
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 432
Title
A descriptive phrase which references both the Molecular Initiating Event and Adverse Outcome.It should take the form “MIE leading to AO”. For example, “Aromatase inhibition leading to reproductive dysfunction” where Aromatase inhibition is the MIE and reproductive dysfunction the AO. In cases where the MIE is unknown or undefined, the earliest known KE in the chain (i.e., furthest upstream) should be used in lieu of the MIE and it should be made clear that the stated event is a KE and not the MIE.
More help
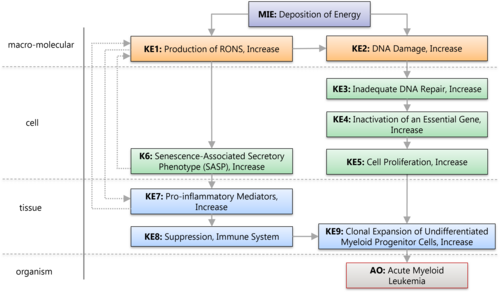
Deposition of Energy by Ionizing Radiation leading to Acute Myeloid Leukemia
Short name
A name that succinctly summarises the information from the title. This name should not exceed 90 characters.
More help
Ionizing Radiation-Induced AML
The current version of the Developer's Handbook will be automatically populated into the Handbook Version field when a new AOP page is created.Authors have the option to switch to a newer (but not older) Handbook version any time thereafter.
More help
Handbook Version v2.0
Graphical Representation
A graphical representation of the AOP.This graphic should list all KEs in sequence, including the MIE (if known) and AO, and the pair-wise relationships (links or KERs) between those KEs.
More help
Point of Contact
The user responsible for managing the AOP entry in the AOP-KB and controlling write access to the page by defining the contributors as described in the next section.
More help
Dag Anders Brede
(email point of contact)
Contributors
Users with write access to the AOP page. Entries in this field are controlled by the Point of Contact.
More help
- Dag Anders Brede
- Kimberly Applegate
- Maria Gomolka
- Dmitry Klokov
- Katalin Lumniczky
- Christophe Badie
Coaches
This field is used to identify coaches who supported the development of the AOP.Each coach selected must be a registered author.
More help
OECD Information Table
Provides users with information concerning how actively the AOP page is being developed and whether it is part of the OECD Workplan and has been reviewed and/or endorsed. OECD Project: Assigned upon acceptance onto OECD workplan. This project ID is managed and updated (if needed) by the OECD. OECD Status: For AOPs included on the OECD workplan, ‘OECD status’ tracks the level of review/endorsement of the AOP . This designation is managed and updated by the OECD. Journal-format Article: The OECD is developing co-operation with Scientific Journals for the review and publication of AOPs, via the signature of a Memorandum of Understanding. When the scientific review of an AOP is conducted by these Journals, the journal review panel will review the content of the Wiki. In addition, the Journal may ask the AOP authors to develop a separate manuscript (i.e. Journal Format Article) using a format determined by the Journal for Journal publication. In that case, the journal review panel will be required to review both the Wiki content and the Journal Format Article. The Journal will publish the AOP reviewed through the Journal Format Article. OECD iLibrary published version: OECD iLibrary is the online library of the OECD. The version of the AOP that is published there has been endorsed by the OECD. The purpose of publication on iLibrary is to provide a stable version over time, i.e. the version which has been reviewed and revised based on the outcome of the review. AOPs are viewed as living documents and may continue to evolve on the AOP-Wiki after their OECD endorsement and publication.
More help
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 29, 2023 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Deposition of Energy | August 23, 2024 09:20 |
| Increase in reactive oxygen and nitrogen species (RONS) | May 08, 2019 12:30 |
| Increase, DNA damage | May 08, 2019 12:28 |
| Increased Pro-inflammatory mediators | September 17, 2024 08:35 |
| Increase, Mutations in Critical Genes | September 16, 2017 10:16 |
| Acute Myeloid Leukemia | December 03, 2021 07:45 |
| Inadequate DNA repair | March 08, 2024 12:15 |
| Increase, Cell Proliferation | December 10, 2024 15:01 |
| Suppression, Immune system | December 03, 2016 16:37 |
| Ionizing Radiation | May 07, 2019 12:12 |
Abstract
A concise and informative summation of the AOP under development that can stand-alone from the AOP page. The aim is to capture the highlights of the AOP and its potential scientific and regulatory relevance.
More help
AOP Development Strategy
Context
Used to provide background information for AOP reviewers and users that is considered helpful in understanding the biology underlying the AOP and the motivation for its development.The background should NOT provide an overview of the AOP, its KEs or KERs, which are captured in more detail below.
More help
Strategy
Provides a description of the approaches to the identification, screening and quality assessment of the data relevant to identification of the key events and key event relationships included in the AOP or AOP network.This information is important as a basis to support the objective/envisaged application of the AOP by the regulatory community and to facilitate the reuse of its components. Suggested content includes a rationale for and description of the scope and focus of the data search and identification strategy/ies including the nature of preliminary scoping and/or expert input, the overall literature screening strategy and more focused literature surveys to identify additional information (including e.g., key search terms, databases and time period searched, any tools used).
More help
Summary of the AOP
This section is for information that describes the overall AOP.The information described in section 1 is entered on the upper portion of an AOP page within the AOP-Wiki. This is where some background information may be provided, the structure of the AOP is described, and the KEs and KERs are listed.
More help
Events:
Molecular Initiating Events (MIE)
An MIE is a specialised KE that represents the beginning (point of interaction between a prototypical stressor and the biological system) of an AOP.
More help
Key Events (KE)
A measurable event within a specific biological level of organisation.
More help
Adverse Outcomes (AO)
An AO is a specialized KE that represents the end (an adverse outcome of regulatory significance) of an AOP.
More help
| Type | Event ID | Title | Short name |
|---|
| MIE | 1686 | Deposition of Energy | Energy Deposition |
| KE | 1632 | Increase in reactive oxygen and nitrogen species (RONS) | Increase in RONS |
| KE | 1194 | Increase, DNA damage | Increase, DNA Damage |
| KE | 155 | Inadequate DNA repair | Inadequate DNA repair |
| KE | 876 | Increase, Mutations in Critical Genes | Increase, Mutations in Critical Genes |
| KE | 870 | Increase, Cell Proliferation | Increase, Cell Proliferation |
| KE | 1493 | Increased Pro-inflammatory mediators | Increased pro-inflammatory mediators |
| KE | 403 | Suppression, Immune system | Suppression, Immune system |
| AO | 1955 | Acute Myeloid Leukemia | AML |
Relationships Between Two Key Events (Including MIEs and AOs)
This table summarizes all of the KERs of the AOP and is populated in the AOP-Wiki as KERs are added to the AOP.Each table entry acts as a link to the individual KER description page.
More help
Network View
This network graphic is automatically generated based on the information provided in the MIE(s), KEs, AO(s), KERs and Weight of Evidence (WoE) summary tables. The width of the edges representing the KERs is determined by its WoE confidence level, with thicker lines representing higher degrees of confidence. This network view also shows which KEs are shared with other AOPs.
More help
Prototypical Stressors
A structured data field that can be used to identify one or more “prototypical” stressors that act through this AOP. Prototypical stressors are stressors for which responses at multiple key events have been well documented.
More help
| Name |
|---|
| Ionizing Radiation |
Life Stage Applicability
The life stage for which the AOP is known to be applicable.
More help
Taxonomic Applicability
Latin or common names of a species or broader taxonomic grouping (e.g., class, order, family) can be selected.In many cases, individual species identified in these structured fields will be those for which the strongest evidence used in constructing the AOP was available.
More help
Sex Applicability
The sex for which the AOP is known to be applicable.
More help
| Sex | Evidence |
|---|---|
| Mixed |
Overall Assessment of the AOP
Addressess the relevant biological domain of applicability (i.e., in terms of taxa, sex, life stage, etc.) and Weight of Evidence (WoE) for the overall AOP as a basis to consider appropriate regulatory application (e.g., priority setting, testing strategies or risk assessment).
More help
Domain of Applicability
Addressess the relevant biological domain(s) of applicability in terms of sex, life-stage, taxa, and other aspects of biological context.
More help
Essentiality of the Key Events
The essentiality of KEs can only be assessed relative to the impact of manipulation of a given KE (e.g., experimentally blocking or exacerbating the event) on the downstream sequence of KEs defined for the AOP. Consequently, evidence supporting essentiality is assembled on the AOP page, rather than on the independent KE pages that are meant to stand-alone as modular units without reference to other KEs in the sequence. The nature of experimental evidence that is relevant to assessing essentiality relates to the impact on downstream KEs and the AO if upstream KEs are prevented or modified. This includes: Direct evidence: directly measured experimental support that blocking or preventing a KE prevents or impacts downstream KEs in the pathway in the expected fashion. Indirect evidence: evidence that modulation or attenuation in the magnitude of impact on a specific KE (increased effect or decreased effect) is associated with corresponding changes (increases or decreases) in the magnitude or frequency of one or more downstream KEs.
More help
Evidence Assessment
Addressess the biological plausibility, empirical support, and quantitative understanding from each KER in an AOP.
More help
Known Modulating Factors
Modulating factors (MFs) may alter the shape of the response-response function that describes the quantitative relationship between two KES, thus having an impact on the progression of the pathway or the severity of the AO.The evidence supporting the influence of various modulating factors is assembled within the individual KERs.
More help
Quantitative Understanding
Optional field to provide quantitative weight of evidence descriptors.
More help
Considerations for Potential Applications of the AOP (optional)
Addressess potential applications of an AOP to support regulatory decision-making.This may include, for example, possible utility for test guideline development or refinement, development of integrated testing and assessment approaches, development of (Q)SARs / or chemical profilers to facilitate the grouping of chemicals for subsequent read-across, screening level hazard assessments or even risk assessment.
More help
References
List of the literature that was cited for this AOP.
More help