
This AOP is licensed under the BY-SA license. This license allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use. If you remix, adapt, or build upon the material, you must license the modified material under identical terms.
AOP: 293
Title
Increased DNA damage leading to increased risk of breast cancer
Short name
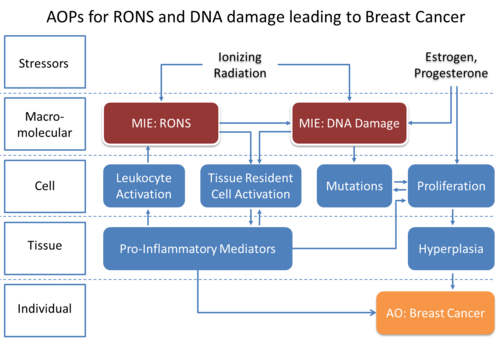
Graphical Representation
Point of Contact
Contributors
- Jessica Helm
- Ruthann Rudel
Coaches
- Tanja Burgdorf
- Shihori Tanabe
OECD Information Table
| OECD Project # | OECD Status | Reviewer's Reports | Journal-format Article | OECD iLibrary Published Version |
|---|---|---|---|---|
| 1.80 | Under Development |
This AOP was last modified on April 29, 2023 16:03
Revision dates for related pages
| Page | Revision Date/Time |
|---|---|
| Increase, DNA damage | May 08, 2019 12:28 |
| N/A, Breast Cancer | February 29, 2024 08:48 |
| Tissue resident cell activation | November 15, 2024 10:51 |
| Increased Pro-inflammatory mediators | September 17, 2024 08:35 |
| Increase in reactive oxygen and nitrogen species (RONS) | May 08, 2019 12:30 |
| Leukocyte recruitment/activation | December 01, 2017 09:33 |
| Increase, Cell Proliferation (Epithelial Cells) | May 08, 2019 12:41 |
| Increased, Ductal Hyperplasia | September 16, 2017 10:17 |
| Increase, Mutations | May 15, 2023 08:47 |
| Increase, DNA Damage leads to Increase, Mutations | May 08, 2019 14:41 |
| Increase, Mutations leads to Increase, Cell Proliferation (Epithelial Cells) | May 08, 2019 14:54 |
| Increase, Cell Proliferation (Epithelial Cells) leads to Increase, Mutations | May 08, 2019 15:02 |
| Increase, Cell Proliferation (Epithelial Cells) leads to Increased, Ductal Hyperplasia | December 03, 2016 16:38 |
| Increased, Ductal Hyperplasia leads to N/A, Breast Cancer | May 08, 2019 15:09 |
| Increase in RONS leads to Tissue resident cell activation | May 09, 2019 15:47 |
| Increase in RONS leads to Increase, DNA Damage | May 08, 2019 14:43 |
| Tissue resident cell activation leads to Increased pro-inflammatory mediators | August 02, 2018 03:37 |
| Increased pro-inflammatory mediators leads to Leukocyte recruitment/activation | November 12, 2018 10:55 |
| Increased pro-inflammatory mediators leads to Increase in RONS | May 09, 2019 16:00 |
| Leukocyte recruitment/activation leads to Increase in RONS | May 09, 2019 15:59 |
| Increase, DNA Damage leads to Tissue resident cell activation | May 10, 2019 17:15 |
| Increased pro-inflammatory mediators leads to Increase, Cell Proliferation (Epithelial Cells) | May 10, 2019 13:30 |
| Increased pro-inflammatory mediators leads to N/A, Breast Cancer | May 10, 2019 13:41 |
| Ionizing Radiation | May 07, 2019 12:12 |
| Other DNA damaging agents | May 10, 2019 14:46 |
Abstract
Knowledge about established breast carcinogens can support improved 21st century toxicological testing methods by identifying key mechanistic events. Ionizing radiation (IR) increases the risk of breast cancer, especially for women and for exposure at younger ages. We used the Adverse Outcome Pathway (AOP) framework to outline and evaluate the evidence linking ionizing radiation with breast cancer from molecular initiating events (MIE) to the adverse outcome (AO) through intermediate key events (KE). We identified prospective key events using recent literature on ionizing radiation and carcinogenesis, focusing on review articles. We searched PubMed for each key event and ionizing radiation, and used references cited in the resulting papers and targeted searches with related key words to identify additional papers. We manually curated publications and evaluated data quality. The AOP specifies that ionizing radiation directly and indirectly causes DNA damage and increases production of reactive oxygen and nitrogen species (RONS), and these are designated as MIEs. RONS lead to DNA damage (MIE) which leads to mutations (KE). Proliferation (KE) amplifies the effects of DNA damage and mutations leading to the AO of breast cancer. Separately, RONS (and DNA damage) also increase inflammation (KE). Inflammation contributes to direct and indirect effects (effects in cells not directly reached by IR) via positive feedback to RONS and DNA damage, and separately increases proliferation and the AO through pro-carcinogenic effects on cells and tissue. These MIEs and KEs overlap at multiple points with events characteristic of “background” induction of breast carcinogenesis, including hormone-responsive proliferation, oxidative activity, and DNA damage. These overlaps make the breast particularly susceptible to ionizing radiation and reinforce the importance of these MIEs and KEs as part of toxicological panels for carcinogenicity. The AOP identifies areas for additional research, including better description of the time and dose-dependence of MIEs and KEs in mammary tissues directly and indirectly exposed to IR.
This AOP extends the characteristics of mammary carcinogens beyond DNA damage, highlighting the important role in breast cancer of chemicals that increase RONS, cell proliferation, and inflammation. Chemicals that increase these biological processes should be considered potential breast carcinogens, and predictive methods should be developed to identify chemicals that increase these processes. Ultimately, this AOP will improve methods that predict chemical breast carcinogens so that exposure can be reduced.
AOP Development Strategy
Context
Breast cancer imposes a significant burden on women worldwide and is an important target for prevention. It is the most common invasive cancer in women with the highest rates found in North America and Europe (Ervik, Lam et al. 2016), and incidence is increasing globally (Forouzanfar, Foreman et al. 2011). In the US, the National Cancer Institute estimates that the total number of new breast cancers will increase from 283,000 to 441,000 between 2011 and 2030 (Rosenberg, Barker et al. 2015). Twin studies suggest that heritable factors explain at most a third of breast cancers and around 60% of all cancers are related to avoidable factors (Ronckers, Erdmann et al. 2005; Colditz and Wei 2012; Moller, Mucci et al. 2016), leaving significant room for prevention efforts focused on environmental factors to reduce new cases. Well-documented risk factors include tobacco and alcohol use as well as obesity, physical activity, and exposure to carcinogens (Colditz and Wei 2012).
Breast cancer incidence and risk varies with age, and hormonal and reproductive factors. Incidence increases with age, with rates among women increasing rapidly after age 30 and peaking around 75 years of age (NCI SEER 2016). Incidence is strongly influenced by the reproductive hormones estrogen and progesterone and by childbirth, which influence the proliferation and number of cells in the breast (Gertig, Stillman et al. 1999; Ronckers, Erdmann et al. 2005; Bijwaard, Brenner et al. 2010; Dall, Risbridger et al. 2017). Breast cancer risk increases with earlier puberty or later menopause (CGHFBC 2012; Bodicoat, Schoemaker et al. 2014), factors that increase cumulative estrogen and progesterone exposure and the number of proliferative menstrual cycles in the breast. Conversely, risk decreases in women with ovariectomies (Olson, Sellers et al. 2004; Press, Sullivan-Halley et al. 2011) and with menopause (CGHFBC 2012). Risk also decreases with number of pregnancies, breastfeeding, and increasing time since childbirth. This decrease in risk is thought to be related to the differentiation of stem cells in the breast during pregnancy and lactation and the decline in epithelial cell number after childbirth (Gertig, Stillman et al. 1999; Dall, Risbridger et al. 2017). Breast cancer incidence in men is less than 1% that of women, a difference attributed to low levels of estrogen and progesterone and few breast epithelial cells (Stang and Thomssen 2008).
Strategy
Summary of the AOP
Events:
Molecular Initiating Events (MIE)
Key Events (KE)
Adverse Outcomes (AO)
| Type | Event ID | Title | Short name |
|---|
| MIE | 1194 | Increase, DNA damage | Increase, DNA Damage |
| KE | 1492 | Tissue resident cell activation | Tissue resident cell activation |
| KE | 1493 | Increased Pro-inflammatory mediators | Increased pro-inflammatory mediators |
| KE | 1632 | Increase in reactive oxygen and nitrogen species (RONS) | Increase in RONS |
| KE | 1494 | Leukocyte recruitment/activation | Leukocyte recruitment/activation |
| KE | 1182 | Increase, Cell Proliferation (Epithelial Cells) | Increase, Cell Proliferation (Epithelial Cells) |
| AO | 185 | Increase, Mutations | Increase, Mutations |
| AO | 1192 | Increased, Ductal Hyperplasia | Increased, Ductal Hyperplasia |
| AO | 1193 | N/A, Breast Cancer | N/A, Breast Cancer |
Relationships Between Two Key Events (Including MIEs and AOs)
| Title | Adjacency | Evidence | Quantitative Understanding |
|---|
Network View
Prototypical Stressors
Life Stage Applicability
Taxonomic Applicability
Sex Applicability
Overall Assessment of the AOP
Domain of Applicability
While the key events described here are likely relevant to all tissues after exposure to IR, it is particularly relevant to the female mammary gland. While ionizing radiation causes many kinds of cancers including leukemia, lung, bladder, and thyroid cancers (BEIR 2006; Preston, Ron et al. 2007), breast cancers are among the cancers most increased by exposure to ionizing radiation (Preston, Ron et al. 2007).
The lengthy and hormone-dependent developmental trajectory of the mammary gland is likely to be a major factor in its susceptibility to breast cancer. Numerous epidemiological and laboratory studies support the requirement for ovarian hormones in the risk of breast cancer from ionizing radiation (Grant, Cologne et al. 2018). Although at first examination breast cancer from ionizing radiation and hormones involve very different processes, in fact the hormone-dependent and ionizing radiation pathways of carcinogenesis intersect at multiple points that are part of breast development leaving the hormone-exposed breast more vulnerable to radiation. Two studies in humans and rats also suggest that IR can increase long term concentrations of circulating estrogen which would further amplify any additive effects, although additional evidence is needed (Suman, Johnson et al. 2012; Grant, Cologne et al. 2018).
One major mechanism promoting breast cancer from ionizing radiation is the proliferation of breast stem cells. Stem cells are considered to be important to initiation because of their long life and capacity to pass on mutations to many progeny. Breast tissue is responsive to estrogen and progesterone, reproductive hormones that rise at puberty and stimulate cellular proliferation with each reproductive cycle and in pregnancy. These hormonal proliferative cycles increase the risk of cancer in breast tissue (Brisken, Hess et al. 2015). IR increases the long term proliferation of stem cells in pubertal but not adult mammary gland (Nguyen, Oketch-Rabah et al. 2011; Datta, Hyduke et al. 2012; Snijders, Marchetti et al. 2012; Suman, Johnson et al. 2012; Tang, Fernandez-Garcia et al. 2014). Replication of stem cells in the IR-exposed breast is therefore particularly elevated during puberty, likely contributing to the increased susceptibility to breast cancer from IR at this age.
Another vulnerability of the breast to IR is a byproduct of proliferation: mutations. Replication itself increases the likelihood of mutations, which add to mutations arising from IR and increase the likelihood of oncogenic transformation (Atashgaran, Wrin et al. 2016). Furthermore, the high replication rate of mammary gland epithelial cells during puberty and pregnancy increases reliance on homologous recombination pathways (Kass, Lim et al. 2016). Disruption of these HR processes by IR-induced mutation or increased demand for repair can increase mutation rates and increase tumorigenesis (Mahdi, Huo et al. 2018). This disruption is particularly relevant for mammary stem cells which are highly replicating and dependent on HR but shift to NHEJ to respond to DNA damage from IR (Chang, Zhang et al. 2015). The consequence of mutations in stem cells is significant, since these cells can clonally expand to generate many mutated progeny. However, errors in stem cell division may not be the sole or primary factor driving cancer from radiation, since excess cancer risk for solid cancers at different sites from the atomic bomb are not clearly related to the number of stem cell divisions at that site (Tomasetti, Li et al. 2017).
The elevated estrogen associated with development and the estrous cycle may also have direct effects that further complement the carcinogenic effects of IR. Estrogen directly increases oxidative stress in virgin (but not parous) mice (Yuan, Dietrich et al. 2016), interferes with DNA repair (Pedram, Razandi et al. 2009; Li, Chen et al. 2014) increases mutations (Mailander, Meza et al. 2006), and increases TGF-b (Jerry, Dunphy et al. 2010). Each of these effects would increase the impact of the same events arising from IR alone.
Inflammation from the estrous cycle may also contribute to tumorigenesis following IR. Cytokines and macrophages play an integral role in mammary gland development and ductal elaboration, with alternating inflammatory, immune surveillance, and phagocytic activity occurring over each estrous cycle (Hodson, Chua et al. 2013; Atashgaran, Wrin et al. 2016; Brady, Chuntova et al. 2016). This inflammation could potentially increase IR-induced DNA damage and mutations and promote tumorigenic and invasive characteristics.
The enhancement of IR induced tumorigenesis by the estrous cycle may be replicated or further enhanced by exogenous endocrine disrupting chemicals. Indeed, evidence suggests that BPA (and presumably other estrogenic chemicals) exposure in utero can increase the mammary gland’s response to progesterone during puberty (Brisken, Hess et al. 2015). This enhancement would presumably also increase the risk of breast cancer from ionizing radiation, since that risk increases with estrogen exposure and the number of menstrual cycles.
Uncertainty arising from extrapolating from rodent and human in vitro studies to human biology
Uncertainty in this pathway arises from inconsistencies in carcinogenesis between rodent and mouse species and strains and from incomplete information about the same mechanisms operating in humans. This raises questions about whether all evidence should be weighted equally.
Almost half of the data included here is from in vitro experiments on human primary or cultured cells, which should have a high degree of relevance for this pathway in humans. However, most of the human cells are not from mammary gland, and most of the mammary gland derived cells are cancer or immortalized cells that will not respond in exactly the same way as primary cells. Even this human data should therefore be interpreted with some caution.
Most of the remaining data in this AOP is from mice, with a relatively small number of rat studies. As a breast cancer model, mice share important characteristics with humans (Medina 2007; Imaoka, Nishimura et al. 2009). Mice and humans share similar epithelial cell types (Lim, Wu et al. 2010) and a similar developmental regime with the bulk of epithelial development occurring postnatally and accelerating during puberty, with differentiation during pregnancy (Medina 2007). Tumors in humans originate in the terminal ductal lobular unit, a structure that includes the lobule with secretory alveoli and the start of the collecting duct. The developmental terminal end bud structure is thought to be particularly vulnerable to carcinogens because of the presence of stem cells and proliferation, although it is not the only possible site of initiation. Similarly, tumors in mice originate in predominantly in alveoli as well as terminal end buds and small ducts (Medina 2007). Humans are more susceptible to carcinogens around puberty, and pregnancy is protective. Evidence on the role of development and reproduction in mammary carcinogenesis in mice is limited compared with rats but is consistent with sensitivity to radiation around puberty (Imaoka, Nishimura et al. 2009), and parity is protective for chemical carcinogens (Medina 2007). In addition, proliferation contributes to carcinogenesis in both mice and humans (Medina 2007).
However, mice differ from humans in some notable ways (Medina 2007). Mammary tumors are not common in mice, so susceptible strains or tumor-promoting viruses are used to increase spontaneous incidence and response to carcinogenic stimuli. This difference may be partially attributable to hormone responsiveness of tumors. Although tumors in mice depend on hormones for development, breast cancers in rats and humans are frequently hormone receptor positive, while mammary tumors in mice are not (Nandi, Guzman et al. 1995; Medina 2007; Imaoka, Nishimura et al. 2009).
Medina, D. (2007). "Chemical carcinogenesis of rat and mouse mammary glands." Breast Dis 28: 63-68.
Essentiality of the Key Events
Essentiality of the key events
IR appears to be a “complete” carcinogen in the mammary gland in that the toxin acts as an initiator through the formation of oxidative stress and pro-mutagenic DNA damage and (the MIEs) and as a promoter through increasing inflammation and proliferation, similar to many chemical carcinogens (Russo and Russo 1996). We have high confidence in the evidence linking stressor (IR) with adverse outcome (breast cancer). The weight of evidence for the first pathway from RONS to DNA damage, Mutation, and Proliferation is High while the weight of evidence for the second pathway from RONS to Inflammation to Proliferation and Breast Cancer is Moderate. These evaluations are based on the supporting evidence for all KEs and the considerations in Annex 1, and based on the need for additional evidence in the essentiality of Inflammation for the genesis of breast cancer.
|
Defining question |
High (Strong) |
Moderate |
Low (Weak) |
|
|
Support for essentiality of KEs |
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from specifically designed experimental studies illustrating essentiality for at least one of the important KEs |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE |
No or contradictory experimental evidence of the essentiality of any of the KEs. |
|
MIE1: Increase in reactive oxygen and nitrogen species (RONS) |
Essentiality is High. The most significant support comes from the relatively large number of studies using antioxidants or other interventions to reduce RONS, which show a reduction in DNA damage and mutations. Additional support comes from experiments increasing external oxidants like H2O2, which show that RONS are independently capable of causing DNA damage and mutations. Uncertainties arise from the smaller effects of RONS on DNA damage compared with ionizing radiation. Mammary gland relevance is less certain due to the relatively few experiments in breast tissue. |
|||
|
MIE2: Increase in DNA damage |
Essentiality is High. The essentiality of this MIE to cancer is generally accepted. Supporting evidence comes from application of mutagenic agents: the increase in DNA damage precedes mutations, proliferation, and tumorigenesis. Further indirect evidence comes from evidence for MIE1, in which antioxidants that reduce DNA damage also reduce mutations and chromosomal damage. Finally, mutations in DNA repair genes increase the risk of tumors. |
|||
|
KE1: Increase in mutation |
Essentiality is High. The contribution of this MIE to cancer is generally accepted. Evidence comes from knock-out and knock-in experiments, which find that mutations in certain key genes increase tumorigenesis. However, an ongoing debate pits the singular importance of mutations against a significant role for the tissue microenvironment. This debate is fueled by transplant studies that show the importance of tissue environment for tumorigenesis and suggesting that mutations may not be sufficient for tumorigenesis. |
|||
|
KE2: Increase in proliferation |
Essentiality is High. Evidence comes from transplant experiments showing that non-proliferating tissue is less tumorigenic than proliferating lesions, and from interventions that reduce both proliferation and tumors. Further evidence comes from animals that are resistant to both mammary gland proliferation and tumors from ionizing radiation. Uncertainty arises from conflicting evidence on the tumorigenicity of hyperplasia, the absence of hyperplasia observed before some tumors, and spontaneous regression of tumors. |
|||
|
KE3: Increase in inflammation |
Essentiality is Moderate. Evidence comes from using genetic modifications, antibodies, and antioxidants to reduce inflammatory and anti-inflammatory factors. These interventions reduce DNA damage, mutations, and mechanisms contributing to tumorigenesis and invasion. Uncertainty arises from conflicting effects in different genetic backgrounds and in different organs. |
|||
Evidence Assessment
IR appears to be a “complete” carcinogen in the mammary gland in that the toxin acts as an initiator through the formation of oxidative stress and pro-mutagenic DNA damage and (the MIEs) and as a promoter through increasing inflammation and proliferation, similar to many chemical carcinogens (Russo and Russo 1996). We have high confidence in the evidence linking stressor (IR) with adverse outcome (breast cancer). The weight of evidence for the first pathway from RONS and DNA damage to Mutation and Proliferation is High while the weight of evidence for the second pathway from RONS to Inflammation to Proliferation and Breast Cancer is Moderate. These evaluations are based on the supporting evidence for all KEs and the considerations in Annex 1, and based on the need for additional evidence in the essentiality of Inflammation for the genesis of breast cancer.
This AOP could not address the large number of related topics that interact with the key events described here. These topics include events following IR that may interact with these key events such as immune surveillance (which may change with the inflammatory environment after IR (Schreiber, Old et al. 2011; Barcellos-Hoff 2013; Lumniczky and Safrany 2015); IR effect on survival/apoptosis and interactions of apoptosis with inflammation, mutation, compensatory proliferation, and selection process; changes to DNA repair; and the role of epigenetics in carcinogenesis from IR (Daino, Nishimura et al. 2018). This AOP also does not address other influences on these key events beyond reproductive hormones and typical breast development. Subsequent contributions to this AOP should elaborate on these points.
|
Defining question |
High (Strong) |
Moderate |
Low (Weak) |
|||
|
2. Support for essentiality of KEs |
Are downstream KEs and/or the AO prevented if an upstream KE is blocked? |
Direct evidence from specifically designed experimental studies illustrating essentiality for at least one of the important KEs |
Indirect evidence that sufficient modification of an expected modulating factor attenuates or augments a KE |
No or contradictory experimental evidence of the essentiality of any of the KEs. |
||
|
MIE: Increase in DNA damage |
Essentiality is High. The essentiality of this MIE to cancer is generally accepted. Supporting evidence comes from application of mutagenic agents: the increase in DNA damage precedes mutations, proliferation, and tumorigenesis. Further indirect evidence comes from evidence for MIE1, in which antioxidants that reduce DNA damage also reduce mutations and chromosomal damage. Finally, mutations in DNA repair genes increase the risk of tumors. |
|||||
|
KE/AO: Increase in mutation |
Essentiality is High. The contribution of this MIE to cancer is generally accepted. Evidence comes from knock-out and knock-in experiments, which find that mutations in certain key genes increase tumorigenesis. However, an ongoing debate pits the singular importance of mutations against a significant role for the tissue microenvironment. This debate is fueled by transplant studies that show the importance of tissue environment for tumorigenesis and suggesting that mutations may not be sufficient for tumorigenesis. |
|||||
|
KE: Increase, Cell Proliferation (epithelial cells) |
Essentiality is High. Cellular proliferation is a key characteristic of cancer cells and can lead to hyperplasia, an intermediate phase in the development of tumorigenesis. Proliferation also increases the number of cells with mutations, which can further promote proliferation and/or changes to the local microenvironment. |
|||||
|
KE/AO: Increase, Ductal Hyperplasia |
Essentiality is High. Evidence comes from transplant experiments showing that non-proliferating tissue is less tumorigenic than proliferating lesions, and from interventions that reduce both proliferation and tumors. Further evidence comes from animals that are resistant to both mammary gland proliferation and tumors from ionizing radiation. Uncertainty arises from conflicting evidence on the tumorigenicity of hyperplasia, the absence of hyperplasia observed before some tumors, and spontaneous regression of tumors. |
|||||
|
KEs: Tissue Resident Cell Activation, Increased Pro-inflammatory mediators, Leukocyte recruitment/activation |
Essentiality is Moderate. These key events were reviewed as a group. Evidence comes from using genetic modifications, antibodies, and antioxidants to reduce inflammatory and anti-inflammatory factors. These interventions reduce DNA damage, mutations, and mechanisms contributing to tumorigenesis and invasion. Uncertainty arises from conflicting effects in different genetic backgrounds and in different organs. |
|||||
|
KE: Increase in reactive oxygen and nitrogen species (RONS) |
Essentiality is High. The most significant support comes from the relatively large number of studies using antioxidants or other interventions to reduce RONS, which show a reduction in DNA damage and mutations. Additional support comes from experiments increasing external oxidants like H2O2, which show that RONS are independently capable of causing DNA damage and mutations. Uncertainties arise from the smaller effects of RONS on DNA damage compared with ionizing radiation. Mammary gland relevance is less certain due to the relatively few experiments in breast tissue. |
|||||
|
Defining question |
High (Strong) |
Moderate |
Low (Weak) |
|||
|
1. Support for biological plausibility of KERs |
a. Is there a mechanistic relationship between KEup and KEdown consistent with established biological knowledge? |
Extensive understanding of the KER based on extensive previous documentation and broad acceptance |
KER is plausible based on analogy to accepted biological relationships, but scientific understanding is incomplete |
Empirical support for association between KEs, but the structural or functional relationship between them is not understood. |
||
|
MIE DNA damage leads to Increase in Mutations (intermediate AO) |
High. DNA damage in the form of nucleotide damage, single strand and double strand breaks, and complex damage can generate mutations, particularly when a damaged cell undergoes replication. |
|||||
|
Increase in Mutations leads to Increase, Cell Proliferation (epithelial cells) |
High. Multiple mechanisms limit the proliferation of cells in normal biological systems. Mutations in many of the genes controlling these mechanisms promote proliferation. |
|||||
|
Increase, Cell Proliferation (epithelial cells) leads to Increase in Mutation |
High. Biological plausibility is high since proliferation is generally acknowledged to increase mutations through incorporating or amplifying the impact of unrepaired DNA damage as mutations. |
|||||
|
Increase, Cell Proliferation (epithelial cells) leads to Increase, Ductal Hyperplasia (intermediate AO) |
Not Specified. This relationship is generally accepted, but evidence has not been compiled for this AOP. |
|||||
|
Increase, Ductal Hyperplasia increases risk of Breast Cancer (AO) |
High. It is generally accepted that proliferation and hyperplasia increase the risk of breast cancer. Evidence comes from transplant experiments showing that non-proliferating tissue is less tumorigenic than proliferating lesions, and from interventions that reduce both lesions and tumors. Further evidence comes from animals that are resistant to both mammary gland hyperplasia and tumors from ionizing radiation. Uncertainty arises from conflicting evidence on the tumorigenicity of hyperplasia, the absence of hyperplasia observed before some tumors, and spontaneous regression of tumors. |
|||||
|
Increase in DNA damage leads to Tissue resident cell activation |
High. Biological plausibility is high since DNA damage generates inflammatory signals (DAMPS). |
|||||
|
Tissue resident cell activation leads to Increase in Pro-inflammatory mediators |
High. This relationship is well established. |
|||||
|
Pro-inflammatory mediators lead to Leukocyte recruitment/activation |
High. This relationship is well established. |
|||||
|
Pro-inflammatory mediators, Laukocyte recruitment/ activation lead to an Increase in RONS |
High. Inflammation is commonly understood to generate RONS via inflammatory signaling and activated immune cells. |
|||||
|
Increase in RONS leads to Increase in DNA damage (intermediate AO) |
High. Reactive oxygen and nitrogen species from oxygen and respiratory activity are generally acknowledged to damage DNA under a range of cellular conditions. |
|||||
|
Increase in RONS leads to Tissue resident cell activation |
Moderate. Damage from RONS can activate some inflammatory and anti-inflammatory pathways (TLR, TGF-β), and RONS are an essential part of the primary signaling pathways of multiple inflammatory and anti-inflammatory pathways (TLR4, TNF-a, TGF-β, NFkB). |
|||||
|
Pro-inflammatory mediators lead to Increase, Cell Proliferation (epithelial cells) |
High. Inflammation is generally understood to lead to proliferation during recovery from inflammation. |
|||||
|
Pro-inflammatory mediators promote Breast Cancer |
Moderate. Tissue environment is known to be a major factor in carcinogenesis, and inflammatory processes are implicated in the development and invasiveness of breast and other cancers. |
|||||
|
Defining questions |
High (Strong) |
Moderate |
Low (Weak) |
|||
|
3. Empirical support for KERs |
Does empirical evidence support that a change in KEup leads to an appropriate change in KEdown? Does KEup occur at lower doses and earlier time points than KE down and is the incidence of KEup > than that for KEdown? Inconsistencies? |
Multiple studies showing dependent change in both events following exposure to a wide range of specific stressors. No or few critical data gaps or conflicting data |
Demonstrated dependent change in both events following exposure to a small number of stressors. Some inconsistencies with expected pattern that can be explained by various factors. |
Limited or no studies reporting dependent change in both events following exposure to a specific stressor; and/or significant inconsistencies in empirical support across taxa and species that don’t align with hypothesized AOP |
||
|
MIE DNA damage leads to Increase in Mutations (intermediate AO) |
High. It is generally accepted that DNA damage leads to mutations. Empirical support comes in part from the observation that agents which increase DNA damage also cause mutations, that DNA damage precedes the appearance of mutations, and that interventions to reduce DNA damage also reduce mutations. None of the identified studies measure both outcomes over the same range of time points. This constitutes a readily addressable data gap. |
|||||
|
Increase in Mutations leads to Increase, Cell Proliferation (epithelial cells) |
Moderate. Mutations that promote proliferation are frequently found in cancers, and both mutation and proliferation occur in response to tumorigenic stressors like ionizing radiation. Although not measured together after stressors, mutations appear over the same time frame or prior to the appearance of proliferation. Multiple uncertainties and conflicting evidence weaken this key event relationship. The two key events differ in their dose response- mutation but not proliferation increases with ionizing radiation dose. Furthermore, a single mutation is not necessarily sufficient to increase proliferation- proliferation typically requires multiple mutations or a change in the surrounding environment. In mammary tissue, stromal state strongly influences the proliferative nature of epithelial cells – even epithelial cells with mutated tumor suppressors may be unable to form tumors in the absence of stromal changes. |
|||||
|
Increase, Cell Proliferation (epithelial cells) leads to Increase in Mutation |
High. We did not evaluate the empirical support for this KER in response to IR. However proliferation or mitosis is required for some types of DNA damage to be made permanent and heritable, and further DNA damage including mutation promoting double strand breaks can occur when cells divide before DNA repair is complete. |
|||||
|
Increase, Cell Proliferation (epithelial cells) leads to Increase, Ductal Hyperplasia (intermediate AO) |
Not specified. This relationship is generally accepted, but evidence has not been compiled for this AOP. |
|||||
|
Increase, Ductal Hyperplasia increases risk of Breast Cancer (AO) |
High. Carcinogenic agents increase proliferation and hyperplasia as well as tumors. Proliferation and hyperplasia appear prior to or at the same time as tumors, grow into carcinomas, and form mammary tumors more effectively than non-proliferating tissue. Disruption of proliferation is associated with decreased tumor growth, and tumor resistant rats do not show proliferation. However, the discrepancy between the non-linear proliferative and linear mammary tumor response to carcinogen dose coupled with evidence of independent occurrences of proliferation and tumorigenesis suggests that while proliferation and hyperplasia likely promote carcinogenesis, additional factors also contribute to carcinogenesis. |
|||||
|
Increase in DNA damage leads to Tissue resident cell activation |
Not Specified. Biological plausibility is high since DNA damage generates inflammatory signals (DAMPS), but evidence has not been compiled for this AOP. |
|||||
|
Tissue resident cell activation leads to Increase in Pro-inflammatory mediators |
Not specified. This relationship is generally accepted, but empirical evidence has not been compiled for this KER in mammary gland. |
|||||
|
Pro-inflammatory mediators lead to Leukocyte recruitment/activation |
Not specified. This relationship is generally accepted, but evidence has not been compiled for this KER in mammary gland. |
|||||
|
Increase in Pro-inflammatory mediators and Leukocyte recruitment/ activation leads to an Increase in RONS lead to an Increase in RONS |
High. Signals arising from inflammation can be both pro- and anti-inflammatory, and both can have effects on RONS and downstream key events. Multiple inflammation-related factors increase RONS or oxidative damage, and ionizing radiation increases both inflammation-related signaling and RONS or oxidative damage over the same time points. Interventions to reduce inflammation also reduce RONS. The dose-dependence of the response to stressors is generally consistent between the two key events, although this is based on a small number of studies with some conflicting evidence. |
|||||
|
Increase in RONS leads to Increase in DNA damage (intermediate AO) |
High. Multiple studies show an increase in DNA damage with RONS treatment as well as dependent changes in both RONS and DNA damage in response to stressors. DNA damage increases with RONS dose, and temporal concordance between RONS and DNA damage events following ionizing radiation is consistent with a causative relationship, although few studies examine multiple doses and time points. A small number of studies do not find double strand breaks at physiological doses, or report an increase in one key event but not the other. |
|||||
|
Increase in RONS leads to Tissue resident cell activation |
Moderate. Both RONS and inflammation increase in response to agents that increase either RONS or inflammation. Multiple studies show dose-dependent changes in both RONS and inflammation in response to stressors including ionizing radiation and antioxidants. RONS have been measured at the same or earlier time points as inflammatory markers, but additional studies are needed to characterize the inflammatory response at the earliest time points to support causation. Uncertainties come from the positive feedback from inflammation to RONS potentially interfering with attempts to establish causality, and from the large number of inflammation related factors with differing responses to stressors and experimental variation. |
|||||
|
Pro-inflammatory mediators lead to Increase, Cell Proliferation (epithelial cells) |
Not Specified. We did not evaluate the empirical support for this KER in response to IR. However, inflammation is generally understood to promote proliferation and survival |
|||||
|
Pro-inflammatory mediators promote Breast Cancer |
Moderate. Interventions to increase inflammatory factors increase the carcinogenic potential of targeted and non-targeted cells. Inflammation is documented at earlier time points than tumorigenesis or invasion- within minutes or hours compared to days to months for carcinogenesis, consistent with an inflammatory mechanism of tumorigenesis and invasion. Inhibition of cytokines, inflammatory signaling pathways, and downstream effectors of inflammation activity prevent transformation, tumorigenesis, and invasion following IR or stimulation of inflammatory pathways. However, the key event and the adverse outcome differ in their dose-response to ionizing radiation: inflammation always does not increase linearly with dose, while breast cancer and invasion does. Uncertainty arises from the multifunctional nature of inflammation-related pathways which may be pro- or anti-inflammatory and pro- or anti-carcinogenic based on context. Both pro- and anti-inflammatory factors may contribute to carcinogenesis- further research will be required to identify the context of each. |
|||||
Supporting Information for Essentiality Call to be Included in Annex 1:
Overall assessment of KEs
IR appears to be a “complete” carcinogen in the mammary gland in that the stressor acts as an initiator through the formation of oxidative stress and pro-mutagenic DNA damage and (the MIEs) and as a promoter through increasing inflammation and proliferation, similar to many chemical carcinogens (Russo and Russo 1996). We have high confidence in the evidence linking stressor (IR) with adverse outcome (breast cancer). The weight of evidence for the first pathway from RONS to DNA damage, Mutation, and Proliferation is High while the weight of evidence for the second pathway from RONS to Inflammation to Proliferation and Breast Cancer is Moderate. These evaluations are based on the supporting evidence for all KEs and the considerations in Annex 1, and based on the need for additional evidence in the essentiality of Inflammation for the genesis of breast cancer.
MIE: Increase in DNA damage
Essentiality is High. The essentiality of this MIE to cancer is generally accepted. Supporting evidence comes from application of mutagenic agents: the increase in DNA damage precedes mutations, proliferation, and tumorigenesis. Further indirect evidence comes from evidence for MIE1, in which antioxidants that reduce DNA damage also reduce mutations and chromosomal damage. Finally, mutations in DNA repair genes increase the risk of tumors.
Increases or decreases in DNA damage are associated with corresponding increases or decreases in downstream key events in the pathway to breast cancer. An external agent (ionizing radiation) that increases DNA damage (Padula, Ponzinibbio et al. 2016) also causes chromosomal damage and increased mutations (Sandhu and Birnboim 1997; Jones, Riggs et al. 2007; Denissova, Nasello et al. 2012; Fibach and Rachmilewitz 2015), transforms cells (Yang, Craise et al. 1992; Yang, Georgy et al. 1997; Unger, Wienberg et al. 2010), and causes tumors (Poirier and Beland 1994; Little 2009). Polymorphisms or mutations in DNA repair genes affect tumor formation after ionizing radiation in animals (Yu, Okayasu et al. 2001; Umesako, Fujisawa et al. 2005) and in people (Millikan, Player et al. 2005; Andrieu, Easton et al. 2006; Broeks, Braaf et al. 2007; Bernstein, Haile et al. 2010; Brooks, Teraoka et al. 2012; Pijpe, Andrieu et al. 2012; Bernstein, Thomas et al. 2013). Consistent with these findings, antioxidants that reduce DNA damage from stressors like IR also reduce chromosomal aberrations and micronuclei arising from those stressors (Azzam, De Toledo et al. 2002; Choi, Kang et al. 2007; Jones, Riggs et al. 2007).
Evidence in mammary gland
The majority of research on the effects of IR on DNA damage has been performed in tissues other than mammary gland, but several studies suggest that effects in the mammary gland (and its consequences) would be consistent with other tissues. Oxidative DNA damage in mammary cells increases immediately after exposure to IR (Haegele, Wolfe et al. 1998), and double stranded breaks, micronuclei, and (later) chromosomal aberrations appear two hours to six days after IR exposure in vivo and in vitro (Soler, Pampalona et al. 2009; Snijders, Marchetti et al. 2012; Hernandez, Terradas et al. 2013). Genomic instability was reported in genetically susceptible cells after a month of higher doses of IR (4 doses of 1.8 Gy but not 0.75 Gy) (Snijders, Marchetti et al. 2012).
KE/AO: Increase in mutation
Essentiality is High. The contribution of this MIE to cancer is generally accepted. Evidence comes from knock-out and knock-in experiments, which find that mutations in certain key genes increase tumorigenesis. However, an ongoing debate pits the singular importance of mutations against a significant role for the tissue microenvironment. This debate is fueled by transplant studies that show the importance of tissue environment for tumorigenesis and suggesting that mutations may not be sufficient for tumorigenesis.
Mutations increase transformation in culture (Wang, Su et al. 2011) and proliferation and tumors in mice (Radice, Ferreira-Cornwell et al. 1997; Umesako, Fujisawa et al. 2005; de Ostrovich, Lambertz et al. 2008; Podsypanina, Politi et al. 2008; Francis, Bergsied et al. 2009; Gustin, Karakas et al. 2009; Francis, Chakrabarti et al. 2011; Tao, Xiang et al. 2017). Restoring function in mutated genes regresses tumors in animals (Martins, Brown-Swigart et al. 2006; Podsypanina, Politi et al. 2008). Mutations are common in tumors (Haag, Hsu et al. 1996; Greenman, Stephens et al. 2007; Stratton, Campbell et al. 2009; CGAN (Cancer Genome Atlas Network) 2012; Vandin, Upfal et al. 2012; Garraway and Lander 2013; Vogelstein, Papadopoulos et al. 2013; Yang, Killian et al. 2015) and tumors are largely clonal, suggesting that individual mutations offer the tumor evolutionary advantages (Wang, Waters et al. 2014; Yates, Gerstung et al. 2015; Begg, Ostrovnaya et al. 2016).
Evidence in mammary gland
Many of the studies in support of the proliferative and tumorigenic role of mutations are in mammary gland or breast cancers. Further support for including DNA damage and mutation in the mechanistic pathway linking ionizing radiation with breast cancer comes from the observation that variants in DNA repair genes increase the risk of mammary tumors in animals after IR (Yu, Okayasu et al. 2001; Umesako, Fujisawa et al. 2005) and increase breast cancer after IR (Millikan, Player et al. 2005; Andrieu, Easton et al. 2006; Broeks, Braaf et al. 2007; Bernstein, Haile et al. 2010; Brooks, Teraoka et al. 2012; Pijpe, Andrieu et al. 2012; Bernstein, Thomas et al. 2013). BRCA is perhaps the best known DNA repair gene linked with breast cancer risk, and several studies of these studies have suggested a link between BRCA mutation status and increased susceptibility to breast cancer following ionizing radiation, particularly in women exposed at younger ages (Pijpe, Andrieu et al. 2012).
Uncertainties or Inconsistencies
Mutations alone are not sufficient or even essential for tumor growth in mammary glands. Mammary tumor incidence following ionizing radiation varies significantly by sex and depends on the presence of ovarian hormones (Cronkite, Shellabarger et al. 1960; Segaloff and Maxfield 1971; Shellabarger, Stone et al. 1976; Holtzman, Stone et al. 1979; Holtzman, Stone et al. 1981; Welsch, Goodrich-Smith et al. 1981; Clifton, Yasukawa-Barnes et al. 1985; Solleveld, van Zwieten et al. 1986; Broerse, Hennen et al. 1987; Lemon, Kumar et al. 1989; Inano, Suzuki et al. 1991; Inano, Suzuki et al. 1996; Peterson, Servinsky et al. 2005). Tumor growth from transplanted tumor cells varies with age, parity, and lactational status (Maffini, Calabro et al. 2005; McDaniel, Rumer et al. 2006), and stroma treated with carcinogens or IR supports tumors from pre-malignant epithelial cells (Barcellos-Hoff and Ravani 2000; Maffini, Soto et al. 2004; Nguyen, Oketch-Rabah et al. 2011). While the mechanisms underlying these contextual factors have not been clearly identified, the proliferative effect of hormones on the mammary gland may serve to amplify damaged and mutated cells and modify the stromal environment to increase the likelihood of cellular transformation. Inflammatory responses including the release of cytokines and the activation of inflammatory and anti-inflammatory signaling pathways likely also amplify the effects of DNA damage and mutations through many of the same mechanisms.
KE: Increase in proliferation
Essentiality is High. Cellular proliferation is a key characteristic of cancer cells (Hanahan and Weinberg 2011) and can lead to hyperplasia, an intermediate phase in the development of tumorigenesis. Proliferation also increases the number of cells with mutations, which can further promote proliferation and/or changes to the local microenvironment.
Evidence in mammary gland
Multiple studies show that mammary gland proliferates after IR or chemical carcinogen treatment prior to the appearance of mammary tumors. Epithelial cells proliferate following IR in vitro (Mukhopadhyay, Costes et al. 2010) and in vivo (Nguyen, Oketch-Rabah et al. 2011; Snijders, Marchetti et al. 2012; Suman, Johnson et al. 2012; Tang, Fernandez-Garcia et al. 2014). Increasing proliferation leads to hyperplasia (Korkaya, Paulson et al. 2009). Proliferative nodules and hyperplasia appear in mammary terminal end bud, alveolae, and ducts of rats and mice after exposure to chemical carcinogens (Beuving, Bern et al. 1967; Beuving, Faulkin et al. 1967; Russo, Saby et al. 1977; Purnell 1980) and ionizing radiation (Faulkin, Shellabarger et al. 1967; Ullrich and Preston 1991; Imaoka, Nishimura et al. 2006). Proliferating foci precede the development of tumors (Haslam and Bern 1977; Purnell 1980) and form tumors more effectively than non-proliferating tissue (Deome, Faulkin et al. 1959; Beuving 1968; Rivera, Hill et al. 1981).
Supporting the essentiality of these proliferative processes to tumorigenesis, ACI rats that exhibit no mammary proliferation or hyperplasia following IR are resistant to tumors following IR (Kutanzi, Koturbash et al. 2010). Interventions reducing proliferation in susceptible PyVT and BALB/c mice also reduce mammary tumors (Luo, Fan et al. 2009; Connelly, Barham et al. 2011; Tang, Fernandez-Garcia et al. 2014).
Uncertainties or Inconsistencies
Some studies report carcinogenesis in the absence of hyperplasia (Sinha and Dao 1974) and others do not find increased tumorigenesis from transplanted hyperplasia (Beuving, Bern et al. 1967; Haslam and Bern 1977; Sinha and Dao 1977). The failure of some proliferative foci to form tumors and the regression of some tumors when formed (Haslam and Bern 1977; Purnell 1980; Korkola and Archer 1999) suggests that proliferation may not be sufficient for sustained tumorigenesis in mammary gland.
KE/AO: Increase, ductal hyperplasia
Essentiality is High. Evidence comes from transplant experiments showing that non-proliferating tissue is less tumorigenic than proliferating lesions, and from interventions that reduce both proliferation and tumors. Further evidence comes from animals that are resistant to both mammary gland proliferation and tumors from ionizing radiation. Uncertainty arises from conflicting evidence on the tumorigenicity of hyperplasia, the absence of hyperplasia observed before some tumors, and spontaneous regression of tumors.
Hyperplasia signals the presence of excess proliferation (a key characteristic of cancer cells (Hanahan and Weinberg 2011)) and represents an intermediate phase in the development of tumorigenesis.
Evidence in mammary gland
Multiple studies show that mammary gland proliferates after IR or chemical carcinogen treatment prior to the appearance of mammary tumors. Proliferative nodules and hyperplasia appear in mammary terminal end bud, alveolae, and ducts of rats and mice after exposure to chemical carcinogens (Beuving, Bern et al. 1967; Beuving, Faulkin et al. 1967; Russo, Saby et al. 1977; Purnell 1980) and ionizing radiation (Faulkin, Shellabarger et al. 1967; Ullrich and Preston 1991; Imaoka, Nishimura et al. 2006). Proliferating foci precede the development of tumors (Haslam and Bern 1977; Purnell 1980) and form tumors more effectively than non-proliferating tissue (Deome, Faulkin et al. 1959; Beuving 1968; Rivera, Hill et al. 1981). Adenocarcinomas in rats appear to preferentially form from terminal end bud hyperplasia (Haslam and Bern 1977; Russo, Saby et al. 1977; Purnell 1980), similar to the origin of many breast cancers for humans and for some mice after IR (Medina and Thompson 2000).
Supporting the essentiality of these proliferative processes to tumorigenesis, ACI rats that exhibit no mammary proliferation or hyperplasia following IR are resistant to tumors following IR (Kutanzi, Koturbash et al. 2010). Interventions reducing proliferation in susceptible PyVT and BALB/c mice also reduce mammary tumors (Luo, Fan et al. 2009; Connelly, Barham et al. 2011).
Uncertainties or Inconsistencies
Some studies report carcinogenesis in the absence of hyperplasia (Sinha and Dao 1974) and others do not find increased tumorigenesis from transplanted hyperplasia (Beuving, Bern et al. 1967; Haslam and Bern 1977; Sinha and Dao 1977). The failure of some lesions to form tumors and the regression of some tumors when formed (Haslam and Bern 1977; Purnell 1980; Korkola and Archer 1999) suggests that hyperplasia alone may not be sufficient for sustained tumorigenesis in mammary gland.
KEs: Tissue resident cell activation, Increase, Pro-inflammatory mediators, Leukocyte Recruitment/Activation
Essentiality is Moderate. These key events were reviewed as a group. Evidence comes from using genetic modifications, antibodies, and antioxidants to reduce inflammatory and anti-inflammatory factors. These interventions reduce DNA damage, mutations, and mechanisms contributing to tumorigenesis and invasion. Uncertainty arises from conflicting effects in different genetic backgrounds and in different organs.
Tumors and tumor cells exhibit features of inflammation, and inflammation is generally understood to promote transformation and tumor progression by supporting multiple hallmarks of cancer including oxidative activity and DNA damage, survival and proliferation, angiogenesis, and invasion and metastasis (Iliopoulos, Hirsch et al. 2009; Hanahan and Weinberg 2011; Esquivel-Velazquez, Ostoa-Saloma et al. 2015).
Many of these cancer promoting effects of inflammation can be seen following exposure to ionizing radiation (Bisht, Bradbury et al. 2003; Elahi, Suraweera et al. 2009; Nguyen, Oketch-Rabah et al. 2011; Bouchard, Bouvette et al. 2013; Nguyen, Fredlund et al. 2013; Illa-Bochaca, Ouyang et al. 2014). Inflammatory pathways are commonly activated in breast and mammary cancers following IR (Nguyen, Oketch-Rabah et al. 2011; Nguyen, Fredlund et al. 2013; Illa-Bochaca, Ouyang et al. 2014). Polymorphisms in inflammation genes are associated with breast cancer risk from IR in radiation technologists (Schonfeld, Bhatti et al. 2010) and with susceptibility to intestinal adenoma following IR in mice (Elahi, Suraweera et al. 2009). Cytokines TGF-β and IL6 transform primary human mammospheres and pre-malignant mammary epithelial cell lines in vitro and make them tumorigenic in vivo (Sansone, Storci et al. 2007; Iliopoulos, Hirsch et al. 2009; Nguyen, Oketch-Rabah et al. 2011), and inflammation related factors COX2 and TGF-β are required for the full effect of IR on DNA damage and transformation in vitro and mammary tumor growth and invasion in vivo (Bisht, Bradbury et al. 2003; Nguyen, Oketch-Rabah et al. 2011).
One mechanism of cancer promotion involves oxidative activity and DNA damage: inflammation in response to IR increases oxidative activity in a positive feedback loop leading to increased DNA lesions and mutations. Oxidative activity mediates the increase in inflammatory markers (TNF-a and neutrophil markers) in bladder and kidney (Ozyurt, Cevik et al. 2014), and TNF-a and neutrophils increase oxidative activity (Jackson, Gajewski et al. 1989; Stevens, Bucurenci et al. 1992; Zhang, Zhu et al. 2017). Inflammatory activity from neutrophils and TNF-a and NF-kB-dependent COX2 and NO damage DNA and increase mutations by increasing oxidative activity (Jackson, Gajewski et al. 1989; Zhou, Ivanov et al. 2005). The mutations can be reduced by blocking the inflammatory factors NF-kB, COX2, TNF-a, or nitric oxide, or with antioxidants (Jackson, Gajewski et al. 1989; Zhou, Ivanov et al. 2005; Zhou, Ivanov et al. 2008; Zhang, Zhu et al. 2017). Antibodies to TNF-a or TGF-β reduce DNA damage in bone marrow (Burr, Robinson et al. 2010; Rastogi, Coates et al. 2012) and CHO cells (Han, Chen et al. 2010). Inhibiting TNF-a also reduces genomic instability in directly irradiated (but not bystander) lymphocytes (Moore, Marsden et al. 2005) and in bone marrow of CBA/Ca mice susceptible to IR-induced leukemia but not resistant C57BL/6 mice (Lorimore, Mukherjee et al. 2011). Inhibiting inflammatory factors NF-kB or iNOS reduces IR-induced bystander mutations in lung fibroblasts (Zhou, Ivanov et al. 2008).
Inflammatory pathways activated by IR are also capable of promoting tumor growth and metastasis. Exposure to IR or RONS sensitizes mammary epithelial cells to respond to TGF-β - which is widely activated by IR (Ehrhart, Segarini et al. 1997). IR and TGF-β signaling leads to an epithelial to mesenchymal (EMT)-like transition, which disrupts the expression and distribution of cell adhesion molecules and multicellular organization and promotes invasion (Park, Henshall-Powell et al. 2003; Andarawewa, Erickson et al. 2007; Andarawewa, Costes et al. 2011; Iizuka, Sasatani et al. 2017). This mechanism resembles wound healing (Koh and DiPietro 2011; Perez, Vago et al. 2014; Landen, Li et al. 2016), but also resembles malignancy - invasive breast cancer cell lines overexpress TGF-β and respond to TGF-β with increased invasion (Kim, Kim et al. 2004; Gomes, Terra et al. 2012).
The response to TGF-β likely involves an increase in senescence in fibroblasts. IR-induced senescence releases a suite of signaling molecules including pro-inflammatory IL6 and proteases (MMPs) (Tsai, Chuang et al. 2005; Liakou, Mavrogonatou et al. 2016; Perrott, Wiley et al. 2017). The signaling molecules released by IR-senescent fibroblasts promote the disorganized tissue structure of mammary epithelial cells and the growth, EMT, and invasion of breast cancer epithelial cells or mutant epithelial cells (Tsai, Chuang et al. 2005; Liakou, Mavrogonatou et al. 2016; Perrott, Wiley et al. 2017) and 3D mammary tumor models (Sourisseau, Harrington et al. 2011). The induction of senescence in fibroblasts by IR requires TGF-β (Liakou, Mavrogonatou et al. 2016), and the release of the pro-invasive signaling molecules involves an IL-1 dependent activation of NF-kB (Perrott, Wiley et al. 2017). Senescence following IR also selects for a post-senescent variant of epithelial cell that is more conducive to tumorigenesis (Mukhopadhyay, Costes et al. 2010).
Il6 may play an important function in the carcinogenic response to IR. IL6 is expressed in mouse mammary gland after IR (Bouchard, Bouvette et al. 2013). IL6 is produced by IR-senescent fibroblasts, but may also be expressed by epithelial cells after IR since primary human mammospheres and pre-malignant mammary epithelial cell lines respond to IL6 with increased IL6 expression (Sansone, Storci et al. 2007; Iliopoulos, Hirsch et al. 2009). IL6 promotes the mobility and tumorigenesis of normal and breast cancer epithelial cells (Sansone, Storci et al. 2007; Sasser, Sullivan et al. 2007; Studebaker, Storci et al. 2008; Iliopoulos, Hirsch et al. 2009; Iliopoulos, Jaeger et al. 2010). This activity depends on transcription factor NOTCH3, which supports the renewal of stem-like cell populations (Sansone, Storci et al. 2007), and NOTCH has been implicated in multiple other studies in the proliferative response to IR in mammary epithelia (Nguyen, Oketch-Rabah et al. 2011; Marusyk, Tabassum et al. 2014; Tang, Fernandez-Garcia et al. 2014). The NF-kB/IL6/STAT3 signaling pathway generates cancer stem cells in multiple types of breast cancer cells (Iliopoulos, Hirsch et al. 2009; Iliopoulos, Jaeger et al. 2010; Iliopoulos, Hirsch et al. 2011) and is also implicated in colon and other cancers (Iliopoulos, Jaeger et al. 2010). The inflammation related transcription factor NF-kB also contributes to mammary tumorigenesis and metastasis in PyVt mice, in which mammary tumors are induced by expression of an MMTV-driven oncogene (Connelly, Barham et al. 2011). Interestingly, breast cancer fibroblasts and fibroblasts from common sites of breast cancer metastasis (bone, lung) express IL6. IL6 is required for the growth and tumor promoting effects of these fibroblasts on ER-positive cancer cells in vitro and in vivo. ER-negative breast epithelial cells release autocrine IL6 and may therefore be less dependent on IL6 from fibroblasts, although IL6 also transforms these cells (Sasser, Sullivan et al. 2007; Studebaker, Storci et al. 2008; Iliopoulos, Hirsch et al. 2009).
Inflammation is suspected to play a role in the indirect effects of radiation, in which cells not directly targeted by radiation exhibit effects including DNA damage and RONS (Lorimore and Wright 2003; Mukherjee, Coates et al. 2014; Sprung, Ivashkevich et al. 2015). In addition to the IR-induced release of inflammatory signals that are diffusible and can trigger systemic immune responses, inflammatory factors COX2 and TGF-β are produced in bystander cells that are not directly irradiated but are exposed to irradiated cells or media (Zhou, Ivanov et al. 2005; Zhou, Ivanov et al. 2008; Chai, Calaf et al. 2013; Chai, Lam et al. 2013; Wang, Wu et al. 2015).
Inflammatory factors TGF-β, TNF-a, COX2, and NO are implicated in the RONS (Shao, Folkard et al. 2008; Zhou, Ivanov et al. 2008; Wang, Wu et al. 2015), DNA damage (Dickey, Baird et al. 2009; Han, Chen et al. 2010; Dickey, Baird et al. 2012; Chai, Calaf et al. 2013; Chai, Lam et al. 2013; Wang, Wu et al. 2015) and mutations (Zhou, Ivanov et al. 2005; Zhou, Ivanov et al. 2008) observed in bystander cells and in the appearance of genomic instability (Moore, Marsden et al. 2005; Natarajan, Gibbons et al. 2007; Lorimore, Chrystal et al. 2008; Lorimore, Mukherjee et al. 2011) after IR. Further evidence for inflammation in indirect effects of IR come from tumors arising from mammary epithelial cells transplanted into IR exposed cleared fat pads: inflammation-related genes and pathways are upregulated or enriched in the gene expression patters of these indirectly IR-induced tumors (Nguyen, Oketch-Rabah et al. 2011; Nguyen, Fredlund et al. 2013; Illa-Bochaca, Ouyang et al. 2014).
Evidence in mammary gland
Many of the studies above that link inflammatory signals with increased oxidative activity, senescence, EMT, bystander effects, genomic instability, and tumorigenesis, and metastasis use mammary tissue. Since inflammation-related signals are reported after IR in mammary gland (Barcellos-Hoff, Derynck et al. 1994; Dickey, Baird et al. 2009; Datta, Hyduke et al. 2012; Snijders, Marchetti et al. 2012; Bouchard, Bouvette et al. 2013; Wang, Wu et al. 2015) inflammation likely contributes to many of the effects of IR in this tissue.
Uncertainties or inconsistencies
The effects of inflammation can be both pro and anti-tumorigenic. For example, in addition to TGF-β’s role in EMT, in mammary epithelial cells TGF-β is essential to apoptosis of DNA damaged cells including damage following ionizing radiation (Ewan, Henshall-Powell et al. 2002), thus limiting genomic instability (Maxwell, Fleisch et al. 2008). Inflammatory factors TNF-a and COX2 play a similar role in bone marrow of C57BL/6 mice (Lorimore, Rastogi et al. 2013). By eliminating cells with severe DNA damage and curtailing genomic instability, apoptosis (and therefore TGF-β or TNF-a) limits the appearance of major (possibly carcinogenic) mutations following ionizing radiation. However, apoptosis (and thus TGF-β or TNF-a) can indirectly promote tumorigenesis through compensatory proliferation (Loree, Koturbash et al. 2006; Fogarty and Bergmann 2017).
Genetic background also influences the interaction between inflammation and tumorigenesis. Polymorphisms in inflammatory genes influence susceptibility to intestinal cancer following IR (Elahi, Suraweera et al. 2009). In the SPRET outbred mouse higher baseline TGF-β during development decreases tumor incidence following lower doses of IR (0.1 Gy), possibly by reducing ductal branching and susceptibility (Zhang, Lo et al. 2015). Conversely, the BALB/c mouse susceptible to mammary tumors after IR has a lower baseline TGF-β (and a polymorphism in a DNA damage repair-related gene). Early (4 hours) after low dose (0.075 Gy) IR BALB/c mice have suppressed immune pathways and macrophage response but increased IL6, COX2, and TGF-β pathway activation in mammary gland compared to the tumor-resistant C57BL/6 mouse (Snijders, Marchetti et al. 2012; Bouchard, Bouvette et al. 2013). By 1 week after IR, the BALB/c mice show TGF-β -dependent inflammation in the mammary gland, and by 1 month after IR, their mammary glands show proliferation (Nguyen, Martinez-Ruiz et al. 2011; Snijders, Marchetti et al. 2012), suggesting that TGF-β is associated with inflammation, proliferation, and mammary tumorigenesis in these mice. Consistent with this pattern, BALB/c mice that are heterozygous for TGF-β are more resistant to mammary tumorigenesis following IR (Nguyen, Oketch-Rabah et al. 2011). However, the BALB/c mouse also has a polymorphism in a DNA repair gene associated with IR-induced genomic instability (Yu, Okayasu et al. 2001), making it difficult to distinguish potentially overlapping mechanisms.
While inflammatory signals are associated with bystander effects including DNA damage, genomic instability, and mutation, these effects vary between organs in vivo (Chai, Calaf et al. 2013; Chai, Lam et al. 2013), by genotype (Coates, Rundle et al. 2008; Lorimore, Chrystal et al. 2008; Lorimore, Mukherjee et al. 2011), and by cell type (Chai, Calaf et al. 2013). Further research will be required to identify all the underlying factors determining differences in bystander effects, but one variable is the appearance of a protective apoptotic response to cytokines under some conditions (Lorimore, Mukherjee et al. 2011; Lorimore, Rastogi et al. 2013).
One major piece of conflicting evidence comes from a direct test of the essentiality of inflammation to IR-induced carcinogenesis. In a mouse model of lymphoma, a mutation preventing the PIDD/NEMO dependent activation of NF-kB blocks early IR-induced activation of NF-kB (4-24 h) and production of TNF-a (5-48 h) but not lymphoma, suggesting that activation of these inflammatory factors is not essential in this time period (Bock, Krumschnabel et al. 2013). However, this study examined only day one post-IR time points for NF-kB activity, and did not block production of IL6. Later activation of NF-kB or activation of other inflammation-related factors including IL6 and TGF-β could therefore potentially have contributed to lymphoma.
KE: Increase in RONS
Essentiality is High. The most significant support comes from the relatively large number of studies using antioxidants or other interventions to reduce RONS, which show a reduction in DNA damage and mutations. Additional support comes from experiments increasing external oxidants like H2O2, which show that RONS are independently capable of causing DNA damage and mutations. Uncertainties arise from the smaller effects of RONS on DNA damage compared with ionizing radiation. Mammary gland relevance is less certain due to the relatively few experiments in breast tissue.
Multiple studies support the hypothesis that elevated RONS is a key part of the adverse outcome pathway for breast cancer from ionizing radiation. The strongest evidence comes from studies showing that reducing RONS also reduces DNA damage in irradiated cells and bystander cells, including genomic instability observed at later time points after IR. Free radical and NADPH oxidase inhibitors reduce the effect of IR on DNA nucleotide damage, double strand breaks, chromosomal damage, and mutations in isolated DNA and cultured cells (Winyard, Faux et al. 1992; Douki, Ravanat et al. 2006; Choi, Kang et al. 2007; Jones, Riggs et al. 2007; Ameziane-El-Hassani, Boufraqech et al. 2010; Ameziane-El-Hassani, Talbot et al. 2015; Manna, Das et al. 2015) and on nucleotide damage and double strand breaks in vivo (Pazhanisamy, Li et al. 2011; Ozyurt, Cevik et al. 2014). RONS reduction after ionizing radiation also reduces genomic instability in animals and in cloned cell lines (Dayal, Martin et al. 2008; Dayal, Martin et al. 2009; Pazhanisamy, Li et al. 2011; Bensimon, Biard et al. 2016). RONS are similarly implicated in IR effects in bystander cells. Antioxidants (including a nitric oxide scavenger) and oxidase inhibitors added before or after radiation reduce micronuclei and gamma-H2AX formation in bystander cells (Azzam, De Toledo et al. 2002; Yang, Asaad et al. 2005; Yang, Anzenberg et al. 2007). Antioxidant activity also reduces the inflammatory response to IR in animals and cultured skin cells (Berruyer, Martin et al. 2004; Das, Manna et al. 2014; Ozyurt, Cevik et al. 2014; Haddadi, Rezaeyan et al. 2017; Zhang, Zhu et al. 2017).
RONS are sufficient to trigger subsequent key events in this AOP. Extracellularly applied or intracellularly generated ROS (which also facilitates the formation of RNS) are capable of creating DNA damage in vitro including base damage, single and double strand breaks, and chromosomal damage (Oya, Yamamoto et al. 1986; Dahm-Daphi, Sass et al. 2000; Nakamura, Purvis et al. 2003; Gradzka and Iwanenko 2005; Ismail, Nystrom et al. 2005; Driessens, Versteyhe et al. 2009; Berdelle, Nikolova et al. 2011; Lorat, Brunner et al. 2015; Stanicka, Russell et al. 2015) and mutations (Sandhu and Birnboim 1997; Ameziane-El-Hassani, Boufraqech et al. 2010; Seager, Shah et al. 2012; Sharma, Collins et al. 2016). Similarly, decreased antioxidant activity and higher RONS is observed in cells with genomic instability (Dayal, Martin et al. 2008; Buonanno, de Toledo et al. 2011). To our knowledge, no experiments have tested whether elevating intracellular RONS alone in one group of cells can cause bystander effects in another.
Evidence in Mammary Gland
The increase of RONS following IR has been shown in a wide range of cells, in vivo and in vitro, including epithelial cells, and in two studies in mammary epithelial cells (Jones, Riggs et al. 2007; Bensimon, Biard et al. 2016). Both mammary cell studies also show increased RONS and DNA damage over a day after IR in vitro and link DNA damage with elevated RONS.
Uncertainties or Inconsistencies
The mitigating effects of antioxidants on IR-generated DNA damage support the essentiality of RONS in producing DNA damage and mutations. However, externally applied RONS is less effective than IR at generating double strand breaks and mutations (Sandhu and Birnboim 1997; Dahm-Daphi, Sass et al. 2000; Gradzka and Iwanenko 2005; Ismail, Nystrom et al. 2005). One possible explanation for this discrepancy is that IR may elicit a higher concentration of localized RONS than can be achieved with external application of H2O2. IR deposits energy and oxidizes molecules within a relatively small area over a rapid timescale potentially permitting a very high local concentration which could precede or overwhelm local buffering capacity. In contrast, extracellularly applied H2O2 would interact with many antioxidants and other molecules on its way to the nucleus, where the concentration would slowly reach a lower steady state.
As expected for RONS as a key event for DNA damage from IR, DNA damage from IR and H2O2 are additive in cells (Dahm-Daphi, Sass et al. 2000; Driessens, Versteyhe et al. 2009). Unexpectedly however, inhibiting glutathione (which should increase or sustain the effects of RONS), increases DNA damage from H2O2 but not IR. This lack of effect of glutathione inhibition on IR conflicts with multiple studies showing decreased DNA damage from IR with anti-oxidants. One possible explanation is that the concentration or reaction rate of glutathione is already inadequate to buffer the elevated RONS from IR, so further inhibition has no measurable effect.
Known Modulating Factors
Quantitative Understanding
Considerations for Potential Applications of the AOP (optional)
Because of the long latency of mammary tumors, the two-year rodent carcinogenicity bioassay is the primary assay for the adverse outcome of breast cancer. The assay is included in the OECD Test No. 451 and 453 for carcinogenicity and combined toxicity and carcinogenicity. Mammary tumors are also reported in short term, sub-chronic, and chronic toxicity tests, but these tests are less sensitive due to their shorter duration.
This AOP is relevant to guideline tests addressing DNA damage and mutation. MIE2: Increase in DNA damage is relevant to OECD Test Nos. 473, 475, 483, 487, and 489, which detect DNA damage in the form of single and double strand breaks, chromosomal damage and micronuclei, as well as some forms of nucleotide damage. KE1: Increase in mutation is relevant to OECD Test Nos. 471, 476, 488, and 490 for in vitro and in vivo mutations. To our knowledge no guideline tests address increases in RONS, proliferation, or inflammation, although some in vitro tests in ToxCast or in development elsewhere may reflect changes in these key events.
