai Zasshi, 43(12), 1687-92.
Yamamoto, Y., et al. (2004) The orphan nuclear receptor constitutive active/androstane receptor is essential for liver tumor promotion by phenobarbital in mice. Cancer Res, 64(20), 7197-200.