
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 134
Title
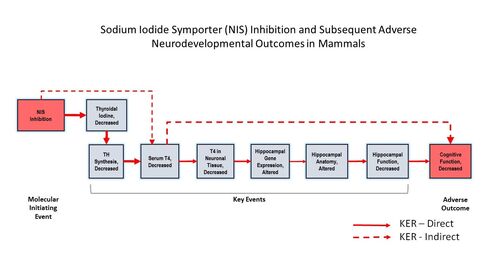
Sodium Iodide Symporter (NIS) Inhibition and Subsequent Adverse Neurodevelopmental Outcomes in Mammals
Short name
Graphical Representation
Point of Contact
Contributors
- Mary Gilbert
- Kevin Crofton
Coaches
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
This AOP was last modified on April 27, 2026 07:27
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Inhibition, Na+/I- symporter (NIS) | April 27, 2026 05:31 |
| Thyroid hormone synthesis, Decreased | November 04, 2022 09:25 |
| Decrease of Thyroidal iodide | April 04, 2019 09:00 |
| Thyroxine (T4) in serum, Decreased | October 10, 2022 08:52 |
| Thyroxine (T4) in neuronal tissue, Decreased | April 04, 2019 09:13 |
| Hippocampal gene expression, Altered | August 11, 2018 09:26 |
| Hippocampal anatomy, Altered | July 24, 2024 23:09 |
| Hippocampal Physiology, Altered | July 24, 2024 23:15 |
| Cognitive function, decreased | July 25, 2024 17:23 |
| Inhibition, Na+/I- symporter (NIS) leads to Thyroidal Iodide, Decreased | May 29, 2018 07:24 |
| Thyroidal Iodide, Decreased leads to TH synthesis, Decreased | June 04, 2018 06:11 |
| TH synthesis, Decreased leads to T4 in serum, Decreased | October 10, 2022 08:56 |
| T4 in serum, Decreased leads to T4 in neuronal tissue, Decreased | April 04, 2019 10:50 |
| T4 in neuronal tissue, Decreased leads to Hippocampal gene expression, Altered | August 11, 2018 19:18 |
| Hippocampal gene expression, Altered leads to Hippocampal anatomy, Altered | August 11, 2018 19:05 |
| Hippocampal anatomy, Altered leads to Hippocampal Physiology, Altered | July 25, 2024 18:38 |
| Hippocampal Physiology, Altered leads to Cognitive function, decreased | July 26, 2024 12:57 |
| T4 in serum, Decreased leads to Cognitive function, decreased | April 27, 2026 05:59 |
Abstract
This AOP describes one adverse outcome that results from the inhibition of the sodium iodine symporter (NIS) during mammalian development. Inhibition of NIS, the molecular-initiating event (MIE), results in decreased iodine uptake, decreased thyroidal iodine content, subsequent decreased synthesis of thyroid hormones (THs), and reduction in circulating levels of THs. THs are essential for normal human brain development, both prenatally and postnatally. Therefore, chemicals that interfere with TH synthesis have the potential to cause TH insufficiency that may result in adverse neurodevelopmental effects in offspring. This AOP includes changes in brain TH concentrations and subsequent impacts on hippocampal development that lead to declines in cognitive spatial behavior. The weight of evidence for this AOP is strong. And, while there are currently computational quantitative models that can predict serum TH levels from NIS inhibition, there is currently a lack of quantitative understanding of the degree of serum TH disruption that leads to the adverse outcome.
AOP Development Strategy
Context
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 424 | Inhibition, Na+/I- symporter (NIS) | Inhibition, Na+/I- symporter (NIS) |
| KE | 277 | Thyroid hormone synthesis, Decreased | TH synthesis, Decreased |
| KE | 425 | Decrease of Thyroidal iodide | Thyroidal Iodide, Decreased |
| KE | 281 | Thyroxine (T4) in serum, Decreased | T4 in serum, Decreased |
| KE | 280 | Thyroxine (T4) in neuronal tissue, Decreased | T4 in neuronal tissue, Decreased |
| KE | 756 | Hippocampal gene expression, Altered | Hippocampal gene expression, Altered |
| KE | 757 | Hippocampal anatomy, Altered | Hippocampal anatomy, Altered |
| KE | 758 | Hippocampal Physiology, Altered | Hippocampal Physiology, Altered |
| AO | 402 | Cognitive function, decreased | Cognitive function, decreased |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
| Inhibition, Na+/I- symporter (NIS) leads to Thyroidal Iodide, Decreased | adjacent | High | High |
| Thyroidal Iodide, Decreased leads to TH synthesis, Decreased | adjacent | High | High |
| TH synthesis, Decreased leads to T4 in serum, Decreased | adjacent | High | High |
| T4 in serum, Decreased leads to T4 in neuronal tissue, Decreased | adjacent | High | Low |
| T4 in neuronal tissue, Decreased leads to Hippocampal gene expression, Altered | adjacent | Moderate | Low |
| Hippocampal gene expression, Altered leads to Hippocampal anatomy, Altered | adjacent | Moderate | Low |
| Hippocampal anatomy, Altered leads to Hippocampal Physiology, Altered | adjacent | Moderate | Low |
| Hippocampal Physiology, Altered leads to Cognitive function, decreased | adjacent | Moderate | Low |
| T4 in serum, Decreased leads to Cognitive function, decreased | non-adjacent | High | Low |
Network View
Prototypical Stressors
Life Stage Applicability
| Life stage | Evidence |
|---|---|
| Perinatal | High |
Taxonomic Applicability
Sex Applicability
| Sex | Evidence |
|---|---|
| Male | High |
| Female | High |
Overall Assessment of the AOP
Domain of Applicability
Taxonomic: According to the evaluation of the empirical taxonomic domain of applicability (tDOA) of an adverse outcome pathway network for thyroid hormone system disruption (THSD) by Haigis et al., 2023, the level of confidence for a linkage between NIS inhibition and reduced thyroid hormone (TH) levels as well as for a linkage between THSD and developmental neurotoxicity (DNT) in general, was considered high for mammals and moderate for amphibians. For fish, the level of confidence was evaluated to be moderate for a linkage between NIS inhibition and reduced TH levels and high for a linkage between THSD and DNT in general. In addition, the level of confidence was considered moderate for birds for a linkage between THSD and DNT in general. This was supported by structural protein conservation analysis by Lalone et al., 2018 and Haigis et al., 2023. Structural protein conservation of mammalian, fish, amphibian, reptilian and avian NIS was found compared to the human (Homo sapiens) protein target using the U.S. Environmental Protection Agency’s Sequence Alignment to Predict Across Species Susceptibility (SeqAPASS v6.0; seqapass.epa.gov/seqapass/) tool, while acknowledging the potential existence of interspecies differences in conservation. However, evidence specifically linking THSD to impaired learning and memory in nonmammalian vertebrates is generally missing. No empirical evidence linking NIS inhibition to THSD was found for reptiles and birds. Furthermore, no empirical evidence linking THSD to decreased cognitive function was found for reptiles, but this taxon is considered part of the plausible tDOA based on the evaluation by Haigis et al., 2023. Scientific inference and the presence of comparable neurological structures indicate that decreased cognitive function can plausibly be measured and is plausibly linked to THSD (Hussan et al., 2022, Naumann et al., 2015, Rivera and Lock, 2008).
Essentiality of the Key Events
Evidence Assessment
Known Modulating Factors
| Modulating Factor (MF) | Influence or Outcome | KER(s) involved |
|---|---|---|
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
References
Haigis A-C., Vergauwen L., LaLone C.A., Villeneuve D.L., O'Brien J.M., Knapen D. (2023). Cross-species applicability of an adverse outcome pathway network for thyroid hormone system disruption. Toxicol Sci. 195, 1-27.
Hussan, M. T., Sakai, A., and Matsui, H. (2022). Glutamatergic pathways in the brains of turtles: A comparative perspective among reptiles, birds, and mammals. Front. Neuroana. 16, 937504.
Lalone, C. A., Villeneuve, D. L., Doering, J. A., Blackwell, B. R., Transue, T. R., Simmons, C. W., Swintek, J., Degitz, S. J., Williams, A. J., and Ankley, G. T. (2018).
Naumann, R. K., Ondracek, J. M., Reiter, S., Shein-Idelson, M., Tosches, M. A., Yamawaki, T. M., and Laurent, G. (2015). The reptilian brain. Curr. Biol. 25, R317–R321
Rivera, S., and Lock, B. (2008). The reptilian thyroid and parathyroid glands. Vet. Clin. North Am. Exot. Anim. Pract. 11, 163–175, viii.